Abstract
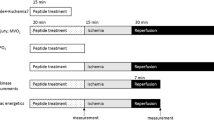
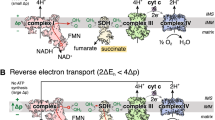
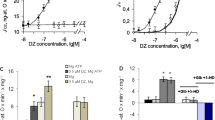
Diazoxide, a mitochondrial ATP-sensitive potassium (mitoKATP) channel opener, protects the heart from ischemia–reperfusion injury. Diazoxide also inhibits mitochondrial complex II-dependent respiration in addition to its preconditioning effect. However, there are no prior studies of the role of diazoxide on post-ischemic myocardial oxygenation. In the current study, we determined the effect of diazoxide on the suppression of post-ischemic myocardial tissue hyperoxygenation in vivo, superoxide (O2 −•) generation in isolated mitochondria, and impairment of the interaction between complex II and complex III in purified mitochondrial proteins. It was observed that diazoxide totally suppressed the post-ischemic myocardial hyperoxygenation. With succinate but not glutamate/malate as the substrate, diazoxide significantly increased ubisemiquinone-dependent O2 −• generation, which was not blocked by 5-HD and glibenclamide. Using a model system, the super complex of succinate-cytochrome c reductase (SCR) hosting complex II and complex III, we also observed that diazoxide impaired complex II and its interaction with complex III with no effect on complex III. UV–visible spectral analysis revealed that diazoxide decreased succinate-mediated ferricytochrome b reduction in SCR. In conclusion, our results demonstrated that diazoxide suppressed the in vivo post-ischemic myocardial hyperoxygenation through opening the mitoKATP channel and ubisemiquinone-dependent O2 −• generation via inhibiting mitochondrial complex II-dependent respiration.








Similar content being viewed by others
Abbreviations
- DCPIP:
-
Dichlorophenolindophenol
- DEPMPO:
-
5-(Diethoxyphosphoryl)-5-methyl-1-pyrroline N-oxide
- DMPO:
-
5,5-Dimethyl-1-pyrroline N-oxide
- DTPA:
-
Diethylenetriaminepentaacetic acid
- EPR:
-
Electron paramagnetic resonance
- FCCP:
-
Carbonyl cyanide-p-tri-fluromethoxyphenyl-hydrazone
- 5-HD:
-
5-hydroxydecanoate
- H2DCF:
-
2′,7′-dichlorodihydrofluorescein
- IPC:
-
Ischemic preconditioning
- mitoKATP :
-
Mitochondrial ATP-sensitive potassium channel
- 3-NPA:
-
3-Nitropropionic acid
- O2 −• :
-
Superoxide
- •OH:
-
Hydroxyl radical
- \( P_{{{\text{O}}_{ 2} }} \) :
-
Tissue oxygen tension
- PPC:
-
Pharmacological preconditioning
- Q:
-
Ubiquinone
- Q−• :
-
Ubisemiquinone
- Q2 :
-
Ubiquinone-2
- QCR:
-
Ubiquinol cytochrome c reductase or complex III
- QH2 :
-
Ubiquinol
- QO −• :
-
Ubisemiquinone at the QO site
- ROS:
-
Reactive oxygen species
- SCR:
-
Succinate-cytochrome c reductase or super complex containing complex II and complex III
- SMP:
-
Sub-mitochondrial particles
- SOD:
-
Superoxide dismutase
- SQR:
-
Succinate ubiquinone reductase or complex II
- TEA+ :
-
Tetraethyl ammonium
References
Murry CE, Jennings RB, Reimer KA (1986) Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation 74:1124–1136
Yellon DM, Downey JM (2003) Preconditioning the myocardium: from cellular physiology to clinical cardiology. Physiol Rev 83:1113–1151
Zhu X, Liu B, Zhou S et al (2007) Ischemic preconditioning prevents in vivo hyperoxygenation in postischemic myocardium with preservation of mitochondrial oxygen consumption. Am J Physiol Heart Circ Physiol 293:H1442–H1450
Carroll R, Gant VA, Yellon DM (2001) Mitochondrial K(ATP) channel opening protects a human atrial-derived cell line by a mechanism involving free radical generation. Cardiovasc Res 51:691–700
Ozcan C, Holmuhamedov EL, Jahangir A et al (2001) Diazoxide protects mitochondria from anoxic injury: implications for myopreservation. J Thorac Cardiovasc Surg 121:298–306
Przyklenk K, Maynard M, Whittaker P (2006) First molecular evidence that inositol trisphosphate signaling contributes to infarct size reduction with preconditioning. Am J Physiol Heart Circ Physiol 291:H2008–H2012
Schafer G, Wegener C, Portenhauser R et al (1969) Diazoxide, an inhibitor of succinate oxidation. Biochem Pharmacol 18:2678–2681
Ovide-Bordeaux S, Ventura-Clapier R, Veksler V (2000) Do modulators of the mitochondrial K(ATP) channel change the function of mitochondria in situ? J Biol Chem 275:37291–37295
Hanley PJ, Mickel M, Loffler M et al (2002) K(ATP) channel-independent targets of diazoxide and 5-hydroxydecanoate in the heart. J Physiol 542:735–741
Lim KH, Javadov SA, Das M et al (2002) The effects of ischaemic preconditioning, diazoxide and 5-hydroxydecanoate on rat heart mitochondrial volume and respiration. J Physiol 545:961–974
Drose S, Brandt U, Hanley PJ (2006) K+-independent actions of diazoxide question the role of inner membrane KATP channels in mitochondrial cytoprotective signaling. J Biol Chem 281:23733–23739
Minners J, Lacerda L, Yellon DM et al (2007) Diazoxide-induced respiratory inhibition—a putative mitochondrial K(ATP) channel independent mechanism of pharmacological preconditioning. Mol Cell Biochem 294:11–18
Bacsi A, Woodberry M, Widger W et al (2006) Localization of superoxide anion production to mitochondrial electron transport chain in 3-NPA-treated cells. Mitochondrion 6:235–244
Gomez-Lazaro M, Galindo MF, Melero-Fernandez de Mera RM et al (2007) Reactive oxygen species and p38 mitogen-activated protein kinase activate Bax to induce mitochondrial cytochrome c release and apoptosis in response to malonate. Mol Pharmacol 71:736–743
Turrens JF (2003) Mitochondrial formation of reactive oxygen species. J Physiol 552:335–344
Swartz HM, Bacic G, Friedman B et al (1994) Measurements of pO2 in vivo, including human subjects, by electron paramagnetic resonance. Adv Exp Med Biol 361:119–128
Friedman BJ, Grinberg OY, Grinberg SA et al (1997) Myocardial oxygen tension in isolated erythrocyte-perfused rat hearts and comparison with crystalloid media. J Mol Cell Cardiol 29:2855–2858
Zhao X, He G, Chen YR et al (2005) Endothelium-derived nitric oxide regulates postischemic myocardial oxygenation and oxygen consumption by modulation of mitochondrial electron transport. Circulation 111:2966–2972
Khan M, Iyyapu KM, Kutala V et al (2008) Sulfaphenazole protects heart against ischemia–reperfusion injury and cardiac dysfunction by overexpression of iNOS leading to enhancement of nitric-oxide bioavailability and tissue oxygenation. Antioxid Redox Signal 11:725–738
Villamena FA, Zweier JL (2004) Detection of reactive oxygen and nitrogen species by EPR spin trapping. Antioxid Redox Signal 6:619–629
Quast U, Cook NS (1989) In vitro and in vivo comparison of two K+ channel openers, diazoxide and cromakalim, and their inhibition by glibenclamide. J Pharmacol Exp Ther 250:261–271
Xu Y, Liu B, Zweier JL et al (2008) Formation of hydrogen peroxide and reduction of peroxynitrite via dismutation of superoxide at reperfusion enhances myocardial blood flow and oxygen consumption in postischemic mouse heart. J Pharmacol Exp Ther 327:402–410
Costa AD, Quinlan CL, Andrukhiv A et al (2006) The direct physiological effects of mitoK(ATP) opening on heart mitochondria. Am J Physiol Heart Circ Physiol 290:H406–H415
Dzeja PP, Bast P, Ozcan C et al (2003) Targeting nucleotide-requiring enzymes: implications for diazoxide-induced cardioprotection. Am J Physiol Heart Circ Physiol 284:H1048–H1056
Ferranti R, da Silva MM, Kowaltowski AJ (2003) Mitochondrial ATP-sensitive K+ channel opening decreases reactive oxygen species generation. FEBS Lett 536:51–55
Han D, Antunes F, Canali R et al (2003) Voltage-dependent anion channels control the release of the superoxide anion from mitochondria to cytosol. J Biol Chem 278:5557–5563
Rigobello MP, Folda A, Scutari G et al (2005) The modulation of thiol redox state affects the production and metabolism of hydrogen peroxide by heart mitochondria. Arch Biochem Biophys 441:112–122
Chen YR, Chen CL, Pfeiffer DR et al (2007) Mitochondrial complex II in the post-ischemic heart: oxidative injury and the role of protein S-glutathionylation. J Biol Chem 282:32640–32654
Lowry OH, Rosebrough NJ, Farr AL et al (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Boveris A, Cadenas E (1975) Mitochondrial production of superoxide anions and its relationship to the antimycin insensitive respiration. FEBS Lett 54:311–314
Salvemini D, Wang ZQ, Zweier JL et al (1999) A nonpeptidyl mimic of superoxide dismutase with therapeutic activity in rats. Science 286:304–306
Iqbal J, Whitney P (1991) Use of cyanide and diethyldithiocarbamate in the assay of superoxide dismutases. Free Radic Biol Med 10:69–77
Chen YR, Chen CL, Yeh A et al (2006) Direct and indirect roles of cytochrome b in the mediation of superoxide generation and NO catabolism by mitochondrial succinate-cytochrome c reductase. J Biol Chem 281:13159–13168
Sato T, O’Rourke B, Marban E (1998) Modulation of mitochondrial ATP-dependent K+ channels by protein kinase C. Circ Res 83:110–114
Hu H, Sato T, Seharaseyon J et al (1999) Pharmacological and histochemical distinctions between molecularly defined sarcolemmal KATP channels and native cardiac mitochondrial KATP channels. Mol Pharmacol 55:1000–1005
Han D, Williams E, Cadenas E (2001) Mitochondrial respiratory chain-dependent generation of superoxide anion and its release into the intermembrane space. Biochem J 353:411–416
Frejaville C, Karoui H, Tuccio B et al (1995) 5-(Diethoxyphosphoryl)-5-methyl-1-pyrroline N-oxide: a new efficient phosphorylated nitrone for the in vitro and in vivo spin trapping of oxygen-centered radicals. J Med Chem 38:258–265
Holmuhamedov EL, Jahangir A, Oberlin A et al (2004) Potassium channel openers are uncoupling protonophores: implication in cardioprotection. FEBS Lett 568:167–170
Kopustinskiene DM, Jovaisiene J, Liobikas J et al (2002) Diazoxide and pinacidil uncouple pyruvate-malate-induced mitochondrial respiration. J Bioenerg Biomembr 34:49–53
Andrukhiv A, Costa AD, West IC et al (2006) Opening mitoKATP increases superoxide generation from complex I of the electron transport chain. Am J Physiol Heart Circ Physiol 291:H2067–H2074
Velayutham M, Muthukumaran RB, Sostaric JZ et al (2007) Interactions of the major metabolite of the cancer chemopreventive drug oltipraz with cytochrome c: a novel pathway for cancer chemoprevention. Free Radic Biol Med 43:1076–1085
Halliwell B, Whiteman M (2004) Measuring reactive species and oxidative damage in vivo and in cell culture: how should you do it and what do the results mean? Br J Pharmacol 142:231–255
Dikalov S, Griendling KK, Harrison DG (2007) Measurement of reactive oxygen species in cardiovascular studies. Hypertension 49:717–727
Fernandes MA, Santos MS, Moreno AJ et al (2004) Glibenclamide interferes with mitochondrial bioenergetics by inducing changes on membrane ion permeability. J Biochem Mol Toxicol 18:162–169
Engbersen R, Masereeuw R, van Gestel MA et al (2005) Glibenclamide depletes ATP in renal proximal tubular cells by interfering with mitochondrial metabolism. Br J Pharmacol 145:1069–1075
Cadenas E, Boveris A (1980) Enhancement of hydrogen peroxide formation by protophores and ionophores in antimycin-supplemented mitochondria. Biochem J 188:31–37
Brookes PS (2005) Mitochondrial H(+) leak and ROS generation: an odd couple. Free Radic Biol Med 38:12–23
Lesnefsky EJ, Gudz TI, Migita CT et al (2001) Ischemic injury to mitochondrial electron transport in the aging heart: damage to the iron-sulfur protein subunit of electron transport complex III. Arch Biochem Biophys 385:117–128
Petrosillo G, Ruggiero FM, Di Venosa N et al (2003) Decreased complex III activity in mitochondria isolated from rat heart subjected to ischemia and reperfusion: role of reactive oxygen species and cardiolipin. Faseb J 17:714–716
Becker LB, vanden Hoek TL, Shao ZH et al (1999) Generation of superoxide in cardiomyocytes during ischemia before reperfusion. Am J Physiol 277:H2240–H2246
Acknowledgments
This work was supported by American Heart Association Grant 0435299N and National Heart, Lung, and Blood Institute Grants HL-081630 (He) and HL-083237 (Chen). The LiPc was made with the support of PO1 EB2180 (Swartz).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, B., Zhu, X., Chen, CL. et al. Opening of the mitoKATP channel and decoupling of mitochondrial complex II and III contribute to the suppression of myocardial reperfusion hyperoxygenation. Mol Cell Biochem 337, 25–38 (2010). https://doi.org/10.1007/s11010-009-0283-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-009-0283-2