Abstract
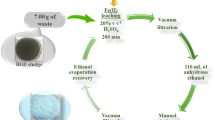
Flotation tailings of the zinc processing plant contain a high amount of iron. Recovery of iron from these tailings is a promising approach to reduce process waste and provide the steel industry's raw material. This study investigated the reduction kinetics of self-reducing briquettes composed of fine hematite particles and iron concentrate; a material obtained from processing zinc tailing by magnetic separation. The investigation was performed using the forced stepwise isothermal analysis method, and mass loss data from 973 to 1373 K was provided by the reduction of the briquettes in a thermogravimetric scale. The addition of iron concentrate as part of the raw material showed an increase in the reduction rate compared to briquettes composed of pure hematite. In the temperature range of 973–1073 K, the reduction was controlled by nucleation, with an activation energy of 327.60 kJ mol−1, and from 1123 to 1223 K by diffusion, with an activation energy of 402.3–665.0 kJ mol−1. For a batch process, as performed in the TG analysis, the gasification of charcoal was a step that increased the apparent activation energy of the process.







Similar content being viewed by others
References
Lei C, Yan B, Chen T, Quan SX, Xiao XM. Comprehensive utilization of lead-zinc tailings, part 1: pollution characteristics and resource recovery of sulfur. J Environ Chem Eng. 2015;3:862–9.
Sidle RC, Chambers JC, Amacher MC. Fate of heavy metals in an abandoned lead-zinc tailings pond: II. Sediment J Environ Qual. 1991;20:752–8.
Ye ZH, Shu WS, Zhang ZQ, Lan CY, Wong MH. Evaluation of major constraints to revegetation of lead/zinc mine tailings using bioassay techniques. Chemosphere. 2002;47:1103–11.
Húngaro RC, Vinhal JT, Espinosa DCR, Tenório JAS. Caracterização do resíduo da flotação do minério de zinco. 20° Simpósio Mineração. Editora Blucher; 2019. p. 17–25.
Vinhal JT, Húngaro RC, Espinosa DCR, Tenório JAS (2019) Caracterização de rejeito proveniente do processo de flotação do minério willemítico. XXVIII Encontro Nac Trat Minérios e Metal Extrativa, pp 1–8
Junca E, Restivo TAG, Espinosa DCR, Tenório JAS. Application of stepwise isothermal analysis method in the kinetic study of reduction of basic oxygen furnace dust. J Therm Anal Calorim. 2015;120:1913–9.
Gotor FJ, Perez-Maqueda LA, Ortega A, Criado JM. Kinetic analysis of solid state reactions by means of stepwise isothermal analysis (SIA) and constant rate thermal analysis (crta)—a compartive study. J Therm Anal. 1998;53:389–96.
Tiernan MJ, Barnes PA, Parkes GMB. Reduction of iron oxide catalysts: the investigation of kinetic parameters using rate perturbation and linear heating thermoanalytical techniques. J Phys Chem B. 2001;105:220–8.
Sørensen OT, Rouquerol J. Sample controlled thermal analysis origin, goals, multiple forms, applications and future. In: Sorensen OT, Rouquerol J, editors. Hot top thermal analysis calorimeter. US: Springer; 2003.
Chen F, Sørensen OT, Meng G, Peng D. Thermal decomposition of BaC2O·0.5H2O studied by stepwise isothermal analysis and non-isothermal thermogravimetry. J Therm Anal Calorim. 1998;53:397–410.
Sørensen OT. Thermogravimetric and dilatometric studies using stepwise isothermal analysis and related techniques. J Therm Anal. 1992;38:213–28.
Husum PL, Sørensen OT. Computer controlled forced stepwise isothermal analysis. Thermochim Acta. 1987;114:131–8.
Tenório Vinhal J, Costa Húngaro R, Luís Coleti J, Espinosa DCR. Iron recovery from zinc mine tailings by magnetic separation followed by carbothermal reduction of self-reducing briquettes. Can J Chem Eng. 2020;99:166–77.
Esezobor DE, Balogun SA. Zinc accumulation during recycling of iron oxide wastes in the blast furnace. Ironmak Steelmak. 2013;33:419–25.
Coleti JL, Manfredia GVP, Vinhal JT, Junca E, Espinosa DCR, Tenório JAS. Kinetic investigation of self-reduction basic oxygen furnace dust briquettes using charcoals from different biomass. J Mater Res Technol. 2020;9:13282–93.
Roduit B, Maciejewski M, Baiker A. Influence of experimental conditions on the kinetic parameters of gas-solid reactions—parametric sensitivity of thermal analysis. Thermochim Acta. 1996;282–283:101–19.
Abraham MC, Ghosh A (1973) Kinetics of reduction of iron oxides and ores by carbon. pp 107–116
Hunt J, Ferrari A, Lita A, Crosswhite M, Ashley B, Stiegman AE. Microwave-specific enhancement of the carbon-carbon dioxide (boudouard) reaction. J Phys Chem C Am Chem Soc. 2013;117:26871–80.
Rat’Ko AI, Ivanets AI, Kulak AI, Morozov EA, Sakhar IO. Thermal decomposition of natural dolomite. Inorg Mater. 2011;47:1372–7.
Junca E, Restivo TAG, de Oliveira JR, Espinosa DCR, Tenório JAS. Reduction of electric arc furnace dust pellets by simulated reformed natural gas. J Therm Anal Calorim. 2016;126:1889–97.
Pineau A, Kanari N, Gaballah I. Kinetics of reduction of iron oxides by H2: part I: low temperature reduction of hematite. Thermochim Acta. 2006;447:89–100.
Wang G, Zhang J, Zhang G, Wang H, Zhao D. Experiments and kinetic modeling for reduction of ferric oxide-biochar composite pellets. ISIJ Int. 2017;57:1374–83.
Spreitzer D, Schenk J. Reduction of iron oxides with hydrogen—a review. Steel Res Int. 2019;90:1900108.
Marsh H, Kuo K. Kinetics and catalysis of carbon gasification. Introduction to carbon science. Elsevier; 1989. p. 107–51.
Struis RPWJ, Von Scala C, Stucki S, Prins R. Gasification reactivity of charcoal with CO2. Part I: Conversion and structural phenomena. Chem Eng Sci. 2002;57:3581–92.
Guo D, Hu M, Pu C, Xiao B, Hu Z, Liu S, et al. Kinetics and mechanisms of direct reduction of iron ore-biomass composite pellets with hydrogen gas. Int J Hydrogen Energy. 2015;40:4733–40.
Kowitwarangkul P, Babich A, Senk D. Reduction kinetics of self-reducing pellets of iron ore. AISTech Proc. 2014;1:611–22.
He K, Zheng Z, Chen Z. Multistep reduction kinetics of Fe3O4 to Fe with CO in a micro fluidized bed reaction analyzer. Powder Technol. 2020;360:1227–36.
Man Y, Xiao FJ. Effect of iron ore-coal pellets during reduction with hydrogen and carbon monoxide. Powder Technol. 2016;301:1213–7.
Acknowledgements
The authors acknowledge the FAPESP grants 2012/51871-9, Sao Paulo Research Foundation (FAPESP), for the financial support. This work was also supported by a CAPES/BRAZIL EMBRAPII scholarship. The information presented in this study are those of the authors and do not necessarily reflect the opinion of FAPESP, CAPES or EMBRAPII.
Funding
Fundação de Amparo à Pesquisa do Estado de São Paulo, 2012/51871-9, Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, 88887.507766/2020-00, Jonathan Tenório Vinhal.
Author information
Authors and Affiliations
Contributions
JTV: conceptualization, validation, formal analysis, data curation, writing and editing—original draft; JLC: validation, writing—reviewing; LKT: conceptualization, validation, supervision, writing—reviewing and editing. DCRE: validation, supervision, writing—reviewing and editing.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vinhal, J.T., Coleti, J.L., Khajavi, L.T. et al. Kinetic investigation on the reduction of iron concentrate-charcoal composite briquette by forced stepwise isothermal analysis (FSIA). J Therm Anal Calorim 147, 14449–14458 (2022). https://doi.org/10.1007/s10973-022-11743-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-022-11743-4