Abstract
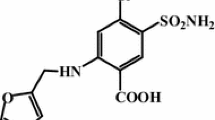
The current study was performed to investigate possible interactions between triazole antifungal drug itraconazole (ITR) with selected excipients commonly used for development of solid lipid nanoparticles. The excipients included common lipids (glyceryl behenate (Compritol 888 ATO™), glyceryl monostearate, stearic acid, and cetyl palmitate), charge inducers (dicetyl phosphate and stearlyamine), and surfactants (sodium cholate and sodium deoxycholate). Differential scanning calorimetry, isothermal stress testing, Fourier transform infrared spectral analysis, optical microcopy, and X-ray powder diffraction analysis were performed for assessing the compatibility between the drug and the excipients. Results of the study suggest that the stearic acid exhibited drug–excipient interactions, whereas all other excipients used in the study were found to be compatible with ITR.












Similar content being viewed by others
References
Verma RK, Garg S. Selection of excipients for extended release formulations of glipizide through drug–excipient compatibility testing. J Pharm Biomed Anal. 2005;38:633–44.
Verma RK, Garg S. Compatibility studies between isosorbide mononitrate and selected excipients used in the development of extended release formulations. J Pharm Biomed Anal. 2004;35:449–58.
Pani NR, Nath LK, Acharya S. Compatibility studies of nateglinide with excipients in immediate release tablets. Acta Pharm. 2011;61:237–47.
Abbas D, Kaloustian J, Orneto C, Piccerelle P, Portugal H, Nicolay A. DSC and physico-chemical properties of a substituted pyridoquinoline and its interaction study with excipients. J Therm Anal Calorim. 2008;93(2):353–60. doi:10.1007/s10973-008-9062-7.
Drebushchak VA, Shakhtshneider TP, Apenina SA, Medvedeva AS, Safronova LP, Boldyrev VV. Thermoanalytical investigation of drug–excipient interaction. J Therm Anal Calorim. 2006;86(2):303–9. doi:10.1007/s10973-005-7440-y.
Pani N, Nath L, Acharya S, Bhuniya B. Application of DSC, IST, and FTIR study in the compatibility testing of nateglinide with different pharmaceutical excipients. J Therm Anal Calorim. 2012;108(1):219–26. doi:10.1007/s10973-011-1299-x.
Bruni G, Berbenni V, Milanese C, Girella A, Marini A. Drug–excipient compatibility studies in binary and ternary mixtures by physico-chemical techniques. J Therm Anal Calorim. 2010;102(1):193–201. doi:10.1007/s10973-009-0382-z.
Marini A, Berbenni V, Pegoretti M, Bruni G, Cofrancesco P, Sinistri C, et al. Drug–excipient compatibility studies by physico-chemical techniques: the case of atenolol. J Therm Anal Calorim. 2003;73(2):547–61. doi:10.1023/a:1025478129417.
Tita D, Jurca T, Fulias A, Marian E, Tita B. Compatibility study of the acetylsalicylic acid with different solid dosage forms excipients. J Therm Anal Calorim. 2013;112(1):407–19. doi:10.1007/s10973-013-2937-2.
De Beule K, Van Gestel J. Pharmacology of itraconazole. Drugs. 2001;61(S1):27–37.
Dobaria N, Badhan A, Mashru R. A novel itraconazole bioadhesive film for vaginal delivery: design, optimization, and physicodynamic characterization. AAPS PharmSciTech. 2009;10(3):951–9. doi:10.1208/s12249-009-9288-0.
Heykants J, Van Peer A, Van de Velde V, Van Rooy P, Meuldermans W, Lavrijsen K, et al. The clinical pharmacokinetics of itraconazole: an overview. Mycoses. 1989;32:67–87. doi:10.1111/j.1439-0507.1989.tb02296.x.
Buchanan CM, Buchanan NL, Edgar KJ, Klein S, Little JL, Ramsey MG, et al. Pharmacokinetics of itraconazole after intravenous and oral dosing of itraconazole-cyclodextrin formulations. J Pharm Sci. 2007;96(11):3100–16. doi:10.1002/jps.20878.
Barone JA, Moskovitz BL, Guarnieri J, Hassell AE, Colaizzi JL, Bierman RH, et al. Food interaction and steady-state pharmacokinetics of itraconazole oral solution in healthy volunteers. Pharmacotherapy. 1998;18(2):295–301.
Singh AK, Nath LK. Evaluation of compatibility of tablet excipients and novel synthesized polymer with lamivudine. J Therm Anal Calorim. 2012;108:263–7.
Kumar N, Shishu, Bansal G, Kumar S, Jana AK. Preparation and cyclodextrin assisted dissolution rate enhancement of itraconazolium dinitrate salt. Drug Dev Ind Pharm. 2012;. doi:10.3109/03639045.2012.681382.
Kumar N, Shishu, Bansal G, Kumar S, Jana AK. Ditosylate salt of itraconazole and dissolution enhancement using cyclodextrins. AAPS PharmSciTechnol. 2012;. doi:10.1208/s12249-012-9804-5.
Kumar N, Shishu, Kapoor VR. Facile syntheses of novel salts of a triazole antifungal agent with enhanced solubility. J Heterocycl Chem. 2012;. doi:10.1002/jhet.1120.
DiNunzio JC, Brough C, Miller DA, Williams RO, McGinity JW. Fusion processing of itraconazole solid dispersions by kinetisol® dispersing: a comparative study to hot melt extrusion. J Pharm Sci. 2010;99(3):1239–53. doi:10.1002/jps.21893.
Six K, Daems T, de Hoon J, Van Hecken A, Depre M, Bouche M-P, et al. Clinical study of solid dispersions of itraconazole prepared by hot-stage extrusion. Eur J Pharm Sci. 2005;24(2–3):179–86.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, N., Goindi, S., Saini, B. et al. Thermal characterization and compatibility studies of itraconazole and excipients for development of solid lipid nanoparticles. J Therm Anal Calorim 115, 2375–2383 (2014). https://doi.org/10.1007/s10973-013-3237-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-013-3237-6