Abstract
The understanding of the thermal stability of zinc carbonates and the relative stability of hydrous carbonates including hydrozincite and hydromagnesite is extremely important to the sequestration process for the removal of atmospheric CO2. The hydration-carbonation or hydration-and-carbonation reaction path in the ZnO-CO2-H2O system at ambient temperature and atmospheric CO2 is of environmental significance from the standpoint of carbon balance and the removal of green house gases from the atmosphere.
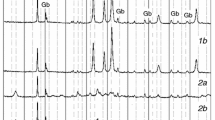
The dynamic thermal analysis of hydrozincite shows a 22.1% mass loss at 247°C. The controlled rate thermal analysis (CRTA) pattern of hydrozincite shows dehydration at 38°C, some dehydroxylation at 170°C and dehydroxylation and decarbonation in a long isothermal step at 190°C. The CRTA pattern of smithsonite shows a long isothermal decomposition with loss of CO2 at 226°C. CRTA technology offers better resolution and a more detailed interpretation of the decomposition processes of zinc carbonate minerals via approaching equilibrium conditions of decomposition through the elimination of the slow transfer of heat to the sample as a controlling parameter on the process of decomposition. The CRTA technology offers a mechanism for the study of the thermal decomposition and relative stability of minerals such as hydrozincite and smithsonite.
Similar content being viewed by others
References
J. W. Anthony, R. A. Bideaux, K. W. Bladh and M. C. Nichols, Handbook of Mineralogy, Mineral Data Publishing, Tiscon, Arizona, USA 2003.
V. C. Farmer, Mineralogical Society Monograph 4: The Infrared Spectra of Minerals, 1974.
M. Bouchard and D. C. Smith, Asian Chem. Lett., 5 (2001) 157.
M. M. Harding, B. M. Kariuki, R. Cernik and G. Cressey, Acta Crystallogr., Section B: Structural Sci., B50 (1994) 673.
W. Zabinski, Can. Mineral., 8 (1966) 649.
S. Ghose, Acta Cryst., 17 (1964) 1051.
A. K. Alwan and P. A. Williams, Transition Metal Chem. (Dordrecht, Netherlands), 4 (1979) 128.
P. A. Williams, Oxide Zone Geochemistry, Ellis Horwood Ltd., Chichester, West Sussex, England 1990.
J. L. Jambor, Can. Mineral., 8 (1964) 92.
F. Zhu, D. Persson and D. Thierry, Corrosion (Houston, TX, United States), 57 (2001) 582.
R. L. Frost, S. J. Palmer, J. M. Bouzaid and B. J. Reddy, J. Raman Spectrosc., 38 (2007) 68.
R. L. Frost, D. A. Henry, M. L. Weier and W. Martens, J. Raman Spectrosc., 37 (2006) 722.
R. L. Frost, A. W. Musumeci, J. T. Kloprogge, M. O. Adebajo and W. N. Martens, J. Raman Spectrosc., 37 (2006) 733.
R. L. Frost, J. Cejka, M. Weier and W. N. Martens, J. Raman Spectrosc., 37 (2006) 879.
R. L. Frost, M. L. Weier, J. Cejka and J. T. Kloprogge, J. Raman Spectrosc., 37 (2006) 585.
R. L. Frost, J. Cejka, M. L. Weier and W. Martens, J. Raman Spectrosc., 37 (2006) 538.
R. L. Frost, M. L. Weier, B. J. Reddy and J. Cejka, J. Raman Spectrosc., 37 (2006) 816.
R. L. Frost, M. L. Weier, W. N. Martens, J. T. Kloprogge and J. Kristóf, J. Raman Spectrosc., 36 (2005) 797.
R. L. Frost, J. Raman Spectrosc., 37 (2006) 910.
R. L. Frost, R.-A. Wills, M. L. Weier and W. Martens, J. Raman Spectrosc., 36 (2005) 435.
R. L. Frost, A. W. Musumeci, W. N. Martens, M. O. Adebajo and J. Bouzaid, J. Raman Spectrosc., 36 (2005) 925.
R. L. Frost and M. L. Weier, J. Therm. Anal. Cal., 75 (2004) 277.
R. L. Frost, K. Erickson and M. Weier, J. Therm. Anal. Cal., 77 (2004) 851.
R. L. Frost, S. J. Mills and K. L. Erickson, Thermochim. Acta, 419 (2004) 109.
R. L. Frost, M. L. Weier and W. Martens, J. Therm. Anal. Cal., 82 (2005) 373.
R. L. Frost, M. L. Weier, W. Martens and S. Mills, J. Mol. Struct., 752 (2005) 178.
R. L. Frost, A. W. Musumeci, J. Bouzaid, M. O. Adebajo, W. N. Martens and J. T. Kloprogge, J. Solid State Chem., 178 (2005) 1940.
J. Bouzaid and R. L. Frost, J. Therm. Anal. Cal., 89 (2007) 133.
J. M. Bouzaid, R. L. Frost and W. N. Martens, J. Therm. Anal. Cal., 89 (2007) 511.
J. M. Bouzaid, R. L. Frost, A. W. Musumeci and W. N. Martens, J. Therm. Anal. Cal., 86 (2006) 745.
R. L. Frost, J. M. Bouzaid, A. W. Musumeci, J. T. Kloprogge and W. N. Martens, J. Therm. Anal. Cal., 86 (2006) 437.
R. L. Frost, J. Kristóf, W. N. Martens, M. L. Weier and E. Horváth, J. Therm. Anal. Cal., 83 (2006) 675.
R. L. Frost, J. Kristóf, M. L. Weier, W. N. Martens and E. Horváth, J. Therm. Anal. Cal., 79 (2005) 721.
R. L. Frost, A. W. Musumeci, M. O. Adebajo and W. Martens, J. Therm. Anal. Cal., 89 (2007) 95.
R. L. Frost, A. W. Musumeci, J. T. Kloprogge, M. L. Weier, M. O. Adebajo and W. Martens, J. Therm. Anal. Cal., 86 (2006) 205.
R. L. Frost, M. L. Weier and W. Martens, J. Therm. Anal. Cal., 82 (2005) 115.
R. L. Frost, R.-A. Wills, J. T. Kloprogge and W. Martens, J. Therm. Anal. Cal., 84 (2006) 489.
R. L. Frost, R.-A. Wills, J. T. Kloprogge and W. N. Martens, J. Therm. Anal. Cal., 83 (2006) 213.
A. W. Musumeci, G. G. Silva, W. N. Martens, E. R. Waclawik and R. L. Frost, J. Therm. Anal. Cal., 88 (2007) 885.
Y. Xi, W. Martens, H. He and R. L. Frost, J. Therm. Anal. Cal., 81 (2005) 91.
C. W. Beck, Am. Mineral., 35 (1950) 985.
L. B. Railsback, Carbonates Evaporites, 14 (1999) 1.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vágvölgyi, V., Hales, M., Martens, W. et al. Dynamic and controlled rate thermal analysis of hydrozincite and smithsonite. J Therm Anal Calorim 92, 911–916 (2008). https://doi.org/10.1007/s10973-007-8846-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-007-8846-5