Abstract
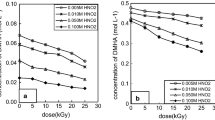
N,N-dimethylhydroxylamine (DMHA) is an effective novel salt-free reductant used in spent fuel reprocessing. This paper reports the effect of monomethylhydrazine (MMH) on γ-ray radiolysis and radiolytic by-products of DMHA in HNO3. The presence of MMH improves the stability of DMHA against radiation significantly, the main radiolytic by-products are N-methylhydroxylamine, HCHO, HCOOH and HNO2, which concentration increases with the dose. The concentration of N-methylhydroxylamine increases with the MMH concentration, but that of HCHO, HCOOH and HNO2 decreases with increasing MMH concentration. These results will provide considerable reference for the application of DMHA in the PUREX process.





Similar content being viewed by others
References
Adamantiades A, Kessides I (2009) Nuclear power for sustainable development: current status and future prospects. Energy Policy 37:5149–5166
Mossini E, Macerata E, Giola M et al (2015) Radiation-induced modifications on physico chemical properties of diluted nitric acid solutions within advanced spent nuclear fuel reprocessing. J Radioanal Nucl Chem 304:395–400
Choppin GR, Morgenstern AJ (2000) Radionuclide separations in radioactive waste disposal. J Radioanal Nucl Chem 243:45–51
Schlea CS, Caverly MR, Henry HE, et al (1963) Uranium(IV) nitrate as a reducing agent for plutonium(IV) in the purex process. DP-808:1-20
Sze YK, Clegg LJ, Gerwing AF et al (1982) Oxidation of Pu(III) by nitric acid in tri-n-butyl phosphate solutions. Part I. Kinetics of the reaction and its effect on plutonium losses in countercurrent liquid–liquid extraction. J Nucl Technol 56:527–534
Sze YK, Gosselin JA (1983) Oxidation of Pu(III) by nitric acid in tri-n-butyl phosphate solutions. Part II. Chemical methods for the suppression of oxidation to improve plutonium separation in contactor operation. J Nucl Technol 63:431–441
McKibben JM, Bercaw JE (1971) Hydroxylamine nitrate as a plutonium reductant in the purex solvent extraction process. DP-1248:1-22
Ochsenfeld W, Petrich G (1983) Neptunium decontamination in a uranium purification cycle of a spent fuel reprocessing plant. Separ Sci Technol 18:1685–1698
Biddle P, Miles JH (1968) Rate of reaction of nitrous acid with hydrazine and with sulphamic acid. J Inorg Nucl Chem 30:1291–1297
Zheng WF, Zhang ZF, Lin ZJ et al (2001) Study on separation of Np from U by acetohydroxamic acid in purex process. Chin J Nucl Sci Eng 21:369–374 (in Chinese)
Ye GA (2004) Review on the study and application of organic salt-free reagent in purex process. Atom Energy Sci Technol 38:152–158 (in Chinese)
Taylor RJ, May I, Koltunov VS et al (1998) Kinetic and solvent extraction studies of the selective reduction of Np(VI) by new salt-free reducing agents. Radiochim Acta 81:149–156
Chen YX, Tang HB, Liu JP et al (2011) The kinetics of the reduction reaction of plutonium(IV) with N,N-dimethylhydroxylamine. J Radioanal Nucl Chem 289:41–47
Liu JP, He H, Tang HB et al (2011) The application of N,N-dimethylhydroxylamine as reductant for the separation of plutonium from uranium. J Radioanal Nucl Chem 288:351–356
Li XG, He H, Ye GA et al (2010) Reaction kinetics between N,N-dimethylhydroxylamine and neptunium(VI). J Nucl Radiochem 32:262–267 (in Chinese)
Wang JH, Wu MH, Bao BR et al (2007) Study on hydrogen and carbon monoxide produced by radiation degradation of N,N-dimethylhydroxylamine. J Radioanal Nucl Chem 273:371–373
Wang JH, Li Ch, Wu MH et al (2010) Gaseous products of aqueous N,N-dimethyl hydroxylamine degraded by radiation. Nucl Sci Techniq 21:233–236
Wang JH, Li Q, Wu MH et al (2012) Radiolysis of N,N-dimethylhydroxylamine and its radiolytic liquid organics. J Radioanal Nucl Chem 292:249–254
Wang JH, Cao XJ, Li Ch et al (2015) Effect of HNO3 on the γ radiolysis and radiolytic liquid products of N,N-dimethylhydroxylamine. Acta Phys Chim Sin 31:559–565
Koltunov VS, Zhuravleva GI, Marchenko VI et al (2007) Kinetics and mechanism of the plutonium catalysed nitric acid oxidation of U(IV) ions in 30% tributyl phosphate solution. Radiochim Acta 95:559–567
Perrott JR, Stedman G, Uysal N (1976) Kinetic and product study of the reaction between nitrous acid and hydrazine. J Chem Soc, Dalton Trans 20:2058–2064
Wu GP, Wei FS (1992) Determination of nitrite in the wet precipitation—N-(1-naphthyl)-1,2-diaminoethane dihydrochloride spectrophotometry. GB-13580.7-92:1-2 (in Chinese)
Li C (2010) Study on γ-irradiation stability and radiolysis products of N,N-dimethylhydroxylamine at low doses. Master thesis, Shanghai University (in Chinese)
Li HB, Su Zh, Cong HF et al (2012) α and γ irradiation stability of 30% TBP-kerosene-HNO3 systems. J Nucl Radiochem 34:281–285 (in Chinese)
Karraker DG (2002) Radiation chemistry of acetohydroxamic acid in the Urex process (WSRC-TR-2002-00283), U.S. Department of Commerce, National Technical Information Service: Springfielf, pp 1–8
Wang JH, Wang P, Li Ch et al (2019) Effect of nitrous acid on the γ radiolysis and radiolytic products of N,N-dimethylhydroxylamine. J Radioanal Nucl Chem 319:759–765
Wei Y, Wang H, Pan YJ et al (2009) Reaction kinetics of monomethylhydrazine with nitrous acid in perchloric acid solution. J Nucl Radiochem 31:198–201 (in Chinese)
Li GL, He H (2011) Study on mechanism for oxidation of N,N-dimethylhydroxylamine by nitrous acid. J Radioanal Nucl Chem 287:673–678
Burton M (1951) An introduction to radiation chemistry. J Chem Educ 28:404–420
KozŁowska-Milner E, Broszkiewicz RK (1978) Pulse radiolysis of HNO3 and HNO3(aq). Radiat Phys Chem 11:253–260
Katsumura Y, Jiang PY, Nagaishi R, Oishi T et al (1991) Pulse radiolysis study of aqueous nitric acid solutions. Formation mechanism, yield, and reactivity of NO3 radical. J Phys Chem 95:4435–4439
Koltunov VS, Baranov SM (1995) Organic derivatives of hydrazine and hydroxylamine in future technology of spent nuclear fuel reprocessing. In: International conference on evaluation of nuclear fuel cycle systems, France, pp 577–582
Li CB, Zhi Cao, Yan TH et al (2018) Secondary reaction of MMH in DMHAN-MMH solution system. J Nucl Radiochem 40:30–36 (in Chinese)
Acknowledgements
The authors would like to express our sincere acknowledgement for the financial assistance of National Natural Science Foundation of China (No. 20771074).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, S.Y., Wang, J.H., Li, C. et al. Effect of monomethylhydrazine on γ-ray radiolysis and radiolytic by-products of DMHA in nitric acid. J Radioanal Nucl Chem 327, 259–267 (2021). https://doi.org/10.1007/s10967-020-07489-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-020-07489-8