Abstract
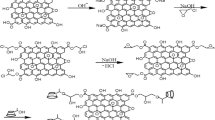
A specialized ethylamine-bridged β-cyclodextrins (EB β-CD) was synthetized. EB β-CD’s adsorption properties of thorium ions were performed. Experimental results show that the adsorption of thorium using the EB β-CD can achieve equilibrium within 60 min at room temperature under the condition of pH 4, the adsorbance is 10.49 mg g−1. The results of SEM and BET nitrogen adsorption show that, EB β-CD is typical mesoporous material. According to pseudo-second-order kinetics, the adsorption of Th(IV) indicating the influence of textural properties of EB β-CD on the rate of adsorption. The thermodynamic parameters obtained showed that the adsorption process is exothermic, spontaneous process.
















Similar content being viewed by others
References
Lin P, Guo L, Chen M (2014) Adsorption and fractionation of thorium and protactinium on nanoparticles in seawater. Mar Chem 162:50–59
Liu P, Qi W, Du Y, Li Z, Wang J, Bi J, Wu W (2014) Adsorption of thorium(IV) on magnetic multi-walled carbon nanotubes. Sci China Chem 57(11):1483–1490
Piech R, Paczosa-Bator B (2014) New adsorptive stripping determination of ATP with thorium(IV) on renewable silver amalgam film electrode. Int J Electrochem Sci 9:4287–4296
Aly A, Amer HA, Shawky S, Kandil AT (2014) Separation of thorium from aqueous solution by non living water hyacinth roots. Tech J Eng Appl Sci 4:1–13
Humelnicu D, Blegescu C, Ganju D (2014) Removal of uranium(VI) and thorium(IV) ions from aqueous solutions by functionalized silica: kinetic and thermodynamic studies. J Radioanal Nucl Chem 299:1183–1190
Sadeek SA, Moussa EMM, El-Sayed MA (2014) Uranium(VI) and thorium(IV) adsorption studies on chelating resin containing pentaethylenehexamine as a functional group. J Dispers Sci Technol 35(7):926–933
Zhou L, Jia Y, Peng J, Liu Z, Al-Zaini E (2014) Competitive adsorption of uranium(VI) and thorium(IV) ions from aqueous solution using triphosphate-crosslinked magnetic chitosan resins. J Radioanal Nucl Chem 302(1):331–340
Ji XZ, Liu HJ, Wang LL, Sun YK, Wu YW (2012) Study on adsorption of Th(IV) using surface modified dibenzoylmethane molecular imprinted polymer. J Radioanal Nucl Chem 295(1):265–270
Liu Y, Chen Y (2006) Cooperative binding and multiple recognition by bridged bis(β-cyclodextrin)s with functional linkers. Acc Chem Res 39(10):68–691
Zhao Y, Liu X-Q, Gu J (2008) Synthesis of novel bis(β-cyclodextrin)s linked with aromatic diamine and their molecular recognition with model substrates. J Phys Org Chem 21(6):440–448
Zhang Y-H, Zhang Y-M, Chen Y, Yang Y, Liu Y (2014) Phenanthroline bridged bis(β-cyclodextrin)s/adamantane-carboxylic acid supramolecular complex as an efficient fluorescence sensor to Zn2+. Org Chem Front 1(4):355–360
Kremer C, Lutzen A (2014) Allosteric binding of capsaicin by a bis(beta-cyclodextrin)-2,2′-bipyridine receptor. Chem Eur J 20(29):8852–8855
Zhang YM, Wang Z, Chen Y, Chen HZ, Ding F, Liu Y (2014) Molecular binding behavior of a bispyridinium-containing bis(beta-cyclodextrin) and its corresponding [2]rotaxane towards bile salts. Org Biomol Chem 12(16):2559–2567
Liu Y, You C-C, Li B (2001) Synthesis and molecular recognition of novel oligo(ethylenediamino) bridged bis(β-cyclodextrin) s and their Copper(ii) Complexes: enhanced molecular binding ability and selectivity by multiple recognition. Chem Eur J 7(6):1281–1288
Gangadhar T, Bhoi VI, Kumar S, Murthy CN (2013) Supramolecular self-assembly and nanoencapsulation of [60]fullerene by bis-β-cyclodextrin. J Incl Phenom Macrocycl Chem 79:215–223
Casas-Solvas JM, Ortiz-Salmeron E, Fernandez I, Garcia-Fuentes L, Santoyo-Gonzalez F, Vargas-Berenguel A (2009) Ferrocene-beta-cyclodextrin conjugates: synthesis, supramolecular behavior, and use as electrochemical sensors. Chem Eur J 15(33):8146–8162
Yin JJ, Sharma S, Shumyak SP, Wang ZX (2013) Synthesis and biological evaluation of novel folic acid receptor-targeted, beta-cyclodextrin-based drug complexes for cancer treatment. PLoS One 8(5):1–20
Sun M, Zhang H-Y, Liu B-W, Liu Y (2013) Construction of a supramolecular polymer by bridged bis(permethyl-β-cyclodextrin) s with porphyrins and its highly efficient magnetic resonance imaging. Macromolecules 46(11):4268–4275
Liu Y, You C-C, Inoue Y (2000) Molecular recognition of fluorescent dyes with novel triethylenetetraamine-tethered bis(β-cyclodextrin) and its copper(II) complex: enhanced binding and selectivity by tether ligation. Tetrahedron Lett 41:6869–6873
Zhao Z-G (2004) Surface fractal analysis of mesoporous adsorbents. Acta Chim Sin 62:219–223
Zhao D (2008) Adsorption of thorium(IV) on MX-80 bentonite: effect of pH, ionic strength and temperature. Appl Clay Sci 41:17–23
Yang SK, Tan N, Yan XM, Chen F, Long W, Lin YC (2013) Thorium(IV) removal from aqueous medium by citric acid treated mangrove endophytic fungus Fusarium sp. #ZZF51. Mar Pollut Bull 74(1):213–219
Tong KS, Kassim MJ, Azraa A (2011) Adsorption of copper ion from its aqueous solution by a novel biosorbent Uncaria gambir: equilibrium, kinetics, and thermodynamic studies. Chem Eng J 170(1):145–153
Sheng G, Hu J, Wang X (2008) Sorption properties of Th(IV) on the raw diatomite–effects of contact time, pH, ionic strength and temperature. Appl Radiat Isot 66(10):1313–1320
Zafar S, Khalid N, Daud M, Mirza ML (2015) Kinetic studies of the adsorption of thorium ions onto rice husk from aqueous media: linear and nonlinear approach. Nucleus 52(1):14–19
Mellah A, Chegrouche S, Barkat M (2006) The removal of uranium(VI) from aqueous solutions onto activated carbon: kinetic and thermodynamic investigations. J Colloid Interface Sci 296(2):434–441
Zhao D (2008) Adsorption of thorium(IV) on MX-80 bentonite: effect of pH, ionic strength and temperature. Appl Clay Sci 41(1–2):17–23
Acknowledgments
The authors gratefully acknowledge the financial support from National Natural Science Foundation of China (No. 11375084).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hu, C., Liu, Hj., Peng, L. et al. Synthesis of ethylamine-bridged β-cyclodextrins and adsorption properties of thorium. J Radioanal Nucl Chem 308, 251–259 (2016). https://doi.org/10.1007/s10967-015-4306-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-015-4306-z