Abstract
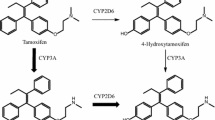
Tamoxifen [TAM; ([Z]-2-[4-(1,2-diphenyl-1-di-butenyl)-phenoxy]-N,N-dimethylethanamine) has been used as an antiestrogen drug for treatment and prevention of human breast cancer. The aim of this study is conjugation of hydrophilic glucuronic acid to the starting substance TAM and labeling with 131I using iodogen as oxidizing agent. The reactions are completed in three steps, including enzymatic reaction, with the following sub-steps; preparation of microsomal fraction from rat liver and subsequent purification of UDP-glucuronyl transferase (UDPGT), estimation of protein amount in microsomal samples and glucuronidation reaction. Synthesized glucuronide derivative (TAM-G) was purified using high performance liquid chromatography (HPLC). Mass spectroscopy of cold standard showed that the labeling most probably occurs in ortho position to the aromatic ring containing the ether group of TAM-G as expected. Radiochemical yield of the 131I labeled TAM-G ([131I]TAM-G) is determined by using Thin Layer Radio Chromatography (TLRC). The radiopharmaceutical potential of [131I]TAM-G is examined by biodistribution studies that is run on normal female Albino Wistar rats. According to biodistribution results 131I labeled TAM-G may be proposed as a new antiestrogen glucuronide imaging agent for breast and uterus.







Similar content being viewed by others
References
Martin EA, Brown K, Gaskell M, Al-Azzawi F, Garner RC, Boocock DJ et al (2003) Tamoxifen DNA damage deteted in human endometrium using accelerator mass spectrometry. Cancer Res 63:8461–8465
Musa MA, Khan MOF, Cooperwood JS (2007) Medicinal chemistry and emerging strategies applied to the development of selective estrogen receptor modulators (SERMs). Curr Med Chem 14:1249–1261
Marcsek Z, Kocsis Z, Jakab M, Szende B, Tompa A (2004) The efficacy of tamoxifen in estrogen receptor-positive breast cancer cells is enhanced by a medical nutriment. Cancer Biother Radiopharm 19:746–753
Sperker B, Backman JT, Kroemer HK (1997) The role of beta-glucuronidase in drug disposition and drug targeting in humans. J Clin Pharmacol 33:18–31
Houba PHJ, Leenders RGG, Boven E, Scheeren JW, Pinedo HM, Haisma HJ (1996) Characterization of novel anthracycline prodrugs activated by human β-glucuronidase for use in antibody-directed enzyme prodrug therapy. Biochem Pharmacol 9:455–463
Kamal A, Tekumalla V, Raju P, Naidu VGM, Diwan PV, Sistla R (2008) Pyrrolo[2,1-c][1,4]benzodiazepine-β-glucuronide prodrugs with a potential for selective therapy of solid tumors by PMT and ADEPT strategies. Bioorg Med Chem Lett 18:3769–3773
Starkey DE, Han A, Bao JJ, Ahn CH, Wehmeyer KR, Prenger MC et al (2001) Fluorogenic assay for β-glucuronidase using microchip-based capillary electrophorrsis. J Chromatogr 762:33–41
Minton JP, Walaszek Z, Schooley W, Hanausek WM, Webb TE (1986) Beta-Glucuronidase levels in patients with fibrocystic breast disease. Breast Cancer Res Treat 8:217–222
Delpassand SE, Yang JD, Wallace S, Cherıf A, Quadrı MS, Price J et al (1996) Synthesis, biodistribution and estrogen receptor scintigraphy of Indium-111-diethylenetriamine pentaacetic acid-tamoxifen analogue. J Pharm Sci 85:553–559
Yurt A, Biber Muftuler FZ, Unak P, Enginar H, Yolcular S, Acar C (2009) Synthesis of a novel anti estrogen radioligand (99mTc-TOR-DTPA). Cancer Biother Radiopharm 24:707–716
Zihnioglu FEU (1992) Graduate school of natural and applied sciences chemistry department biochemistry. Doctorate Thesis; 169
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Alkharfy KM, Frye RF (2002) Sensitive liquid chromatographic method using fluorescence detection for the determination of estradiol 3- and 17-glucuronides in rat and human liver microsomal incubations: formation kinetics. J Chromatogr B Analyt Technol Biomed Life Sci 1:33–38
Stevenson DE, Hubl U (1999) Optimization of β-glucuronide synthesis using UDP-glucuronyl transferase. Enzyme Microbial Technol 24:388–396
Stevenson DE (1999) Optimisation of UDP-glucuronyl transferase-catalysed synthesis of testosterone-β-D-glucuronide by inhibition of contaminating ß-glucuronidase. Biotechnol Tech 13:17–21
Mihailescu R, Aboul-Enein HY, Efstatide MD (2000) Identification of tamoxifen and metabolites in human male urine by GC/MS. Biomed Chromatogr 14:180–183
Ünak T, Akgün Y, Duman Y, Erenel G (2001) Self-radioiodination of iodogen. Appl Radiat Isot 54:749–752
Biber Muftuler FZ, Unak P, Teksoz S, Acar C, Yolcular S, Yürekli Y (2008) 131I labeling of tamoxifen and biodistribution studies in rats. Appl Radiat Isot 66:178–187
Biber FZ, Unak P, Ertay T, Medine EI, Zihnioglu F, Tascı C et al (2006) Synthesis of an estradiol glucuronide derivative and investigation of its radiopharmaceutical potential. Appl Radiat Isot 64:778–788
Biber Muftuler FZ, Unak P, Yolcular S, Yurt Kilcar A, Ichedef C, Enginar H, Sakarya S (2010) Synthesis, radiolabeling and in vivo tissue distribution of an anti-oestrogen glucuronide compound, 99mTc-TOR-G. Anticancer Res 30:1249–1256
Delpassand ES, Yang DG, Wallace S, Cherif A, Quadri SM, Price G et al (1996) Synthesis, biodistribution, and estrogen receptor scintigraphy of indium-111-diethyleneaminpentaacetic acid-tamoxifen analogue. J Pharm Sci 85:553–559
Kon OL (1983) An antiestrogen binding protein in human tissues. J Biol Chem 258:3173–3177
Winborn WB, Sheridan PJ, McGill HC (1987) Sex steroid in the stomach, liver, pancreas, and gastrointestinal tract of the baboon. Gastroenterology 92:23–32
Ciocca DR, Vargas LM (1995) Estrogen receptors in human nontarget tissues: biological and clinical implications. Endocr Rev 16:35–62
Enginar H, Unak P, Lambrecht FY, Biber FZ, Medine IE, Çetinkaya B (2005) Synthesis of I-131 labeled estrone derivatives and biodistribution studies on rats. J Radioanal Nucl Chem 264:535–539
Unak T, Avcıbaşı U, Yıldırım Y, Cetinkaya B (2003) Attempts to develop a new nuclear measurement technique of β-glucuronidase levels in biological samples. Czechoslovak J Phys 53:A797–A802
Fabian C, Sternson L, El-serafi M, Cain L, Hearne E (1981) Clinical pharmacology of Tamoxifen in patients with breast cancer: correlation with clinical data. Cancer 48:876–882
Silva MC, Luciana P, Gano L, SáMelo ML, Inohae E, Shuntaro M et al (2001) Synthesis and biological evaluation of two new radiolabelled estrogens: (125I)(E)-3-methoxy-17α-iodovinylestra-1,3,5(10),6-tetraen-17β-ol and (125I)(Z)-3-methoxy-17α-iodovinylestra-1,3,5(10),6-tetraen-17β-ol. Appl Radiat Isot 54:227–239
Strickland LA, Ponce YZ, Hunter DH, Zabel PL, Powe JE, Morrissey G et al (1990) Amino and iodotamoxifens: synthesis estrogen receptor affinity and biodistribution. Drug Des Deliv 6:195–212
Acknowledgements
The authors thank Prof. Dr. Tamerkan OZGEN and Research Assistant Murat ERDOGAN for GS/MS/MS measurements and Assoc. Prof. Cavit KAZAZ and Dr. Murat ACAR for NMR spectra and MSc. Feriha TOKSOZ and Research Assistant Ayfer YURT KILCAR for technical helping.
Disclosure
This work financially supported by T.R. Prime Ministry State Planning Organization Contract No 06 DPT 06 and Ege University Research Fund contact no 2007 NBE 006.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Muftuler, F.Z.B., Unak, P., Içhedef, Ç. et al. Synthesis of a radioiodinated antiestrogen glucuronide compound (TAM-G). J Radioanal Nucl Chem 287, 679–689 (2011). https://doi.org/10.1007/s10967-010-0932-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-010-0932-7