Abstract
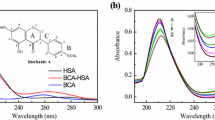
Cuminaldehyde, an oxidized aldehyde monoterpene, present in green cumin seeds (Cuminum cyminum Linn, Family—Apiaceae), is traditionally used for the treatment of abdominal colic, dyspepsia, diarrhea, and jaundice. Also, many studies have reported the antioxidant, antibacterial and antifungal effects of cuminaldehyde. Serum albumins are the major soluble and small molecule-binding proteins, present in abundance in the circulatory system of a wide variety of organisms. Studies on the interaction of bioactive molecules with bovine serum albumin (BSA) and human serum albumin (HSA) have attracted enormous interest due to its direct consequence on drug delivery, pharmacokinetics, pharmacodynamics, therapeutic efficacy and drug designing. Our present study is carried out to understand the mechanism of interaction of pharmaceutically important component of spices, cuminaldehyde with BSA and HSA. Fluorescence spectroscopic measurements confirmed that cuminaldehyde interacted with BSA and HSA and quenched its fluorescence intensity via static quenching mechanism. UV–Visible absorption studies and CD-spectroscopy showed the change in secondary conformation of BSA and HSA upon interaction with cuminaldehyde. CD-spectroscopy revealed that HSA is unfolded at lower concentration of cuminaldehyde compared to BSA. The location of binding site for cuminaldehyde in BSA and HSA was investigated by site probe displacement experiments and the results indicated that cuminaldehyde preferred to bind site-I, located in subdomain IIA of both BSA and HSA. Thermodynamic studies revealed that vander Waal’s interaction and hydrogen bonding play a major role in cuminaldehyde-BSA system while hydrophobic interactions play vital role in cuminaldehyde-HSA system. The molecular dockings of cuminaldehyde with BSA/HSA further confirmed the formation of the stable BSA/HSA–cuminaldehyde complex and cuminaldehyde binds at site-I of HSA. On the other hand, docking study showed that cuminaldehyde interacts with some residues close to site-I of BSA. Both experimental and theoretical results showed that the ΔG0 values are comparable for both the proteins, which indicate almost equal stability of cuminaldehyde-BSA and cuminaldehyde–HSA complex.












Similar content being viewed by others
References
Ebada, M.E.: Cuminaldehyde: A potential drug candidate. J. Pharmacol. Clin. Res. 2(2), 1–4 (2017)
Morshedi, D., Aliakbari, F., Tayaranian-Marvian, A., Fassihi, A., Pan-Montojo, F., Pérez-Sánchez, H.: Cuminaldehyde as the major component of cuminum cyminum, a natural aldehyde with inhibitory effect on alpha-synuclein fibrillation and cytotoxicity. J. Food Sci. 80(10), 2336–2345 (2015)
Al-Snafi, A.E.: Medicinal plants possessed anti-Parkinsonian effects with emphasis on their mechanisms of action. GSC Biol. Pharmaceut. Sci. 17(1), 232–237 (2021)
Monteiro-Neto, V., de Souza, C.D., Gonzaga, L.F., da Silveira, B.C., Sousa, N.C., Pontes, J.P., Fernandes, E.S.: Cuminaldehyde potentiates the antimicrobial actions of ciprofloxacin against Staphylococcus Aureus and Escherichia coli. PLoS ONE 15(5), e0232987 (2020)
Li, H., Zhang, M., Addo, K.A., Yu, Y., Xiao, X.: Action mode of cuminaldehyde against Staphylococcus aureus and its application in sauced beef. LWT. 155, 112924 (2022)
Basu, A., Kumar, G.S.: Thermodynamics of the interaction of the food additive tartrazine with serum albumins: A microcalorimetric investigation. Food Chem. 175, 137–142 (2015)
Basu, A., Kumar, G.S.: Study on the interaction of the toxic food additive carmoisine with serum albumins: A microcalorimetric investigation. J. Hazard. Mater. 273, 200–206 (2014)
Yu, J., Liu, J.Y., Xiong, W.M., Zhang, X.Y., Zheng, Y.: Binding interaction of sodium benzoate food additive with bovine serum albumin: Multi-spectroscopy and molecular docking studies. BMC Chem. 13(1), 1–8 (2019)
Zhang, G., Ma, Y., Wang, L., Zhang, Y., Zhou, J.: Multispectroscopic studies on the interaction of Maltol, a food additive, with bovine serum albumin. Food Chem. 133(2), 264–270 (2012)
Javaheri-Ghezeldizaj, F., Mahmoudpour, M., Yekta, R., Dolatabadi, J.E.N.: Albumin binding study to sodium lactate food additive using spectroscopic and molecular docking approaches. J. Mol. Liq. 310, 113259 (2020)
Al-Shabib, N.A., Khan, J.M., Ali, M.S., Al-Lohedan, H.A., Khan, M.S., Al-Senaidy, A.M., Shamsi, M.B.: Exploring the mode of binding between food additive “butylated hydroxytoluene (BHT)” and human serum albumin: Spectroscopic as well as molecular docking study. J. Mol. Liq. 230, 557–564 (2017)
Mohammadzadeh-Aghdash, H., Dolatabadi, J.E.N., Dehghan, P., Panahi-Azar, V., Barzegar, A.: Multi-spectroscopic and molecular modeling studies of bovine serum albumin interaction with sodium acetate food additive. Food Chem. 228, 265–269 (2017)
Taheri, R., Hamzkanlu, N., Rezvani, Y., Niroumand, S., Samandar, F., Amiri-Tehranizadeh, Z., Chamani, J.: Exploring the HSA/DNA/lung cancer cells binding behavior of p-synephrine, a naturally occurring phenyl ethanol amine with anti-adipogenic activity: multi spectroscopic, molecular dynamic and cellular approaches. J. Mol. Liq. 368, 120826 (2022)
Sharifi-Rad, A., Mehrzad, J., Darroudi, M., Saberi, M.R., Chamani, J.: Oil-in-water nanoemulsions comprising berberine in olive oil: Biological activities, binding mechanisms to human serum albumin or holo-transferrin and QMMD simulations. J. Biomol. Struct. Dyn. 39(3), 1029–1043 (2021)
Chamani, J., Vahedian-Movahed, H., Saberi, M.R.: Lomefloxacin promotes the interaction between human serum albumin and transferrin: A mechanistic insight into the emergence of antibiotic’s side effects. J. Pharm. Biomed. Anal. 55(1), 114–124 (2011)
Goncharenko, N.A., Dmytrenko, O.P., Kulish, M.P., Pavlenko, O.L., Lesiuk, A.I., Busko, T.O., Denis, L.V.: Mechanisms of the interaction of bovine serum albumin with anticancer drug gemcitabine. Mol. Cryst. Liq. Cryst. 701(1), 59–71 (2020)
Chilom, C.G., David, M., Florescu, M.: Monitoring biomolecular interaction between folic acid and bovine serum albumin. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 230, 118074 (2020)
Soltanabadi, O., Atri, M.S., Bagheri, M.: Spectroscopic analysis, docking and molecular dynamics simulation of the interaction of cinnamaldehyde with human sserum albumin. J. Incl. Phenom. Macrocycl. Chem. 91, 189–197 (2018)
Sun, Q., Yang, H., Tang, P., Liu, J., Wang, W., Li, H.: Interactions of cinnamaldehyde and its metabolite cinnamic acid with human serum albumin and interference of other food additives. Food Chem. 243, 74–81 (2018)
Soares, S., Mateus, N., De Freitas, V.: Interaction of different polyphenols with Bovine Serum Albumin (BSA) and Human Salivary α-amylase (HSA) by fluorescence quenching. J. Agric. Food Chem. 55(16), 6726–6735 (2007)
Ali, M.S., Rehman, M.T., Al-Lohedan, H., Alajmi, M.F.: Spectroscopic and molecular docking investigation on the interaction of cumin components with plasma protein: Assessment of the comparative interactions of aldehyde and alcohol with human serum albumin. Int. J. Mol. Sci. 23(8), 4078 (2022)
Ali, M.S., Rehman, M.T., Al-Lohedan, H.A., Alajmi, M.F.: Study of the binding of cuminaldehyde with bovine serum albumin by spectroscopic and molecular modeling methods. J. Spectrosc. (2023). https://doi.org/10.1155/2023/4191046
Yao, D., Yu, J., Pan, Y., Huang, F., Bian, H., Yu, Q., Liang, H.: Spectroscopic studies on the binding of kaempferol-3, 7-α-L-rhamnopyranoside to bovine serum albumin. Chin. J. Chem. 30(2), 438–444 (2012)
Zhang, Y.Z., Zhou, B., Liu, Y.X., Zhou, C.X., Ding, X.L., Liu, Y.: Fluorescence Study on the Interaction of Bovine Serum Albumin with P-Aminoazobenzene. J. Fluoresc. 18, 109–118 (2008)
Berman, H.M., Battistuz, T., Bhat, T.N., Bluhm, W.F., Bourne, P.E., Burkhardt, K., Feng, Z., Gilliland, G.L., Iype, L., Jain, S., Fagan, P., Marvin, J., Padilla, D., Ravichandran, V., Schneider, B., Thanki, N., Weissig, H., Westbrook, J.D., Zardecki, C.: The protein data bank. Acta Crystallogr. D Biol. Crystallogr. 58(6), 899–907 (2002)
O’Boyle, N.M., Banck, M., James, C.A., Morley, C., Vandermeersch, T., Hutchison, G.R.: Open Babel: an open chemical toolbox. J. Cheminf. 3(1), 1–14 (2011)
Liu, Y., Yang, X., Gan, J., Chen, S., Xiao, Z.X., Cao, Y.: CB-dock 2: improved protein-ligand blind docking by integrating cavity detection, docking and homologous template fitting. Nucleic Acids Res. 50, 159–164 (2022)
Laskowski, R.A., Swindells, M.B.: LigPlot+: multiple ligand-protein interaction diagrams for drug discovery. J. Chem. Inf. Model. 51(10), 2778–2786 (2011)
Wang, Z., Pan, H., Sun, H., Kang, Y., Liu, H., Cao, D., Hou, T.: FASTDRH: A webserver to predict and analyze protein-ligand complexes based on molecular docking and MM/PB (Gb)SA computation. Brief. Bioinform. 23(5), 1–10 (2022)
Papadopoulou, A., Green, R.J., Frazier, R.A.: Interaction of flavonoids with bovine serum albumin: A fluorescence quenching study. J. Agric. Food Chem. 53(1), 158–163 (2005)
Bi, S., Sun, Y., Qiao, C., Zhang, H., Liu, C.: Binding of several anti-tumor drugs to bovine serum albumin: Fluorescence study. J. Lumin. 129(5), 541–547 (2009)
Bakar, K.A., Feroz, S.R.: A critical view on the analysis of fluorescence quenching data for determining ligand–protein binding affinity. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. Mol. Biomol. Spectrosc. 223, 117337 (2019)
Feldman, I., Young, D., McGuire, R.: Static and dynamic quenching of protein fluorescence: I—Bovine serum albumin. Biopolymers: Original Res. Biomol. 14(2), 335–351 (1975)
Jayabharathi, J., Thanikachalam, V., Srinivasan, N., Perumal, M.V.: Luminescent study on the binding interaction of bioactive imidazole with bovine serum albumin—A static quenching mechanism. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 84(1), 233–237 (2011)
Kandagal, P.B., Ashoka, S., Seetharamappa, J., Shaikh, S.M.T., Jadegoud, Y., Ijare, O.B.: Study of the interaction of an anticancer drug with human and bovine serum albumin: Spectroscopic approach. J. Pharm. Biomed. Anal. 41(2), 393–399 (2006)
Bulusu, G., Desiraju, G.R.: Strong and weak hydrogen bonds in protein-ligand recognition. J. Indian Inst. Sci. 100, 31–41 (2020)
Hubbard, R.E., Haider, M.K.: Hydrogen bonds in proteins: role and strength. eLS. (2010). https://doi.org/10.1002/9780470015902.a0003011.pub2
Williams, M.A., Ladbury, J.E.: Hydrogen Bonds in Protein-Ligand Complexes. In: Protein-Ligand Interactions: from Molecular Recognition to Drug Design, pp. 137–161. Wiley, NY (2003)
Leckband, D., Israelachvili, J.: Intermolecular forces in biology. Q. Rev. Biophys. 34(2), 105–267 (2001)
Zhang, R.J., Kou, S.B., Hu, L., Li, L., Shi, J.H., Jiang, S.L.: Exploring binding interaction of baricitinib with bovine serum albumin (BSA): Multi-spectroscopic approaches combined with theoretical calculation. J. Mol. Liq. 354, 118831 (2022)
Chen, W.Y., Huang, H.M., Lin, C.C., Lin, F.Y., Chan, Y.C.: Effect of temperature on hydrophobic interaction between proteins and hydrophobic adsorbents: Studies by isothermal titration calorimetry and the van’t Hoff equation. Langmuir 19(22), 9395–9403 (2003)
Lou, Y.Y., Zhou, K.L., Pan, D.Q., Shen, J.L., Shi, J.H.: Spectroscopic and molecular docking approaches for investigating conformation and binding characteristics of clonazepam with bovine serum albumin (BSA). J. Photochem. Photobiol. B. 167, 158–167 (2017)
Zhang, Y.Z., Zhou, B., Zhang, X.P., Huang, P., Li, C.H., Liu, Y.: Interaction of malachite green with bovine serum albumin: Determination of the binding mechanism and binding site by spectroscopic methods. J. Hazard. Mater. 163(2–3), 1345–1352 (2009)
Dufour, C., Dangles, O.: Flavonoid-serum albumin complexation: determination of binding constants and binding sites by fluorescence spectroscopy. Biochim. Biophys. Acta (BBA) 1721(1–3), 164–173 (2005)
Sudlow, G.D.J.B., Birkett, D.J., Wade, D.N.: The characterization of two specific drug binding sites on human serum albumin. Mol. Pharmacol. 11(6), 824–832 (1975)
Sudlow, G.D.J.B., Birkett, D.J., Wade, D.N.: Further characterization of specific drug binding sites on human serum albumin. Mol. Pharmacol. 12(6), 1052–1061 (1976)
Chuang, V.T.G., Otagiri, M.: Stereoselective binding of human serum albumin. Chirality 18(3), 159–166 (2006)
Pettersen, E.F., Goddard, T.D., Huang, C.C., Couch, G.S., Greenblatt, D.M., Meng, E.C., Ferrin, T.E.: UCSF chimeraa visualization system for exploratory research and analysis. J. Comput. Chem. 25(13), 1605–1612 (2004)
Acknowledgements
The author is grateful to Indian Institute of Technology (IIT), Guwahati and Pandu College, Assam, for providing instrument facilities.
Funding
This work was supported by a Grant from Department of Biotechnology (DBT), India (NECBH/2019-20/160 & Date: 29-04-2019)
Author information
Authors and Affiliations
Contributions
RB designed and carried out all the experiments, analyzed the results and prepared the manuscripts. VSKM performed the molecular docking. GH performed CD Spectroscopy.
Corresponding author
Ethics declarations
Competing interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Borah, R., Mattaparthi, V.S.K. & Hazarika, G. Assessment of the Comparative Interactions of Cuminaldehyde with Bovine Serum Albumin and Human Serum Albumin Through Spectroscopic and Molecular Docking Investigation. J Solution Chem (2024). https://doi.org/10.1007/s10953-024-01367-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10953-024-01367-7