Abstract
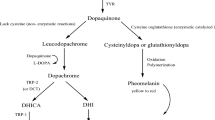
As a safe substitute for hydroquinone, β-arbutin, a natural plant substance, and its synthetic counterpart, α-arbutin, are used in depigmentation formulations. However, there are debatable points regarding the impact of arbutin on tyrosinase and the pigmentation process. To shed light on this issue, the effects of Pyrus biossieriana leaves extract (PbLE) and β-arbutin, extracted from PbLE, on mushroom tyrosinase (MT) were comprehensively examined. The study was focused on cresolase activity as the characteristic reaction of a tyrosinase. Kinetics studies disclosed that β-arbutin can modulate MT monophenolase activity from inhibition to activation or vice versa. β-Arbutin inhibited l-tyrosine (LTy) oxidation at concentrations < 0.3 mM but it increased (more than 400%) the enzymatic oxidation of l-tyrosine at the concentrations > 0.3 mM. An opposite pattern (activation then inhibition) was observed when a synthetic substrate was used instead of LTy. Computational studies, focused on the heavy chain of MT, indicated that β-arbutin effect could be overruled by the enzyme's ability to provide the ligand with a non-specific binding site (MTPc). A plausible mechanism was presented to show the influence of MTPc on the substrate pose in the active site. The possible determinant correlation between the findings of this research and the current studies on human tyrosinase role in the pigmentation process has been presented.





Similar content being viewed by others
Data Availability
The authors declare that the data supporting the findings of this study are available within the article and its supplementary information file.
Abbreviations
- hT:
-
Human tyrosinase
- LTy:
-
l-Tyrosine
- MePAPh:
-
4-[(4-Methylphenyl) azo]-phenol
- MT:
-
Mushroom tyrosinase
- MTPa:
-
MT active site pocket
- MTPc:
-
Non-specific binding site c
- PbLE:
-
Pyrus biossieriana leaves extract
References
Kim Y-J, Uyama H (2005) Tyrosinase inhibitors from natural and synthetic sources: structure, inhibition mechanism and perspective for the future. Cell Mol Life Sci CMLS 62(15):1707–1723. https://doi.org/10.1007/s00018-005-5054-y
Zolghadri S, Bahrami A, Hassan Khan MT, Munoz-Munoz J, Garcia-Molina F, Garcia-Canovas F, Saboury AA (2019) A comprehensive review on tyrosinase inhibitors. J Enzym Inhib Med Chem 34(1):279–309. https://doi.org/10.1080/14756366.2018.1545767
Couteau C, Coiffard L (2016) Overview of skin whitening agents: drugs and cosmetic products. Cosmetics 3(3):27. https://doi.org/10.3390/cosmetics3030027
Migas P, Krauze-Baranowska M (2015) The significance of arbutin and its derivatives in therapy and cosmetics. Phytochem Lett 13:35–40. https://doi.org/10.1016/j.phytol.2015.05.015
Nakajima M, Shinoda I, Fukuwatari Y, Hayasawa H (1998) Arbutin increases the pigmentation of cultured human melanocytes through mechanisms other than the induction of tyrosinase activity. Pigm Cell Res 11(1):12–17. https://doi.org/10.1111/j.1600-0749.1998.tb00705.x
Chakraborty AK, Funasaka Y, Komoto M, Ichihashi M (1998) Effect of arbutin on melanogenic proteins in human melanocytes. Pigm Cell Res 11(4):206–212. https://doi.org/10.1111/j.1600-0749.1998.tb00731.x
Sugimoto K, Nishimura T, Nomura K, Sugimoto K, Kuriki T (2004) Inhibitory effects of α-arbutin on melanin synthesis in cultured human melanoma cells and a three-dimensional human skin model. Biol Pharm Bull 27(4):510–514. https://doi.org/10.1248/bpb.27.510
Draelos ZD (2007) Skin lightening preparations and the hydroquinone controversy. Dermatol Ther 20(5):308–313. https://doi.org/10.1111/j.1529-8019.2007.00144.x
Qin L, Wu Y, Liu Y, Chen Y, Zhang P (2014) Dual effects of alpha-arbutin on monophenolase and diphenolase activities of mushroom tyrosinase. PLoS ONE 9(10):e109398. https://doi.org/10.1371/journal.pone.0109398
Garcia-Jimenez A, Teruel-Puche JA, Berna J, Rodriguez-Lopez JN, Tudela J, Garcia-Canovas F (2017) Action of tyrosinase on alpha and beta-arbutin: a kinetic study. PLoS ONE 12(5):e0177330. https://doi.org/10.1371/journal.pone.0177330
Walger E, Marlin N, Mortha G, Molton F, Duboc C (2021) Hydroxyl radical generation by the h2o2/cuii/phenanthroline system under both neutral and alkaline conditions: an EPR/Spin-Trapping Investigation. Appl Sci 11(2):687. https://doi.org/10.3390/app11020687
Pillar EA, Camm RC, Guzman MI (2014) Catechol oxidation by ozone and hydroxyl radicals at the air–water interface. Environ Sci Technol 48(24):14352–14360. https://doi.org/10.1021/es504094x
Pillar EA, Guzman MI (2017) Oxidation of substituted catechols at the air–water interface: production of carboxylic acids, quinones, and polyphenols. Environ Sci Technol 51(9):4951–4959. https://doi.org/10.1021/acs.est.7b00232
Hassani S, Gharechaei B, Nikfard S, Fazli M, Gheibi N, Hardré R, Legge RL, Haghbeen K (2018) New insight into the allosteric effect of l-tyrosine on mushroom tyrosinase during l-dopa production. Int J Biol Macromol 114:821–829. https://doi.org/10.1016/j.ijbiomac.2018.03.185
Shahaboddin M-E, Pouramir M, Moghadamnia A-A, Parsian H, Lakzaei M, Mir H (2011) Pyrus biossieriana Buhse leaf extract: an antioxidant, antihyperglycaemic and antihyperlipidemic agent. Food Chem 126(4):1730–1733. https://doi.org/10.1016/j.foodchem.2010.12.069
Haghbeen K, Rastgar Jazii F, Karkhane AA, Shareefi Borojerdi S (2004) Purification of tyrosinase from edible mushroom. Iran J Biotechnol 2:189–194
Haghbeen K, Tan EW (1998) Facile synthesis of catechol azo dyes. J Org Chem 63(13):4503–4505. https://doi.org/10.1021/jo972151z
Sasaki C, Ichitani M, Kunimoto K-K, Asada C, Nakamura Y (2014) Extraction of arbutin and its comparative content in branches, leaves, stems, and fruits of Japanese pear Pyrus pyrifolia cv. Kousui. Biosci Biotechnol Biochem 78(5):874–877. https://doi.org/10.1080/09168451.2014.893185
Prabhu K, Karar P, Hemalatha S, Ponnudurai K (2011) Isolation and spectral identification of arbutin from the roots of Viburnum erubescens Wall. ex DC. Int J Res Ayurveda Pharm (IJRAP) 2(3):889–892
Haghbeen K, Tan EW (2003) Direct spectrophotometric assay of monooxygenase and oxidase activities of mushroom tyrosinase in the presence of synthetic and natural substrates. Anal Biochem 312(1):23–32. https://doi.org/10.1016/S0003-2697(02)00408-6
Spada A, Palavicini S, Monzani E, Bubacco L, Casella L (2009) Trapping tyrosinase key active intermediate under turnover. Dalton Trans 33:6468–6471. https://doi.org/10.1039/B911946A
Ismaya WT, Rozeboom HJ, Weijn A, Mes JJ, Fusetti F, Wichers HJ, Dijkstra BW (2011) Crystal structure of Agaricus bisporus mushroom tyrosinase: identity of the tetramer subunits and interaction with tropolone. Biochemistry 50(24):5477–5486. https://doi.org/10.1021/bi200395t
Thomsen R, Christensen MH (2006) MolDock: a new technique for high-accuracy molecular docking. J Med Chem 49(11):3315–3321. https://doi.org/10.1021/jm051197e
Maki-Yonekura S, Yonekura K (2008) Electron digital imaging toward high-resolution structure analysis of biological macromolecules. Microsc Microanal 14(4):362. https://doi.org/10.1017/S1431927608080665
Wallace AC, Laskowski RA, Thornton JM (1995) LIGPLOT: a program to generate schematic diagrams of protein-ligand interactions. Protein Eng Des Sel 8(2):127–134. https://doi.org/10.1093/protein/8.2.127
Van der Spoel D, Lindahl E, Hess B (2013) The GROMACS development team, GROMACS User Manual version 4.6. 5. http://www.gromacs.org
Schüttelkopf AW, Van Aalten DM (2004) PRODRG: a tool for high-throughput crystallography of protein–ligand complexes. Acta Crystallogr D 60(8):1355–1363. https://doi.org/10.1107/S0907444904011679
SDBS. Spectral database for organic compounds. https://sdbs.db.aist.go.jp/sdbs/cgi-bin/cre_index.cgi
Hori I, Ki N, Kubo I (2004) Structural criteria for depigmenting mechanism of arbutin. Phytother Res 18(6):475–479. https://doi.org/10.1002/ptr.1456
Mann T, Gerwat W, Batzer J, Eggers K, Scherner C, Wenck H, Stäb F, Hearing VJ, Röhm K-H, Kolbe L (2018) Inhibition of human tyrosinase requires molecular motifs distinctively different from mushroom tyrosinase. J Invest Dermatol 138(7):1601–1608. https://doi.org/10.1016/j.jid.2018.01.019
Maeda K, Fukuda M (1996) Arbutin: mechanism of its depigmenting action in human melanocyte culture. J Pharmacol Exp Ther 276(2):765–769
Gheibi N, Saboury A, Haghbeen K, Rajaei F, Pahlevan A (2009) Dual effects of aliphatic carboxylic acids on cresolase and catecholase reactions of mushroom tyrosinase. J Enzym Inhib Med Chem 24(5):1076–1081. https://doi.org/10.1080/14756360802632658
de Faria RO, Rotuno Moure V, Balmant W, Lopes de Almeida Amazonas MA, Krieger N, Mitchell DA (2007) The tyrosinase produced by Lentinula boryana (Berk. & Mont.) Pegler suffers substrate inhibition by L-DOPA. Food Technol Biotechnol 45(3):334–340
Pettersson G (1986) Mechanistic origin of the sigmoidal rate behaviour of glucokinase. Biochem J 233(2):347–350. https://doi.org/10.1042/bj2330347
Seo S-Y, Sharma VK, Sharma N (2003) Mushroom tyrosinase: recent prospects. J Agric Food Chem 51(10):2837–2853. https://doi.org/10.1021/jf020826f
Haghbeen K, Khalili MB, Nematpour FS, Gheibi N, Fazli M, Alijanianzadeh M, Jahromi SZ, Sariri R (2010) Surveying allosteric cooperativity and cooperative inhibition in mushroom tyrosinase. J Food Biochem 34(2):308–328. https://doi.org/10.1111/j.1745-4514.2009.00280.x
Levitzki A, Koshland D (1969) Negative cooperativity in regulatory enzymes. Proc Natl Acad Sci USA 62(4):1121–1128. https://doi.org/10.1073/pnas.62.4.1121
Wilcox DE, Porras AG, Hwang YT, Lerch K, Winkler ME, Solomon EI (1985) Substrate analog binding to the coupled binuclear copper active site in tyrosinase. J Am Chem Soc 107(13):4015–4027. https://doi.org/10.1021/ja00299a043
Sendovski M, Kanteev M, Shuster V, Adir N, Fisherman A (2011) Crystal structure of tyrosinase from Bacillus magaterium in complex with inhibitor kojic acid. J Mol Biol 405:227–237. https://doi.org/10.2210/pdb3NQ1/pdb
Deri B, Kanteev M, Goldfeder M, Lecina D, Guallar V, Adir N, Fishman A (2016) The unravelling of the complex pattern of tyrosinase inhibition. Sci Rep 6(1):1–10. https://doi.org/10.1038/srep34993
Hassani S, Haghbeen K, Fazli M (2016) Non-specific binding sites help to explain mixed inhibition in mushroom tyrosinase activities. Eur J Med Chem 122:138–148. https://doi.org/10.1016/j.ejmech.2016.06.013
Ramsden CA, Riley PA (2014) Tyrosinase: the four oxidation states of the active site and their relevance to enzymatic activation, oxidation and inactivation. Bioorg Med Chem 22(8):2388–2395. https://doi.org/10.1016/j.bmc.2014.02.048
Hearing VJ Jr, Ekel TM, Montague PM, Nicholson JM (1980) Mammalin tyrosinase. Stoichiometry and measurement of reaction products. Biochim Biophys Acta 611(2):251–268. https://doi.org/10.1016/0005-2744(80)90061-3
Hornyak TJ (2018) Next time, save mushrooms for the pizza! J Invest Dermatol 138(7):1470–1472. https://doi.org/10.1016/j.jid.2018.03.003
Acknowledgements
This work was funded by National Institute for Genetic Engineering and Biotechnology of I.R. Iran through the annual research grant of Professor K. Haghbeen (Project Number 713).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest with the contents of this article, financial or otherwise.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ghofrani, N.S., Sheikhi, M., Amirzakaria, J.Z. et al. New Insight into the Interactions of Arbutin with Mushroom Tyrosinase. Protein J 40, 689–698 (2021). https://doi.org/10.1007/s10930-021-10004-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-021-10004-x