Abstract
The ERBB tyrosine kinase receptors and their ligands belong to a complex family that has diverse biological effects and expression profiles in the developing mammary glands, where its members play an essential role in translating hormone signals into local effects. While our understanding of these processes stems mostly from mouse models, there is the potential for differences in how this family functions in the mammary glands of other species, particularly in light of their unique histomorphological features. Herein we review the postnatal distribution and function of ERBB receptors and their ligands in the mammary glands of rodents and humans, as well as for livestock and companion animals. Our analysis highlights the diverse biology for this family and its members across species, the regulation of their expression, and how their roles and functions might be modulated by varying stromal composition and hormone interactions. Given that ERBB receptors and their ligands have the potential to influence processes ranging from normal mammary development to diseased states such as cancer and/or mastitis, both in human and veterinary medicine, a more complete understanding of their biological functions should help to direct future research and the identification of new therapeutic targets.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In his most acclaimed play Six Characters in Search of an Author, the Italian dramatist Luigi Pirandello brings six people to a stage where a company is rehearsing. These characters, who belong to the same extended family, claim to be “incomplete” and demand the director of the play to listen to their stories. An intricate family drama arises, ultimately leaving the director confused and the truth uncertain. In this review the characters are fifteen and the stage is the mammary gland. As for the mentioned author, the combined efforts of many scientists is needed to unravel the truth….
The mammary glands are unique in that they undergo a large proportion of their development after puberty under the influence of the reproductive hormones, where estrogen (E) directs ductal elongation into the surrounding stroma, while progesterone (P) and prolactin (PRL) promote branching, alveologenesis and lactogenesis. Despite the essential role of steroid hormones during mammary morphogenesis, only a subpopulation of mammary epithelial cells (MEC) are hormone receptor positive (HR+) due to their expression of receptors for E and P. A conclusion from genetically modified mice is that HR- epithelial cells, stromal cells or adipocytes rely on HR+ MEC to translate endocrine signals into paracrine cues [1, 2]. The mammary parenchyma in mice is very simple in its structure and originates from a branched epithelial tree surrounded by a predominantly adipose stroma (Fig. 1). Club shaped structures referred to as terminal end buds (TEBs) coordinate elongation of the mammary epithelium in pubescent mice. Similar structures are absent in cows, sheep and goats (collectively referred to as ruminants), where their mammary glands are characterized by complex ductal-lobular epithelial structures referred to as terminal ductal lobular units (TDLU), a more fibrous stroma, and less proximity to adipocytes (reviewed by Hovey et al. [3]). The mammary glands of pigs and humans have the combination of murine and ruminant elements, namely both TEB and TDLU, surrounded by a primarily fibrous stroma [4, 5]. The co-presence of TEB and TDLU has also been described in the mammary glands of dogs [6]. Several lines of evidence suggest that hormonal stimuli acting via local regulators underlie this strikingly-diverse morphological variation [7].
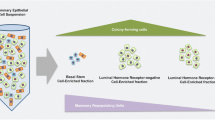
Mammary gland histomorphology and the epidermal growth factor (EGF) receptor (ERBB) family of receptors and their ligands in mice (left), ruminants (middle) and humans (right) during puberty and sexual maturity. The main systemic hormones involved during pre-gestational development are summarized. Asterisks link individual ERBB receptors to specific biological effects (blue arrows). Question marks represent incomplete information regarding ERBBs and their ligands. Abbreviations = AREG, amphiregulin; BTC, betacellulin; EGF, epidermal growth factor; EREG, epiregulin; HBEGF, heparin-binding EGF; NRG, neuregulin; Pituitary, pituitary gland; TEB, terminal end bud; TDLU, terminal ductal lobular unit; TGFα, transforming growth factor-α. Abbreviations for graphics are described in the figure. Figure created with CorelDraw X8.
Central to this local growth regulatory mechanism is the family of ERBB receptors and their ligands [8]. The ERBB family comprises four known receptors and numerous binding factors that are dynamically expressed throughout mammary gland development in mice [9] and are essential for postnatal mammary development. For instance, amphiregulin (AREG) mediates the actions of E at the onset of puberty by activating EGFR in the mammary stroma [10]. In a similar way, activation of ERBB4 is indispensable during lobuloalveolar development and lactogenesis [11]. Nevertheless, the role of ERBB receptors and their ligands has not been investigated for all stages of development, where numerous gaps remain, including for their spatial distribution. Likewise, the literature has an abundance of data for rodents relative to other species that have important and unique histomorphological characteristics [12].
Here we provide a comparative review of the expression and function of the ERBB receptors and their ligands during various stages of postnatal mammary development across several species. We also review how interactions between systemic hormones, ERBB receptors and various signaling pathways can impact development of the mammary gland and may account for differences in its morphology across species. By highlighting the paucity of data available for non-rodent species, we seek to convey that a thorough appreciation of ERBB biology in the mammary glands of different animal models is a necessary and overdue background for future research in both mammary physiology and pathology.
The Protagonists: The ERBB Receptors
Many excellent reviews detail the structure and activation of ERBB receptors elsewhere [13,14,15]. That said, there are several key points about the ERBB receptors with regard to their signaling versus other receptor tyrosine kinases that are worth highlighting. First, the EGF/ERBB family is notably large, having 11 known EGF-like growth factors that can variably bind 4 ERBB receptors. Second, the ERBBs are exclusively activated by either homo- or heterodimerization. Finally, a wide range of signaling pathways can be triggered in response to different ERBB homo- and heterodimers, leading to diverse outcomes. Aspects of these features are described below, with a focus on their implications for mammary biology.
Structure of the ERBB Receptors
The ERBB1/EGFR is an archetypal transmembrane protein that can activate tyrosine kinase activity. ERBB1 is a 170 kDa protein with a glycosylated extracellular domain that contains 4 subdomains arranged in tandem [16, 17], namely the homologous subdomains I and III (L1 and L2), and the cysteine-rich homologous subdomains II and IV (CR1 and CR2) [18,19,20]. Subdomain III is primarily involved in ligand binding (Fig. 2A). The other regions of ERBB1 include a single transmembrane domain, and an intracellular tyrosine kinase domain that confers the ability to activate autophosphorylation. Importantly, ERBB1 can translocate to the nucleus and function as a transcriptional regulator, or can interact with transcriptional factors such as signal transducer and activator of transcription (STAT) 3 and STAT5 [21], although the role of this mechanism during mammary morphogenesis has not been clarified. The ERBB2/human epidermal growth factor receptor 2 (HER2)/neu (185 kDa) is commonly referred to as orphan receptor: while it has an intrinsic kinase activity, the extracellular domain of ERBB2 has a permanent active conformation that prevents ligand binding (Fig. 2B), while also ensuring that the dimerization loop of subdomain IV remains exposed. The third family member, ERBB3 (160/180 kDa), has a defective cytoplasmic domain due to the absence of several key residues (Fig. 2B), which gives this receptor only 1/1000th the kinase activity of ERBB1 [22]. As a result, a role for either ERBB2 or ERBB3 at any stage of mammary development always implies their heterodimerization with another partner. Conversely, ERBB4/HER4 (180 kDa) can form both homodimers and heterodimers with other ERBB receptors, and can consequently undergo proteolytic processing to generate an 80 kDa cleaved intracellular region, which can translocate to the nucleus to regulate gene expression [23]. As will be discussed later, both the entire ERBB4 and the cleaved intracellular region have two isoforms (Cyt-1 and Cyt-2 for the cleaved domain; or JM-a, Cyt-1, and Cyt-2 for the full-length receptor) that confer the ability of ERBB4 to mediate both proliferative and differentiative processes in the developing gland.
(A) Structure of the epidermal growth factor (EGF) receptor (ERBB1), the archetype of the ERBB family. (B) Binding potential of EGF-like factors. Blue, extracellular region divided into L1-4 domains; green, intracellular tyrosine kinase domain; purple, intracellular C-terminus containing phosphorylatable tyrosine residues. Asterisks = defective extracellular domain of ERBB2, and defective intracellular domain of ERBB3. Figure created with CorelDraw X8.
ERBB Dimerization and Activation
Signaling downstream of the various ERBBs is triggered by ligand binding and subsequent receptor homo/heterodimerization [24, 25]. Only ERBB1 and ERBB4 have the ability to homodimerize to initiate signaling [26], whereas ERBB2 and ERBB3 can only heterodimerize [27,28,29]. As a result, ten different ERBB dimers are possible. Within these combinations, ERBB2 is the preferred partner for all other ERBB receptors. For example, ERBB2 and ERBB1 dimerize with the same affinity as the ERBB1 homodimer [30], and overexpression of ERBB2 and its heterodimerization with ERBB3 are both essential to induce breast tumor cell proliferation [31].
Due to the rigidity of its extracellular region, ERBB1 can only exist in an inactive (tethered) or active (extended) conformation. In the inactive form, subdomains II and IV are in close contact, whereas subdomains I and III are spaced apart, forming a potential ligand-binding “pocket”. After ligand binding, the ERBB1 “clicks” to its active form wherein subdomains I and III align, whereas subdomain II moves away from subdomain IV to expose its dimerization loop. Binding between the loops on the two dimerizing receptors induces the phosphorylation of intracellular residues. Each ERBB has a characteristic collection of phosphorylatable tyrosine residues (reviewed by Roskoski [32]). In addition, ERBB ligands have different affinities for ERBB receptors and trigger the phosphorylation of different sets of tyrosine residues within the intracellular non-catalytic region [33].
The ERBB Ligands
Eleven EGF-like peptide growth factors share a range of structural and functional properties. This family can be divided into three groups based on their binding affinity for one or more of the ERBB receptors [34]. One group of ligands, including epidermal growth factor (EGF), transforming growth factor alpha (TGFα), AREG and epigen (EPGN), can only bind ERBB1. The second group binds both ERBB1 and ERBB4, and includes epiregulin (EREG), betacellulin (BTC) and heparin-binding EGF (HBEGF), while the third group includes the neuregulins (NRG) that play a crucial role in neural development. Among these ligands, NRG1 and NRG2 can bind ERBB3 and ERBB4, whereas NRG3 and NRG4 can only bind ERBB4 (Fig. 2B).
The EGF-like ligands are expressed as type I transmembrane precursor proteins comprising an extracellular component, a transmembrane segment and a short intracellular region [35, 36]. The extracellular region includes one or more EGF-like motif(s), consisting of a hexameric cysteine sequence that creates three intramolecular disulfide bonds. This motif is essential and sufficient for ERBB-binding [37, 38]. In addition, the extracellular region of AREG and HBEGF includes an amino-terminal heparin-binding domain. The extracellular domain of ERBB ligands can be cleaved by various members of the A disintegrin and metalloproteinases (ADAMs) in a process referred to as protein ectodomain shedding [39, 40]. While the proteolytic release of this domain is generally necessary for binding ERBB receptors, some ligands that share structural similarities with HBEGF are biologically active even when bound to the membrane [41].
Distribution of ERBB Receptors and Ligands on the Mammary Stage
Mice
Puberty
Edery et al. [42] first reported the expression of two classes of EGF-binding receptors that had either low or high EGF-binding affinity in the epithelial and stromal compartment of the mammary glands in nulliparous mice, respectively. Receptor levels were highest in the glands of nulliparous mice, except for another peak during mid-gestation. A detailed profile of receptor expression then became available once the Erbb genes were identified (Table 1). The expression of Erbb1, Erbb2 and Erbb3 mRNA was often detected in the mammary glands of nulliparous females, even though only the phosphorylated form of ERBB1 and ERBB2 proteins was detected by immunoblotting of ERBB-immunoprecipitates, which raises questions about the sensitivity of these assays. The presence of Erbb4 mRNA and the ERBB3 protein in the mammary glands of nulliparous mice remains controversial [9, 43, 44]. Based on immunohistochemical analyses, ERBB1 and ERBB2 are equally-abundant in the epithelium and stroma of the mammary glands from pubescent female mice, then respectively become predominant in the stroma and epithelium by the time of sexual maturity [9]. The expression of ERBB1 mainly localizes to stromal cells surrounding the ducts and, to a lesser extent, to the cap cells of TEBs [45].
Using microarray analysis of the epithelium and stroma from the mammary glands of 5-week old female mice, Sternlicht and colleagues showed that of the various ERBB ligands, only Areg was abundant in peripubertal animals [46]. By comparison, others had previously indicated that Btc, Ereg, Hbegf, and Tgfa mRNA were expressed in the mammary glands of nulliparous female mice, and that expression for both Areg and Ereg was induced during puberty [9, 47]. While mRNA transcripts and protein for Areg and Tgfa predominate in the epithelium, HBEGF has been detected in both the epithelium and adipocytes [48].
Gestation
During gestation, the mammary glands of mice undergo a massive increase in secondary and tertiary ductal branching followed by the progression of alveolar buds into mature alveoli. Transcripts for all ERBB receptors have been identified in the glands of pregnant mice [9, 44, 49, 50] (Table 2). Compared to nulliparous females, approximately the same level of Erbb1, Erbb2 and Erbb3 gene expression was sustained in the mammary glands during gestation, while Erbb4 gene expression was induced at the onset of gestation [51, 52]. As gestation progresses, the abundance of all ERBB proteins increases in the mammary glands, where ERBBs are primarily expressed in the epithelium, and are either scarce or absent in the stroma [9, 44, 49].
Expression of mRNA for the ligands Areg, Btc, Ereg, Hbegf and Tgfa is abundant in the mammary glands of mice at the onset of gestation. While Areg mRNA abundance decreases thereafter, Btc, Ereg and Tgfa expression are only downregulated at the end of gestation, while Hbegf and Nrg1 levels are maintained until parturition. More specifically, Nrg1 gene expression peaks at day 15 of gestation in mice, while the level of TGFα protein decreases, which was hypothesized to stimulate the onset of whey acidic protein (WAP) expression [47, 53]. Compared to other ligands, the expression of Egf mRNA within the mammary glands progressively increases from mid-gestation [9].
Lactation and Involution
Abundance of transcripts encoding all four ERBB receptors generally decreases in the mammary gland during lactation (Table 3). Among the ERBB ligands, only the expression of Egf mRNA was consistently elevated within the mammary glands throughout lactation [9, 54, 55], where an EGF precursor was mostly detected within lactating alveolar cells in the apical part of the membrane [56]. During involution, there is a general re-expression of all ERBB receptors, and of Tgfa, Hbegf, Btc and Nrg1 mRNA [9].
Rats
The distribution of ERBB receptors and ligands in the mammary glands of rats warrants a separate description given that development of their glands differs subtly to that in mice (Table 1–3). Several studies have reported the differential expression of ERBB receptors in rats across the course of mammary development [57,58,59,60]. According to Darcy et al. [58, 59], proteins for all ERBB receptors are expressed in the mammary glands of peripubertal rats. Of note, ERBB1 was primarily expressed in TEBs, which contrasts to findings in mice, while ERBB3 was primarily expressed in the ducts and stroma. During puberty, ERBB4 was mostly expressed in TEBs, the surrounding stroma, and ducts. By contrast, ERBB2 was evenly distributed across epithelial and stromal compartments, consistent with its putative role as the preferred heterodimerization partner for other ERBB receptors. Interestingly, Dati et al. did not detect ERBB2 in the mammary glands of nulliparous rats, perhaps because its levels were below the detection limit of the immunoblot analysis. All the six possible heterodimers of ERBB1-4 were detected in the mammary glands of adult nulliparous rats [58].
During gestation, ERBB1, -2 and − 3 could be found in both the mammary epithelium of the ducts and in the stroma, whereas only ERBB1 was detectable in the alveoli. With the onset of lactation, expression of ERBB1 was downregulated in all mammary cell types, while the expression of ERBB2 and ERBB3 was minimal. By contrast, others reported that ERBB2 expression was increased in the mammary glands during lactation, alongside the presence of an ERBB2-MUC4/sialomucin complex, although the localization of this complex depended on the antibody used [60, 61].
By contrast, there is limited data for the spatial distribution of ERBB ligands in the mammary glands of rats. In fact, a complete profile of gene expression only exists for Tgfa mRNA, which localized to both the ductal and alveolar epithelium, and to 10–25% of stromal fibroblasts, in the mammary glands of virgin female rats [62]. These findings contrast to those of others who found that Tgfa mRNA expression was abundant in myoepithelial cells and fibroblasts at various stages of mammary development, including in the glands of nulliparous rats [55]. Proteins for TGFα and EGF were also found in the epithelium, stroma and adipose tissue in the mammary glands of pubescent rats [63], while transcripts for Areg, Ereg, Hbegf and Btc were also detected in the mammary glands of adult virgin nulliparous rats [64]. Akin to what was seen in pubescent rats, Tgfa mRNA was present in all MEC of pregnant mice, and in only a small proportion of fibroblasts. With the onset of lactation, the expression of Tgfa was markedly increased, and was exclusive to the epithelium [62].
Livestock Species
Several considerations emphasize how the normal mammary glands of livestock species might be more useful than those in rodents for studying the normal processes that dictate breast cancer risk. More specifically, domestic ruminants (cows, sheep and goats) are attractive models for study given the possibility of sampling repeated tissue and milk samples from single quarters of their udders. Furthermore, their mammary parenchyma is composed of TDLU that are also present in the human breast, but that are absent from the murine mammary gland. Likewise, different connective tissue components in the mammary glands of cattle and mice might explain why, when either human or bovine MEC were grafted into the mammary fat pads of mice, they failed to develop [65] versus mouse cells. Nevertheless, there is only sparse information regarding ERBB receptors in the mammary glands of ruminants (Tables 1, 2 and 3). From binding assays, ERBB1 was identified on purified plasma membranes from the mammary glands of pregnant and lactating cows [66]. Similarly, the capacity to bind TGFα was used to estimate the levels of ERBB1 in the mammary glands of sheep, which were higher in nulliparous females [67]. Lee and colleagues sought to describe the expression of ERBB receptors in the mammary glands of dairy cows prior to and during puberty (at 8 and 13 months of age, respectively). Expression of ERBB2, ERBB3, and to a lesser extent ERBB4, was detected in the mammary glands from both prepubertal and pubescent females, whereas the level of ERBB1 was much lower [68]. Despite several limitations of this study, the work highlights there may be a contrasting profile of expression for ERBB receptors in the bovine mammary glands compared to in mice.
Investigation of potential ERBB ligands in the mammary glands of ruminants has only focused on three members of the EGF-like protein family, namely EGF, TGFα and AREG. Evidence for the presence of EGF in the bovine mammary glands stems from a single study that reported elevated expression for both EGF and TGFA mRNA during mastitis [69]. In goats, EGF was also detected in prepartum mammary secretion [70], consistent with its presence in bovine colostrum [71]. Within the mammary glands of cows, TGFA was detected primarily at the onset of the dry period during regression and subsequent regeneration of the gland prior to parturition, while the presence of other transcripts suggested that other EGF-like factors may be expressed across all stages of mammary development [72, 73]. In contrast with those findings, Plath et al. (1997) described the highest levels of TGFA mRNA in the mammary glands of post-lactational non-pregnant cows during involution and, to a lesser extent, in the mammary glands of virgin heifers. In sheep, transcripts for AREG were detected in the mammary glands across a range of states, including in lambs, at late gestation (140d of a 150d gestation), and during lactation. The AREG protein localized to the nuclei and cytosol of luminal cells and in connective tissue of pregnant sheep, and in the nuclei of luminal cells in lactating sheep, but not in the mammary glands of female lambs [75].
The histomorphology of the mammary glands in female pigs displays striking similarities with that of the human breast [4], especially TDLU that are comparable, in both aspect and number, to those in humans. Even though a complete spatiotemporal description of ERBB receptors and ligands is not available for this species, our preliminary data indicate that the ERBB receptors and EGF-like ligands in the mammary glands of pigs are developmentally and hormonally coordinated.
Carnivores
The scarce information about ERBB receptors and ligands in the normal mammary glands of canids is often contradictory, and mostly derives from studies investigating the role of ERBB receptors in mammary tumors. The ERBB1 protein was detected in both the normal [76,77,78,79] and cancerous mammary glands of bitches [78, 80,81,82,83], while gene expression for ERBB1 has only been reported in mammary tumors [84]. Gama et al. localized membrane-associated and cytoplasmic staining for ERBB1 to myoepithelial cells and the perilobular stroma of the normal mammary glands. No variations in ERBB1 levels were apparent across physiological stages, except for a possible upregulation during estrus and the luteal phase [76]. Both ERBB2 protein and ERBB2 mRNA were expressed in the normal mammary glands of bitches, predominantly in the epithelium [85, 86]. Whereas ERBB2 is commonly overexpressed in canine mammary tumors, there is no clear association between its protein status and histological grade [82, 87,88,89], clinical parameters [86, 88] or prognosis [90, 91]. Likewise, there are inconsistent relationships between the level of ERBB2 mRNA and protein in both normal and cancerous tissues [85, 86, 92], perhaps reflecting either diversity among the studied populations or the use of different antibodies [87].
Mammary cancer in felids is commonly described as a model for HER2-overexpressing-hormone independent human breast cancers, which is why information about the ERBB family in these species almost exclusively focuses on ERBB2. Expression of ERBB2 in the normal mammary glands of queens, at both the mRNA and protein levels, ranges from absent to very low [93,94,95,96]. Higher epithelial immunoreactivity for ERBB2 was observed in malignant tumors compared to benign lesions, although not necessarily alongside higher gene expression [93, 95], or gene amplification [94, 97, 98].
Humans
The ERBB receptors have long been implicated in the etiology of breast cancer. Overexpression of ERBB1, which is associated with increased tumor size and poor outcome, occurs in 15 to 30% of breast cancers [99]; ERBB1 is also overexpressed in anywhere from 13 to 76% of triple negative breast cancers [100,101,102]. Overexpression of ERBB2 occurs in 20–30% of breast cancers, often due to ERBB2 gene amplification [103]. While mutations in ERBB1 are rare, mutations in ERBB2 do exist, leading to its constitutive activation that enhances cell growth and tumorigenicity. Overexpression of ERBB3 is also common in human breast cancers [104] and likely enhances ERBB1-signaling [105], whereas both the overexpression and downregulation of ERBB4 expression has been reported in mammary tumors. The ability of ERBB4 to both inhibit and promote epithelial proliferation in the normal mammary gland reflects its different roles in pathology, whereby ERBB4 expression is commonly related to a less aggressive phenotype, while activated ERBB4 can also promote proliferation, likely mediated by its intracellular domain [106].
Despite a clear role for ERBB receptors and their overexpression in breast cancers, much less is known about their biology in the normal human breast (Tables 1, 2 and 3). Certainly, ERBB1 is expressed in the normal breast given its transcript was detected in epithelial cells isolated from nonpregnant, nonlactating women [107, 108]. In normal tissue and hyperplastic or cancerous mammary lesions, ERBB1 localized to myoepithelial cells, alongside its variable presence in the normal epithelium [109,110,111]. Consistent with the presence of ERBB1, the ERBB1-ligands AREG and TGFα are either transcribed or translated by cultured normal breast epithelial cells [107, 108, 112], and TGFA levels were increased in pregnant as compared to nulliparous woman [62]. Moreover, EREG gene expression was specifically induced in the mammary glands of premenopausal women during the luteal phase, suggesting that EREG may mediate the actions of P in the human breast [113]. Regarding the role of ERBB1 in normal mammary glands, the activation of ERBB1 was required for differentiation of the myoepithelial lineage in immortalized human progenitor MEC that could differentiate into both luminal and myoepithelial cells in the presence of EGF. By contrast, AREG could only induce the luminal lineage, while TGFα only induced the myoepithelial lineage [114, 115]. Despite an absence of data regarding the role of ERBB-mediated signaling during lactogenesis, both EGF and HBEGF were detected in human milk and were associated with a protective effect against intestinal injury, which may further imply a potential role for all ERBB homo- and heterodimers in neonatal gut health [116].
Function of ERBB and Their Ligands in the Developing Mammary Glands: A Family Drama
Puberty and Maturity
Rodents
The mammary glands of pubescent mice and rats initiate their allometric development around 4 and 7 weeks of age respectively, as the ductal epithelium ramifies into the surrounding stroma. In mice there is a clear role for both E and P in developing the ductal network, whereas the mammary glands of virgin rats develop a more ductal-lobular morphology that is insensitive to P [117, 118]. Several lines of evidence indicate that this hormone-directed ductal expansion is mediated by local ERBB receptors and their ligands that not only amplify and modulate upstream systemic stimuli, but also confer variable morphogenic responses (Fig. 1).
Aspects of this ductal morphogenesis have been explored in vitro with a focus on ERBB1-dependent signaling. Different studies using primary cells and organoids from virgin mice highlighted a role for both EGF and TGFα in promoting ductal development [119, 120], and the potential for EGF to synergize with either P or PRL [121]. Another aspect highlighted by these experiments in vitro is that the regulation of ERBB-signaling likely depends on the composition of the mammary stroma. In fact, EGF-stimulated MEC produced their own basement membrane (BM) when cultured on collagen I-coated dishes, while deposition of BM was impaired on collagen IV, alongside the downregulation of ERBB1 [122].
A role for ERBB1 as a downstream effector of systemic hormone action in the mouse mammary gland in vivo is undisputed. Mice overexpressing a C-terminal-truncated ERBB1 had restricted growth of their mammary ducts as early as 5 weeks of age that was accompanied by fewer and smaller TEBs, followed by decreased ductal branching and density by 16 weeks [123]. On the other hand, mammary stroma expressing wild type ERBB1 could sustain the branching of either wildtype or Erbb1-null epithelium [43, 46, 124], consistent with the localization and activation of ERBB1 in the periductal stroma [9, 45]. There is also likely a role for multiple EGF-like ligands during this stage of development. Although EGF, TGFα and AREG all promote ductal morphogenesis, only AREG is indispensable for ductal growth [48]. This finding aligns with the demonstration that Areg is the only ERBB-ligand that is upregulated by E during puberty, and that shedding of AREG by ERα-positive epithelial cells, as induced by ADAM17, is essential for activating ERBB1 in the stroma prior to the stimulation of ductal elongation [1, 46]. However, questions remain as to how these receptors and ligands mediate the local response to various endocrine cues. First, additional endocrine or local factors besides E likely regulate the local synthesis of AREG. For example, both E and P can induce ductal growth and TEB formation in ovariectomized peripubertal mice, where P-induced development is associated with increased AREG expression, and is blocked by the inhibition of ERBB1 [125]; moreover, the recruitment of macrophages and eosinophils into the developing mammary glands of pubescent mice is triggered by both E and P, and is likely mediated by AREG-induced nuclear factor kappa B (NF-kB) - dependent cytokine expression [126]. Second, ERBB1 is also present in the epithelium, raising questions about its specific function in that cell type. Furthermore, the presence of other ERBBs in the epithelium may create potential heterodimerization partners for ERBB1, consistent with the demonstration that ductal development is impaired when epithelial Erbb1 is silenced. Indeed, the transplantation of Erbb2-null mammary epithelium into the cleared mammary fat pad of wildtype mice led to impaired ductal development, delayed TEB appearance (despite an increased number of branch points) and severe disruption of TEB histomorphology that corresponded to altered distribution of smooth muscle actin, as well as P- and E-cadherins [127]. A similar phenotype was recorded when epithelial expression of Erbb2 was silenced [128]. However, as mentioned earlier, ERBB2 is an orphan receptor, raising the prospect that either heterodimerization of epithelial ERBB1 with ERBB2 plays a minor role in mammary development during puberty, as proposed by Sternlicht et al. [46], or that ERBB3 and/or ERBB4 fulfil specific roles during this stage. Indeed, when Erbb3-null mammary epithelium was transplanted into the wildtype mammary fat pad, the developmental deficits were comparable to those seen in Erbb2-null transplants, and persisted into adulthood [129]. Reduced outgrowth of the ductal network was also recorded in mice homozygous for Erbb3Δ85 (an allele lacking PI3K-binding sites), further confirming a role for the ERBB3/PI3K pathway during glandular morphogenesis [130]. Given these findings, the hypothesis that ERBB3 functions as an ERBB2 heterodimerization partner is further supported by the demonstration that implants of heregulin (isoform of NRG1) could induce ductal development in prepubescent mice [131], and could stimulate ductal morphogenesis even when Erbb1 was deleted [46]. Among the various ERBB, there are also reasons to speculate that ERBB4 may serve a more precocious role than generally acknowledged. Indeed, the Cyt-1 and Cyt-2 splice variants of ERBB4 can either inhibit growth while enhancing differentiation, or promote proliferation. For example, overexpression of ERBB4-Cyt-1 (either as its full-length form, or as the intracellular domain of JMa-Cyt-1) accounts for the anti-proliferative effects of ERBB4 during puberty that results in suppressed ductal elongation and TEB development, followed by enhanced lobuloalveolar budding in sexually-mature female mice. The overexpression of Cyt-2 (intracellular domain of ERBB4) caused hyperproliferation of epithelial cells in both ducts and lobuloalveoli, which did not occur with overexpression of full length JMa-Cyt-2, suggesting that the membrane-associated ERBB4 might interact with the Cyt-2 intracellular domain [132, 133]. Taken together, these findings suggest that, despite the low level of ERBB4 expression in the mammary glands during puberty, ERBB4 might serve a mild growth-suppressing effect on the epithelium during this phase, followed by a potential differentiative effect during gestation and lactation. Interestingly, Schroeder et al. only detected ERBB3 protein in the mammary glands of pregnant mice, while the expression of ERBB4 in the mammary glands during puberty was uncertain [9]. Combined, these observations highlight the need for a more detailed spatiotemporal analysis of ERBB receptors in the murine mammary gland.
Lastly, there is a compelling likelihood that other EGF-like factors besides AREG also contribute to mammary gland development during puberty. For instance, EGF and TGFα both induce ductal morphogenesis in vivo [45, 54, 134]. Furthermore, both inflammatory and ERBB1-signaling related molecules such as Erbb1, Hbegf and Adam17 were upregulated in a unique E-independent phenotype of ductal growth induced by dietary conjugated linoleic acid [135]. Indeed, E-independent activation of ERBB1 by AREG or other growth factors such as HBEGF might contribute to normal mammary gland development, an effect that may be even more pronounced during obesity-associated inflammation that promotes tumorigenesis of certain E-independent cancer subtypes.
As previously mentioned, all ERBB receptors were detected in the mammary glands of pubertal and adult virgin rats, consistent with the effect of ERBB-dependent signaling on both growth and differentiation of mammary epithelial organoids from virgin rats cultured in reconstituted BM [58, 59]. Similar to findings in mice [125], the expression of Areg in the mammary epithelium of 18 week old virgin rats could be stimulated by either E or P, but was maximal in response to their combination. Crosstalk between E, P and ERBB1 was also proposed whereby AREG (released in response to E and P) and P (leading to activation of the P receptor-B) could synergize through various ERBB1-downstream signaling pathways to stimulate epithelial proliferation [64]. Interestingly, in the same study gene expression for other ERBB1 ligands was modulated by either E or P. Specifically, Ereg and Btc were upregulated by E, whereas Tgfa was induced by P, raising the possibility that TGFα may facilitate cross talk between activated P receptor-B and signaling downstream of ERBB1.
Ruminants
Allometric growth of the mammary epithelium in nulliparous female cows (heifers) commences at around 2–3 months of age and prior to puberty, or potentially earlier [136, 137]. Similarly, allometric growth in female sheep can start as early as 4 weeks of age [138]. By the time of puberty (approximately 8 months of age for sheep, and 11 months for cattle), the epithelial ducts have elongated into the surrounding mammary fat pad, which is more fibrous than the predominantly adipose stroma of mice. The mammary parenchyma in pubertal ruminants is characterized by a complex ductal-lobular parenchyma surrounded by distinguishable inter- and intralobular connective tissues [139] (Fig. 1). These unique histomorphological and tissue interactions raise the prospect for differential roles for ERBB receptors and their ligands in these species, like may also occur in humans, and distinct from those in model species such as mice.
Evidence in vitro points to roles for EGF, TGFα and AREG in the regulation of mammary growth in ruminants. When comparing ligands, recombinant human EGF (rhEGF) was more effective than rhTGFα for stimulating the proliferation of bovine MEC [140], whereas both were more effective than rhAREG. By contrast, rhTGFα promoted DNA synthesis in alveolar epithelial cells from nulliparous ewes in a dose-dependent manner more efficiently than rhEGF, independent of developmental stage, although DNA synthesis was delayed in cells from nulliparous females versus those that were pregnant [67, 141]. Similarly, rbTGFα stimulated greater proliferation by bovine MEC than did rhEGF [73, 142, 143], raising the question of whether this different outcome reflected that the cells were from a different physiological state, or whether there is species-specificity at play for human and bovine TGFα. Beyond its direct actions, EGF synergized with insulin-like growth factors (IGF-I, IGF-II and des3-IGF-I) to induce mitosis in undifferentiated MEC in collagen [144, 145], while TGFα synergized with insulin (INS) to induce DNA synthesis in MEC from nulliparous ewes [141].
The positive effect of EGF on ruminant mammary epithelium was confirmed by transplantation of mammary tissue from mid-pregnant heifers into ovariectomized-sialoadenectomized athymic mice. In this model, EGF had almost no effect on epithelial proliferation, whereas it synergized with the combination of E and P to restore the level of DNA synthesis back to that in ovary-intact, steroid-treated mice [146]. These findings raise the prospect that additional interactions between ligand-stimulated proliferation and ovarian hormones, as well as perhaps the nature of the stroma, create a stage- and tissue-specific growth-modulating environment for the mammary epithelium.
Gestation
The mammary glands undergo pronounced and rapid changes during gestation - not only through extensive proliferation of the epithelium that gives rise to alveoli arising from the ductal structures, but also cytologically and biochemically in preparation for the onset of lactation. In this section we review the potential roles for ERBB receptors and their ligands in the mammary glands across various species, while encouraging the reader to keep in mind that the final transition during secretory activation occurs alongside terminal differentiation of the mammary epithelium and a final wave of proliferation.
Rodents
There is clear in vitro evidence that signaling downstream of the ERBB receptors can direct lobuloalveolar development and differentiation of the mammary epithelium in rodents. One illustration of this effect is in whole organ culture, where a first wave of lobuloalveolar development can be induced in mammary glands from steroid-primed pubescent mice following their exposure to INS, PRL, aldosterone and hydrocortisone (HC), while EGF is essential for triggering a second wave of growth [147]. In testing the interrelationships between these hormonal regulators, others investigated the interactive responses of the mammary epithelium to EGF and PRL. Interestingly, PRL inhibited EGF-dependent DNA synthesis in various models in vitro, while also promoting the transcription of Egf itself [148, 149]. Surprisingly, EGF and PRL both independently upregulated the pre- and post-transcriptional levels of β-casein, whereas their combination, in the presence of INS and HC, had the opposite effect [55, 149, 150]. Some have proposed that this negative interaction might be an artefact of a supraphysiological concentration of INS [151], which aligns with evidence showing that EGF enhances the secretion of β-casein in the presence of PRL and dexamethasone [152]. A combined interpretation of these studies is that EGF coordinates epithelial proliferation and differentiation, while PRL progressively inhibits this effect and simultaneously promotes the transcription of Egf as an autocrine factor to sustain secretory differentiation. Variation in the relative abundance of ERBB1 homo- and heterodimers in MEC during this period may well also account for the switching of EGF towards a pro-differentiative function. Neuregulins also specify the functional state of MEC in vitro, as was revealed using antisense nucleotides against endogenous Nrg; these inhibited lobuloalveolar development in cultured whole mammary glands, whereas exogenous Nrg reversed this inhibitory effect, leading to increased expression of β-casein and WAP [44].
Evidence in vivo also suggests that ERBB1 and ERBB2 might support lobuloalveolar development during gestation, despite being unable to compensate for the effects of Erbb3 deletion [49] (Fig. 3). As a matter of fact, mice overexpressing a dominant negative-Erbb2 in their mammary glands displayed abnormal, condensed lobuloalveoli by the end of gestation [51]. Furthermore, several ERBB1-ligands are essential during this developmental stage when deletion of Areg leads to impaired lobuloalveolar development, which is exacerbated by the additional deletion of Egf and Tgfa [40]. More specifically, TGFα was more effective than EGF for inducing lobuloalveolar growth [146, 147]. Unlike ERBB1 and ERBB2, an indispensable role exists for ERBB3 during gestation, as evidenced by the fact that Erbb3-null mice have defective lobuloalveolar expansion, decreased epithelial proliferation and enhanced apoptosis of luminal epithelial cells, together with impaired activation of AKT and STAT5. Intriguingly, failed proliferation and differentiation of the mammary epithelium upon the loss of ERBB3 recovered during late gestation and early lactation, while phosphorylation of ERBB4 (Tyr1056) increased [49, 129]. In fact, ERBB4 might compensate for the defects induced by the loss of ERBB3 in late gestation, where the ERBB4 isoform JMa-Cyt-1 (the full-length, antiproliferative form of ERBB4), which harbors a PI3K-binding site at Tyr1056, would likely heterodimerize with ERBB3 and induce differentiation.
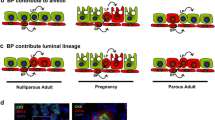
Mammary gland histomorphology and the epidermal growth factor (EGF) receptor (ERBB) family of receptors and their ligands in mice (left), ruminants (middle) and humans (right) during pregnancy and lactation. The main systemic hormones involved during gestation and lactation are summarized. Asterisks link individual ERBB receptors to specific biological effects (blue arrows). Question marks represent incomplete information regarding ERBBs and their ligands. Abbreviations = AREG, amphiregulin; BTC, betacellulin; EGF, epidermal growth factor; EREG, epiregulin; HBEGF, heparin-binding EGF; NRG, neuregulin; Pituitary, pituitary gland; TDLU, terminal ductal lobular unit; TGFα, transforming growth factor-α. Abbreviations for graphics are explained in the figure. Figure created with CorelDraw X8.
Not by chance, ERBB4 plays a major role in the mammary glands during gestation, moreso than other ERBB receptors. Lactating primiparous Erbb4-null mice have scarce lobuloalveolar differentiation with reduced activation of STAT5 shortly after parturition, likely due to deficiencies that arose during gestation [153]. Multiparous female mice harboring Cre-lox mediated deletion of both epithelial Erbb4 alleles had severely-compromised lobuloalveolar growth as early as day 13.5 of gestation, due to both impaired proliferation and defective differentiation [50]. This dual proliferative and differentiative effect for ERBB4 might reflect the opposing functions of ERBB4-Cyt-1 and Cyt-2 isoforms (derived by the alternative splicing of the ERBB4 intracellular domain), as evidenced in HC11 cells overexpressing Cyt-1 or Cyt-2 ICD isoforms that undergo apoptosis or proliferation, respectively [133]. The sparse ductal development described in pubertal mice overexpressing ERBB4-Cyt-1 (either the intracellular domain Cyt-1, or the full-length isoform JM-a-Cyt-1) extended into mid-gestation, after which developmental deficiencies abated. On the other hand, epithelial cells in ducts and lobuloalveoli did not proliferate when the full-length isoform JM-a-Cyt-2 was overexpressed in the mammary glands, perhaps because the membrane-bound fraction of ERBB4-Cyt-2 had a compensatory effect. An interesting model was proposed whereby epithelial expression of Complement subcomponents Clr/Cls, Uegf, Bmpl and zona pellucida-like domain-containing protein 1 (CUZD1) would complex with STAT5 following activation of the PRL receptor (PRLR), increasing local expression of NRG1, EREG (both increased in the mammary gland during gestation [9]) and EPGN; in turn, these ligands would promote epithelial proliferation and differentiation via ERBB1 and ERBB4 [154]. Such an interaction between ERBB4, PRLR and STAT5 would not only enhance the compensatory role of ERBB4 in lobuloalveolar expansion and differentiation (as hypothesized by Williams et al. [49]), but might also partially account for the unique differentiative properties of ERBB4, which may override ERBB1-dependent growth [155]. Finally, there is a clear positive role for NRG1 during lobuloalveolar development, consistent with the pro-differentiative effect of ERBB3 and ERBB4. Specifically, deficiency of the NRG1 isoform NRG1-α leads to the abnormal condensation of lobuloalveoli and decreased epithelial proliferation [156].
Ruminants
Pregnancy in heifers and ewe lambs is typically initiated at 15 months and 12–18 months of age, with corresponding gestation lengths of about 9 months and 5 months, respectively. Extensive alveologenesis and stromal remodeling in the mammary glands of ruminants, including in cattle [157] and small ruminants [158, 159], occurs primarily during gestation, while mammary DNA content remains constant after parturition. There are several lines of in vitro evidence to support a role for the various EGF-like ligands in the mammary glands of pregnant ruminants. Both TGFα and EGF are mitogenic for cultured MEC from pregnant cows [73]. Likewise, treating explants from midpregnant Holstein heifers with EGF or TGFα was sufficient to induce maximal DNA synthesis, despite suboptimal activation of ERBB1 [160]. In a similar way, MEC from pregnant ewes initiated DNA synthesis in response to EGF, TGFα, or AREG; what is more, TGFα compensated for the inhibition of DNA-synthesis induced by heparin. On the other hand, heparin maximized the synergistic response to TGFα and either INS or IGF-1, probably by decreasing the interactions between AREG and INS/IGF-1. In essence, a complex yet unresolved interplay between EGF-like ligands and IGF-related ligands seems to occur during gestation, with the potential for modulation by heparin-like molecules [141].
Other approaches have also been used to demonstrate the mitogenic effects of various EGF-like ligands in the mammary glands of ruminants during gestation in vivo (Fig. 3). Both hEGF and bTGFα induced mammary growth and DNA synthesis in late pregnant cows when they were instilled into half the udder quarters [143]. In a different study, explants of mammary tissue from mid-pregnant cows were transplanted to mice, which were then primed with steroid hormones. In this model, PRL and HC, alone or synergistically, decreased EGF-dependent DNA-synthesis by affecting both abundance and kinase activity of ERBB1. An intriguing question that warrants investigation is whether PRL can simultaneously synergize with EGF to induce epithelial differentiation in the mammary glands of ruminants, as proposed in rodents [160,161,162].
Lactation
Rodents
As outlined above, ERBB4 is indispensable for terminal differentiation of the mammary gland, as highlighted by the fact that Erbb4-dominant negative mice had distended ducts bearing persistently condensed, lipid-filled lobuloalveoli, as well as reduced epithelial proliferation and expression of β-casein, WAP and α-lactalbumin [50, 52, 153]. These defects coincided with impaired activation of STAT5, potentially due to crosstalk between the PRLR and ERBB4 [155]. Not surprisingly, pups nursed by either Erbb4-null dams or those expressing a dominant-negative Erbb4 in their mammary glands had higher mortality and lower body weight due to inadequate milk production by the dams [50, 153]. While the activation of ERBB4 in the mammary glands is crucial for lactation, its requirement is not exclusive (Fig. 3). Specifically, the phenotype of immature, condensed lobuloalveoli filled with fat droplets and scarce proteinaceous material also existed in transgenic Erbb2-dominant negative mice, supporting the notion that ERBB2 might normally heterodimerize with ERBB4 to facilitate terminal differentiation of the alveolar epithelium [51]. Moreover, defects in the mammary glands of Erbb3-null mice appeared primarily around the end of gestation, then progressively improved around parturition, perhaps due to the concurrent upregulation of ERBB4 [49]. However, this proposal requires further validation, given that mice homozygous for Erbb3Δ85 have defective proliferation and differentiation within their mammary glands (giving rise to thicker epithelium, less expanded alveoli, decreased β-casein and WAP protein expression and secretion) that persists to day 3 of lactation [130]. Lastly, the presence of mutated Erbb1, as found in waved-2 mice, is associated with smaller mammary glands, a lower parenchyma/fat ratio, decreased secretions and vacuolation, and higher mortality of pups [163]. A role for ERBB1 also extends to the health of the neonate, where Erbb1-null newborn mice developed hemorrhagic enteritis [164], while EGF supplementation reduced the incidence of necrotizing enterocolitis in newborn rats [165].
Given the potential role(s) for all ERBB receptors during lactation, it is perhaps not surprising that several EGF-like ligands have also been implicated during this period. For example, a deficiency of NRG1 led to the distention of scarcely arranged lobuloalveoli, alongside decreased expression of β-casein and less epithelial proliferation [156], similar to the phenotype in Erbb4-dominant negative mice. Meanwhile, the correlation between TGFα expression and lactation efficiency remains controversial. Specifically, whereas TGFα enhanced progression towards terminal differentiation when it was overexpressed in the mammary glands, in association with precocious lobuloalveolar development (reviewed by Schroeder and Lee [166]), lactation was often impaired in those animals, and multiparous mice often developed hyperplastic lesions. What is more, the extensive lobuloalveolar development in mice overexpressing TGFα was not associated with the expression of milk proteins such as WAP [53, 167], nor with the accumulation of secretory products [131]. One possible explanation for how transgenic overexpression of TGFα suppresses lactation potential is that TGFα impairs the upregulation of PRLR expression around parturition [168]. In the same vein, simultaneous overexpression of TGFβ (a mammary growth suppressor) and TGFα led to the TGFα-transgenic mice having normal mammary glands, but impaired lactation [169]. It should be noted that the role for steroid hormones was not isolated in all the studies, making it difficult to define the local, TGFα-dependent effects on mammary differentiation [170]. In summary, TGFα likely supports terminal differentiation in gestation, but a successful lactation reflects a precise balance between progressive downregulation of TGFα, PRLR and steroid receptor interactions, and the local expression of other growth factors.
Ruminants
There is a paucity of data regarding whether signals downstream of epithelial ERBB receptors affect lactational competence in ruminants (Fig. 3). Some evidence indicates that EGF accumulates in the mammary glands of pre-parturient cows [71] and small ruminants [70, 171], while EGF is detectable in milk at the beginning of lactation, but then declines afterwards. While there was a 10-fold increase in the amount of EGF in the colostrum of lactating ewes a day after parturition, intravenous injection of EGF had no effect on the body weight of their lambs, on colostrum composition, or on milk yield [171]. As in other physiological stages, TGFα is more potent than EGF for inducing DNA synthesis in MEC from lactating ewes, especially in synergy with INS or IGF-1 [141].
Involution
Upon weaning there are massive changes within the gland, including milk stasis and degradation of its components, epithelial apoptosis, remodeling of the extracellular matrix, and phagocytosis, that all proceed over time to varying extents across species. While the first, reversible phase of involution is acutely responsive to milk statis, leading to the up- or downregulation of epithelial STAT3 and STAT5 respectively, the second phase is irreversible and reflects the involvement of systemic factors and macrophages. We posit that activated ERBB receptors are involved in both phases, particularly by virtue of their ability to trigger STAT signaling and to mediate macrophage-dependent tumor cell motility [172].
Rodents
Activated ERBB3 and ERBB4 apparently counteract remodeling of the mammary gland, as demonstrated by accelerated involution in transgenic mice homozygous for Erbb3Δ85 (lacking PI3K-binding sites) [130] and those harboring the epithelial deletion of Erbb4 [50]; in the latter case the remodeling defects are reminiscent of altered AKT signaling. Conversely, deletion of Erbb2 in the mammary epithelium was not constantly associated with defective involution [128].
It is perhaps not surprising that overexpression of TGFα in the mammary glands of mice impairs epithelial apoptosis after parturition (as opposed to the pro-apoptotic effect of TGFβ1) given its aforementioned mitogenic effect during earlier developmental stages [173]. A significant decrease in the abundance of Nrg1 and Nrg2 transcripts was recorded in the involuting mammary glands of heterozygous p63 transgenic mice, consistent with the pro-survival activity of ERBB3 and ERBB4. As a matter of fact, p63 exerts a pro-survival effect on basal cells during early involution by counteracting both the STAT3 and p53 pathways, while promoting the transcription of Nrg1 and Nrg2, which are activators of the ERBB4-STAT5 pathway [174].
Ruminants
Lobuloalveolar regression during involution is never complete in ruminants [175], particularly in dairy cows where lactation and gestation overlap. Surprisingly, there are few insights as to the role of ERBB receptors and their ligands during involution of the mammary glands in ruminants. The levels of TGFA were high in the involuting mammary glands of dairy cows [72], which could potentially contribute to the sustained lobuloalveolar phenotype. In vitro, EGF had an antiapoptotic role on BME-UV cells cultured in FBS-deficient medium, which might reflect a compensatory pro-survival effect in the mammary gland during involution [176]. Interestingly, the transcription of both EGF and TGFA was increased during mastitis [69]. Given the economic impact of this condition on the dairy industry, the role for ERBB-signaling during mastitis and the associated tissue inflammation and repair warrants further investigation.
ERBBs in Dogs and Cats: Only Background Actors?
The mammary glands of dogs and cats are often proposed as models for human breast cancer. Indeed, specific cancer subtypes such as the spontaneous feline carcinoma, which is a possible model of HER2 overexpressing breast cancer [93], and canine hormone-dependent cancers [177], share many similarities with the human disease. These analogies, together with a high incidence of ERBB1 and HER2 overexpression in both canine and feline mammary cancers, explain the considerable interest in ERBB signaling in veterinary medicine. Ironically, almost nothing can be said about the contribution of ERBB receptors towards the physiological development of the canine or feline mammary glands. Logic would suggest that the ERBB receptors have a dynamic profile of expression across the course of mammary development in carnivores, as in mice. Since a major unsolved question in veterinary oncology is whether a precocious ovariectomy significantly decreases the incidence of mammary cancers [178], the contribution of ERBB-signaling around the time of spaying might be a main factor affecting tumorigenesis in our pets.
Behind the Scenes: ERBB-signaling
The many combinations of ERBB receptors and their ligands, ERBB positive/negative effectors, and potential biological outcomes, highlights how ERBB-signaling is a multilayered signaling network [15]. Defining these pathways is critical for understanding the main interactions and their outcomes. To complete the script, we will briefly review the principal signaling pathways that can be activated by homo- or heterodimerizing ERBB receptors, and their contribution during normal mammary development and morphogenesis.
RAS/ERK Pathway
ERBB1, -B2 and -B4 all activate the RAS/ERK pathway due to their multiple binding sites for Grb2 and SHC. The final effector of this signaling cascade, ERK, can either translocate to the nucleus, or activate additional transcriptional factors, apoptosis regulators or hormone receptors. Activation of the RAS/ERK pathway yields a range of phenotypes including proliferation, differentiation, and survival, as well as angiogenesis and cell motility, which all occur in the mammary gland [13, 179]. Unique combinations of ERBB and RAS/ERK may function during mammary gland development. For example, the formation of mammospheres by duct/limited progenitor cells in the murine mammary gland was mediated by ERK1/2, presumably through the release of AREG [180]. In addition, the formation of ductal branches arising from cultured mouse and rat primary MEC in response to TGFα, EGF or AREG was also ERK-dependent [64, 120, 181]. Moreover, specification of the myoepithelial lineage in human breast organoids, which is triggered by EGF-activated ERBB1, depends on ERK1/2 [115]. Based on these findings, activation of the RAS/ERK pathway downstream of ERBB receptors should be essential for mammary development from puberty until adulthood. Indeed, Schroeder and colleagues [9] showed that activated ERK1/2 colocalized with ERBB1 and ERBB2 in the mammary glands of virgin mice, while it was almost absent during gestation and lactation. On the other hand, transgenic mice overexpressing TGFα and/or PRL revealed that these peptides cooperate to activate ERK1/2 resulting in a number of developmental abnormalities, while also rendering the mammary epithelium insensitive to E [182]. The significance of this relationship in the mammary glands of pregnant and lactating animals remains to be addressed. This synergy between TGFα and PRL might involve the modulation of ERBB3 and/or ERBB4 heterodimers, given that the levels of ERBB1 (the only TGFα receptor) are the same in TGFα and TGFα/PRL bitransgenic mice [183].
PI3K/AKT Pathway
Both ERBB3 and the JMa-Cyt-1 isoform of ERBB4 can activate PI3K/AKT signaling due to their multiple binding sites for the p85 subunit of PI3K. Alternatively, PI3K can also be indirectly activated by RAS. Meanwhile AKT, which plays a central role in signaling downstream of ERBB receptors, affects a number of biological processes crucial for mammary gland development, including glucose metabolism, cell survival, protein synthesis, and cell motility [13, 184]. As previously mentioned, activated ERBB3-ERBB4 heterodimers and AKT can interact to direct mammary growth and differentiation. Indeed, the outgrowth of mammary ducts was severely impaired in mice bearing alleles for ERBB3 that lacked P85a subunits [130]. AKT is also an essential mediator for lobuloalveolar expansion during late gestation, as evidenced by the fact that scant lobuloalveolar development in pregnant Erbb3-null mice was also associated with impaired activation of AKT. Furthermore, the ERBB4 isoform JMa-Cyt could partially reverse the lobuloalveolar defects and induction of AKT. While there are no data regarding the role of ERBB-AKT interactions during mammary development beyond gestation, these are likely to be decisive given that the activation of AKT1 and AKT2 isoforms can lead to either delayed or accelerated involution [185, 192, 193].
STATs
The ERBB receptors can activate STAT proteins both directly and indirectly through JAK, which in turn phosphorylates STATs prior to nuclear translocation. Different STAT proteins have precise expression patterns and occupy distinct roles during mammary development [186], although evidence for an interaction between STAT and ERBB proteins only exists for late gestation and lactation. During these stages, STAT5 serves as a hub to signal lobuloalveolar development [187] and milk protein gene expression [188]. Both Erbb3-null [49] and Erbb4-null [50] mice have downregulated STAT5 in their mammary glands, together with the aforementioned developmental and lactogenic defects. While ERBB3 seems unable to induce phosphorylation of STAT5, there is the potential for ERBB4 to be a strong activator. In fact, not only is ERBB4 activated downstream of pSTAT5 in the mammary glands of mice [154], but it can also synergize with the PRLR to activate STAT5 in HC11 cells [131], where the interaction of ERBB4 and STAT5 in the epithelium is crucial for lobuloalveolar development. On the other hand, the ERBB1-dependent phosphorylation of STAT5 is indispensable [189] only in the stroma of pregnant and lactating mice.
During involution, STAT3 is constitutively expressed, but is specifically phosphorylated for its role in apoptosis and remodeling after weaning [190]. While there is no clear link between elements of ERBB-signaling and STAT3 during involution, there is evidence from multiple breast cancer lines that ERBB2-ERBB3 heterodimers can activate STAT3 upon NRG binding, in a signaling network that also involves the PR and the ERK1/2 pathway [191]. In a similar way, STAT1 expression peaks during involution, although its role is not entirely clear [192], while STAT5 is downregulated. Despite being expressed in the mammary glands throughout development, expression of STAT6 is upregulated during gestation, when it contributes to expansion of the alveolar lineage [193]. There is no information about how STAT6 and ERBB receptors interact during these stages, although transactivation of the EGFR by STAT6-mediated release of HBEGF has been described in peritoneal macrophages of mice [194].
Conclusion
The family of characters reviewed here - the ERBB receptors and their ligands - are as complex and dynamic as any human family! A wide body of evidence highlights how in all species the ERBB family finds its strength through the interplay between different ERBBs and their ligands, rather than through the exclusive role for any one receptor or ligand at any stage. On the other hand the literature, albeit patchy, highlights that the biological functions of these molecules differs across species; a case in point being that there is a more diverse expression pattern for the ERBBs and their ligands in the mammary glands of rats and ruminants relative to in mice. In the same way, the relative importance of various EGF-like ligands in regulating mammary development appears to differ across species. We propose that the distinctive stromal composition of the mammary glands is an underlying regulator of ERBB biology and function in this context (Figs. 1 and 3). Finally, we suggest that mediation of upstream hormonal cues by ERBB receptors helps confer the range of histomorphological phenotypes across species. Central to this hypothesis could be the actions of P, which independently is non-proliferative in the mammary glands of primates, ruminants, rats and pigs [4, 195, 196]. While there is mixed information about whether P alone is proliferative in humans [197], the breast is also characterized by a complex ductal-lobular morphology. Elucidating how ERBB signaling converges with the actions of systemic hormones, both on the epithelium and the surrounding stroma, could further define the regulation of breast morphogenesis. At the same time, many unanswered questions remain regarding the role of ERBB receptors and ligands in veterinary pathology, where a deep understanding of their biology would help to identify better targets for treatments against mammary cancers in dogs and cats, as well as against mastitis in dairy animals and even humans.
All in all, while the family of ERBB receptors and ligands is now aged and mature, its members remain partly confused on the mammary gland stage. Like in Pirandello’s theatrical piece Six Characters in Search of an Author, mammary biologists are now asked to give each of these fifteen characters a voice and to fully unravel the roles they are playing in the mammary gland.
References
Ciarloni L, Mallepell S, Brisken C. Amphiregulin is an essential mediator of estrogen receptor alpha function in mammary gland development. Proc Natl Acad Sci U S A 2007;104:5455–60. https://doi.org/10.1073/pnas.0611647104
Brisken C, Park S, Vass T, Lydon JP, O’Malley BW, Weinberg RA. A paracrine role for the epithelial progesterone receptor in mammary gland development. Proc Natl Acad Sci U S A 1998;95:5076–81. https://doi.org/10.1073/PNAS.95.9.5076
Hovey RC, Mcfadden TB, Akers RM. Regulation of Mammary Gland Growth and Morphogenesis by the Mammary Fat Pad: A Species Comparison. vol. 4. 1999.
Horigan KC, Trott JF, Barndollar AS, Scudder JM, Blauwiekel RM, Hovey RC. Hormone interactions confer specific proliferative and histomorphogenic responses in the porcine mammary gland. Domest Anim Endocrinol 2009;37:124–38. https://doi.org/10.1016/j.domaniend.2009.04.002
Russo J, Rivera R, Russo IH. Influence of age and parity on the development of the human breast. Breast Cancer Res Treat 1992;23:2–218.
Santos M, Marcos R, Faustino AMR. Histological study of canine mammary gland during the oestrous cycle. Reprod Domest Anim 2010;45. https://doi.org/10.1111/J.1439-0531.2009.01536.X
Hovey RC, Trott JF, Vonderhaar BK. Establishing a framework for the functional mammary gland: from endocrinology to morphology. J Mammary Gland Biol Neoplasia 2002;7:17–38. https://doi.org/10.1023/a:1015766322258
Brisken C, Ataca D. Endocrine hormones and local signals during the development. Wiley Interdiscip Rev Dev Biol 2015;4:181–95. https://doi.org/10.1002/wdev.172
Schroeder JA, Lee DC. Dynamic expression and activation of ERBB receptors in the developing mouse mammary gland. Cell Growth Differ Mol Biol J Am Assoc Cancer Res 1998;9:451–64.
Sternlicht MD, Sunnarborg SW. The ADAM17-amphiregulin-EGFR axis in mammary development and cancer. J Mammary Gland Biol Neoplasia 2008;13:181–94. https://doi.org/10.1007/s10911-008-9084-6
Muraoka-Cook RS, Feng SM, Strunk KE, Earp SH. ErbB4/HER4: role in mammary gland development, differentiation and growth inhibition. J Mammary Gland Biol Neoplasia 2008;13:235–46. https://doi.org/10.1007/s10911-008-9080-x
Rowson AR, Daniels KM, Ellis SE, Hovey RC. Growth and development of the mammary glands of livestock: a veritable barnyard of opportunities. Semin Cell Dev Biol 2012;23:557–66. https://doi.org/10.1016/j.semcdb.2012.03.018
Wang Z. ErbB receptors and cancer. Methods Mol. Biol., vol. 1652, Humana Press Inc.; 2017, p. 3–35. https://doi.org/10.1007/978-1-4939-7219-7_1
Olayioye MA, Neve RM, Lane HA, Hynes NE. The ErbB signaling network: receptor heterodimerization in developmnet and cancer. EMBO J 2000;19:3159–67. https://doi.org/10.1093/emboj/19.13.3159
Yarden Y, Sliwkowski MX. Untangling the ErbB signalling network. Nat Rev Mol Cell Biol 2001;2:127–37. https://doi.org/10.1038/35052073
Cohen S, Ushiro H, Stoscheck C, Chinkers M. A native 170,000 epidermal growth factor receptor-kinase complex from shed plasma membrane vesicles. J Biol Chem 1982;257:1523–31. https://doi.org/10.1016/S0021-9258(19)68224-4
Ullrich A, Coussens L, Hayflick JS, Dull TJ, Gray A, Tam AW, et al. Human epidermal growth factor receptor cDNA sequence and aberrant expression of the amplified gene in A431 epidermoid carcinoma cells. Nature 1984;309:418–25. https://doi.org/10.1038/309418a0
Bajaj M, Waterfield MD, Schlessinger J, Taylor WR, Blundell T. On the tertiary structure of the extracellular domains of the epidermal growth factor and insulin receptors. Biochim Biophys Acta 1987;916:220–6. https://doi.org/10.1016/0167-4838(87)90112-9
Lax I, Johnson A, Howk R, Sap J, Bellot F, Winkler M, et al. Chicken epidermal growth factor (EGF) receptor: cDNA cloning, expression in mouse cells, and differential binding of EGF and transforming growth factor alpha. Mol Cell Biol 1988;8:1970–8. https://doi.org/10.1128/mcb.8.5.1970
Ward CW, Hoyne PA, Flegg RH. Insulin and epidermal growth factor receptors contain the cysteine repeat motif found in the tumor necrosis factor receptor. Proteins Struct Funct Bioinforma 1995;22:141–53. https://doi.org/10.1002/prot.340220207
Lin SY, Makino K, Xia W, Matin A, Wen Y, Kwong KY, et al. Nuclear localization of EGF receptor and its potential new role as a transcription factor. Nat Cell Biol 2001;3:802–8. https://doi.org/10.1038/ncb0901-802
Shi F, Telesco SE, Liu Y, Radhakrishnan R, Lemmona MA. ErbB3/HER3 intracellular domain is competent to bind ATP and catalyze autophosphorylation. Proc Natl Acad Sci U S A 2010;107:7692–7. https://doi.org/10.1073/pnas.1002753107
Sardi SP, Murtie J, Koirala S, Patten BA, Corfas G. Presenilin-dependent ErbB4 nuclear signaling regulates the timing of astrogenesis in the developing brain. Cell 2006;127:185–97. https://doi.org/10.1016/J.CELL.2006.07.037
Schechter Y, Hernaez L, Schlessingert J, Cuatrecasas P. Local aggregation of hormone-receptor complexes is required for activation by epidermal growth factor. Nature 1979;278:835–8. https://doi.org/10.1038/278835a0
Yarden Y, Schlessinger J. Epidermal growth factor induces rapid, reversible aggregation of the purified epidermal growth factor receptor. Biochemistry 1987;26:1443–51. https://doi.org/10.1021/bi00379a035
Ferguson KM, Darling PJ, Mohan MJ, Macatee TL, Lemmon MA. Extracellular domains drive homo- but not hetero- dimerization of erbB receptors. EMBO J 2000;19:4632–43. https://doi.org/10.1093/emboj/19.17.4632
Berger MB, Mendrola JM, Lemmon MA. ErbB3/HER3 does not homodimerize upon neuregulin binding at the cell surface. FEBS Lett 2004;569:332–6. https://doi.org/10.1016/j.febslet.2004.06.014
Wehrman TS, Raab WJ, Casipit CL, Doyonnas R, Pomerantz JH, Blau HM. A system for quantifying dynamic protein interactions defines a role for Herceptin in modulating ErbB2 interactions. Proc Natl Acad Sci U S A 2006;103:19063–8. https://doi.org/10.1073/pnas.0605218103
Citri A, Skaria KB, Yarden Y. The deaf and the dumb: the biology of ErbB-2 and ErbB-3. Exp Cell Res 2003;284:54–65. https://doi.org/10.1016/S0014-4827(02)00101-5
Hendriks BS, Opresko LK, Wiley HS, Lauffenburger D. Quantitative analysis of HER2-mediated effects on HER2 and epidermal growth factor receptor endocytosis. Distribution of homo- and heterodimers depends on relative HER2 levels. J Biol Chem 2003;278:23343–51. https://doi.org/10.1074/jbc.M300477200
Holbro T, Beerli RR, Maurer F, Koziczak M, Barbas CF, Hynes NE. The ErbB2/ErbB3 heterodimer functions as an oncogenic unit: ErbB2 requires ErbB3 to drive breast tumor cell proliferation. Proc Natl Acad Sci U S A 2003;100:8933–8. https://doi.org/10.1073/pnas.1537685100
Roskoski R. The ErbB/HER family of protein-tyrosine kinases and cancer. Pharmacol Res 2014;79:34–74. https://doi.org/10.1016/j.phrs.2013.11.002
Wilson KJ, Gilmore JL, Foley J, Lemmon MA, Riese Ii DJ. Functional selectivity of EGF family peptide growth factors: implications for cancer. Pharmacol Ther 2008;122:1–8. https://doi.org/10.1016/j.pharmthera.2008.11.008
Jones JT, Akita RW, Sliwkowski MX. Binding specificities and affinities of egf domains for ErbB receptors. FEBS Lett 1999;447:227–31. https://doi.org/10.1016/S0014-5793(99)00283-5
Harris AL, Nicholson S, Richard J, Sainsbury C, Farndon J, Wright C. Epidermal growth factor receptors in breast cancer: Association with early relapse and death, poor response to hormones and interactions with neu. J Steroid Biochem 1989;34:123–31. https://doi.org/10.1016/0022-4731(89)90072-1
Gullick WJ. The type 1 growth factor receptors and their ligands considered as a complex system. Endocr Relat Cancer 2001;8:75–82. https://doi.org/10.1677/erc.0.0080075
Nagata K, Kohda D, Hatanaka H, Ichikawa S, Matsuda S, Yamamoto T, et al. Solution structure of the epidermal growth factor-like domain of heregulin-alpha, a ligand for p180erbB-4. EMBO J 1994;13:3517–23. https://doi.org/10.1002/j.1460-2075.1994.tb06658.x
Jacobsen NE, Abadi N, Sliwkowski MX, Reilly D, Skelton NJ, Fairbrother WJ. High-resolution solution structure of the EGF-like domain of heregulin-α. Biochemistry 1996;35:3402–17. https://doi.org/10.1021/bi952626l
Hinkle CL, Sunnarborg SW, Loiselle D, Parker CE, Stevenson M, Russell WE, et al. Selective roles for tumor necrosis factor α-converting enzyme/ADAM17 in the shedding of the epidermal growth factor receptor ligand family: the juxtamembrane stalk determines cleavage efficiency. J Biol Chem 2004;279:24179–88. https://doi.org/10.1074/JBC.M312141200
Sahin U, Weskamp G, Kelly K, Zhou HM, Higashiyama S, Peschon J, et al. Distinct roles for ADAM10 and ADAM17 in ectodomain shedding of six EGFR ligands. J Cell Biol 2004;164:769–79. https://doi.org/10.1083/JCB.200307137
Dong J, Opresko LK, Chrisler W, Orr G, Quesenberry RD, Lauffenburger DA, et al. The membrane-anchoring domain of epidermal growth factor receptor ligands dictates their ability to operate in Juxtacrine Mode. Mol Biol Cell 2005;16:2984–98. https://doi.org/10.1091/mbc.E04
Edery M, Pang K, Larson L, Colosi T, Nandi S. Epidermal growth factor receptor levels in mouse mammary glands in various physiological states. Endocrinology 1985;117:405–11. https://doi.org/10.1210/endo-117-1-405
Sebastian J, Richards RG, Walker MP, Wiesen JF, Werb Z, Derynck R, et al. Activation and function of the epidermal growth factor receptor and erbB-2 during mammary gland morphogenesis. Cell Growth Differ 1998;9:777–85.
Yang Y, Spitzer E, Meyer D, Sachs M, Niemann C, Hartmann G, et al. Sequential requirement of hepatocyte growth factor and neuregulin in the morphogenesis and differentiation of the mammary gland. J Cell Biol 1995;131:215–26. https://doi.org/10.1083/jcb.131.1.215
Coleman S, Silberstein GB, Daniel CW. Ductal morphogenesis in the mouse mammary gland: evidence supporting a role for epidermal growth factor. Dev Biol 1988;127:304–15. https://doi.org/10.1016/0012-1606(88)90317-X
Sternlicht MD, Sunnarborg SW, Kouros-mehr H, Yu Y, Lee DC, Werb Z, et al. Mammary ductal morphogenesis requires paracrine activation of stromal EGFR via ADAM17-dependent shedding of epithelial amphiregulin. Development 2006;132:3923–33. https://doi.org/10.1242/dev.02314
Herrington EE, Ram TG, Salomon DS, Johnson GR, Gullick W, Kenney N. Expression of epidermal growth factor-related proteins in the aged adult mouse mammary gland and their relationship to tumorigenesis. J Cell Physiol 1997;170:47–56. https://doi.org/10.1002/(sici)1097-4652(199701)170:1%3C47::aid-jcp6%3E3.0.co;2-l.
Luetteke NC, Qiu TH, Fenton SE, Troyer KL, Riedel RF, Chang A, et al. Targeted inactivation of the EGF and amphiregulin genes reveals distinct roles for EGF receptor ligands in mouse mammary gland development. Development 1999;126:2739–50.
Williams MM, Vaught DB, Joly MM, Hicks DJ, Sanchez V, Owens P, et al. ErbB3 drives mammary epithelial survival and differentiation during pregnancy and lactation. Breast Cancer Res 2017;19:105. https://doi.org/10.1186/s13058-017-0893-7
Long W, Wagner K-U, Lloyd KCK, Binart N, Shillingford JM, Hennighausen L, et al. Impaired differentiation and lactational failure of Erbb4-deficient mammary glands identify ERBB4 as an obligate mediator of STAT5. Development 2003;130:5257–68. https://doi.org/10.1242/dev.00715
Jones FE, Stern DF. Expression of dominant-negative ErbB2 in the mammary gland of transgenic mice reveals a role in lobuloalveolar development and lactation. Oncogene 1999;18:3481–90. https://doi.org/10.1038/sj.onc.1202698
Jones FE, Welte T, Fu X, Stern DF. ErbB4 signaling in the mammary gland is required for lobuloalveolar development and Stat5 activation during lactation. J Cell Biol 1999;147:77–87. https://doi.org/10.1083/jcb.147.1.77
Lin CQ, Dempsey PJ, Coffey RJ, Bissell MJ. Extracellular matrix regulates whey acidic protein gene expression by suppression of TGF-α in mouse mammary epithelial cells: studies in culture and in transgenic mice. J Cell Biol 1995;129:1115–26. https://doi.org/10.1083/jcb.129.4.1115
Snedeker SM, Brown CF, DiAugustine RP. Expression and functional properties of transforming growth factor α and epidermal growth factor during mouse mammary gland ductal morphogenesis. Proc Natl Acad Sci U S A 1991;88:276–80. https://doi.org/10.1073/pnas.88.1.276
Fenton SE, Sheffield LG. Lactogenic hormones increase epidermal growth factor messenger RNA content of mouse mammary glands. Biochem Biophys Res Commun 1991;181:1063–9. https://doi.org/10.1016/0006-291X(91)92045-L
Brown CF, Teng CT, Pentecost BT, Diaugustine RP. Epidermal growth factor precursor in mouse lactating mammary gland alveolar cells. Mol Endocrinol 1989;3:1077–83. https://doi.org/10.1210/mend-3-7-1077
Sagsoz H, Ketani MA. The role of estrogen receptors, erbB receptors, vascular endothelial growth factor and its receptors, and vascular endothelial growth inhibitor in the development of the rat mammary gland. Growth Factors 2010;28:379–93. https://doi.org/10.3109/08977194.2010.495718
Darcy KM, Zangani D, Wohlhueter AL, Huang R-Y, Vaughan MM, Russell JA, et al. Changes in ErbB2 (her-2/neu), ErbB3, and ErbB4 during growth, differentiation, and apoptosis of normal rat mammary epithelial cells. J Histochem Cytochem 2000;48:63–80. https://doi.org/10.1177/002215540004800107
Darcy KM, Wohlhueter AL, Zangani D, Vaughan MM, Russell JA, Masso-Welch PA, et al. Selective changes in EGF receptor expression and function during the proliferation, differentiation and apoptosis of mammary epithelial cells. Eur J Cell Biol 1999;78:511–23. https://doi.org/10.1016/S0171-9335(99)80077-6
Dati C, Maggiora P, Puech C, Bortoli M De, Escot C. Expression of the erb B-2 proto-oncogene during differentiation of the mammary gland in the rat. Cell Tissue Res 1996;285:403–10. https://doi.org/10.1007/s004410050656
Price-Schiavi SA, Andrechek E, Idris N, Li P, Rong M, Zhang J, et al. Expression, location, and interactions of ErbB2 and its intramembrane ligand Muc4 (sialomucin complex) in rat mammary gland during pregnancy. J Cell Physiol 2005;203:44–53. https://doi.org/10.1002/jcp.20200
Liscia DS, Merlo G, Ciardiello F, Kim N, Smith GH, Callahan R, et al. Transforming growth factor-α messenger RNA localization in the developing adult rat and human mammary gland by in situ hybridization. Dev Biol 1990;140:123–31. https://doi.org/10.1016/0012-1606(90)90059-R
Brown NM, Wang J, Cotroneo MS, Zhao Y-X, Lamartiniere CA. Prepubertal genistein treatment modulates TGF-α, EGF and EGF-receptor mRNAs and proteins in the rat mammary gland. Mol Cell Endocrinol 1998;144:149–65. https://doi.org/10.1016/s0303-7207(98)00106-3
Kariagina A, Xie J, Leipprandt JR, Haslam SZ. Amphiregulin mediates estrogen, progesterone, and EGFR signaling in the normal rat mammary gland and in hormone-dependent rat mammary cancers. Horm Cancer 2010;1:229–44. https://doi.org/10.1007/s12672-010-0048-0
Sheffield LG. Organization and growth of mammary epithelia in the mammary gland fat pad. J Dairy Sci 1988;71:2855–74. https://doi.org/10.3168/jds.S0022-0302(88)79881-1
Spitzer E, Grosse R. EGF receptors on plasma membranes purified from bovine mammary gland of lactating and pregnant animals. Biochem Int 1987;14:581–8.
Moorby CD, Taylor JA, Forsyth IA. Transforming growth factor-α: receptor binding and action on DNA synthesis in the sheep mammary gland. J Endocrinol 1995;144:165–71. https://doi.org/10.1677/joe.0.1440165
Lee B-W, Kim Y-H, Jeon B-S, Yoon B-I, Singh NK, Kim W-H, et al. Expression of ErbB receptors in the pre-pubertal and pubertal virgin mammary glands of dairy cows. Korean J Vet Res 2012;52:269–73.
Sheffield LG. Mastitis increases growth factor messenger ribonucleic acid in bovine mammary glands. J Dairy Sci 1997;80:2020–4. https://doi.org/10.3168/jds.S0022-0302(97)76146-0
Dehnhard M, Claus R, Munz O, Weiler U. Course of epidermal growth factor (EGF) and insulin-like growth factor I (IGF-I) in mammary secretions of the goat during end-pregnancy and early lactation. J Vet Med Ser A 2000;47:533–40. https://doi.org/10.1046/j.1439-0442.2000.00315.x
Iacopetta BJ, Grieu F, Horisberger M, Sunahara GI. Epidermal growth factor in human and bovine milk. Acta Paediatr 1992;81:287–91. https://doi.org/10.1111/j.1651-2227.1992.tb12227.x
Koff MD, Plaut K. Expression of transforming growth factor-α-like messenger ribonucleic acid transcripts in the bovine mammary gland. J Dairy Sci 1995;78:1903–8. https://doi.org/10.3168/jds.S0022-0302(95)76815-1
Zurfluft LL, Boltent SL, Byatt JC, McGrath MF, Tou JS, Zupec ME, et al. Isolation of genomic sequence encoding a biologically active bovine TGF- α protein. Growth Factors 1990;3:257–66. https://doi.org/10.3109/08977199009003668
Plath A, Einspanier R, Peters F, Sinowatz F, Schams D. Expression of transforming growth factors alpha and beta-1 messenger RNA in the bovine mammary gland during different stages of development and lactation. J Endocrinol 1997;155:501–11. https://doi.org/10.1677/joe.0.1550501
Forsyth IA, Taylor JA, Keable S, Turvey A, Lennard S. Expression of amphiregulin in the sheep mammary gland. Mol Cell Endocrinol 1997;126:41–8. https://doi.org/10.1016/s0303-7207(96)03967-6
Donnay I, Devleeschouwer N, Leclercq G, Verstegen J. Changes in oestrogen, progesterone and epidermal growth factor receptor concentrations and affinities during the oestrous cycle in the normal mammary gland and uterus of dogs 1995;19:101–13. https://doi.org/10.1007/bf01839276
Nerurkar VR, Seshadri R, Mulherkar R, Ishwad CS, Lalitha VS, Naik SN. Receptors for epidermal growth factor and estradiol in canine mammary tumors. Int J Cancer 1987;40:230–2. https://doi.org/10.1002/ijc.2910400218
Queiroga FL, Pérez-Alenza D, Silvan G, Peña L, Peña P, Illera JC. Positive correlation of steroid hormones and EGF in canine mammary cancer. J Steroid Biochem Mol Biol 2009;115:9–13. https://doi.org/10.1016/j.jsbmb.2009.01.018
Gama A, Gärtner F, Alves A, Schmitt F. Immunohistochemical expression of epidermal growth factor receptor (EGFR) in canine mammary tissues. Res Vet Sci 2009;87:432–7. https://doi.org/10.1016/J.RVSC.2009.04.016
Carvalho MI, Pires I, Prada J, Ferreira AF, Queiroga FL. Positive interplay between CD3 + T-lymphocytes and concurrent COX-2/EGFR expression in canine malignant mammary tumors. Anticancer Res 2015;35:2915–20.
Damasceno KA, Ferreira E, Estrela-Lima A, Gamba C de O, Miranda FF, Alves MR, et al. HER-2 and EGFR mRNA expression and its relationship with versican in malignant matrix-producing tumors of the canine mammary gland. PLoS One 2016;11:e0160419. https://doi.org/10.1371/journal.pone.0160419
Guimarães MJ, Carvalho MI, Pires I, Prada J, Gil AG, Lopes C, et al. Concurrent expression of cyclo-oxygenase-2 and epidermal growth factor receptor in canine malignant mammary tumours. J Comp Pathol 2014;150:27–34. https://doi.org/10.1016/j.jcpa.2013.07.005
Carvalho MI, Guimarães MJ, Pires I, Prada J, Silva-Carvalho R, Lopes C, et al. EGFR and microvessel density in canine malignant mammary tumours. Res Vet Sci 2013;95:1094–9. https://doi.org/10.1016/j.rvsc.2013.09.003
Matsuyama S, Nakamura M, Yonezawa K, Shimada T, Ohashi F. Expression patterns of the erbB subfamily mRNA in canine benign and malignant mammary tumors. J Vet Med Sci 2001;63:949–54. https://doi.org/10.1292/jvms.63.949
Martín De Las Mulas J, Ordás J, Millán Y, Fernández-Soria V, Ramón Y Cajal S. Oncogene HER-2 in canine mammary gland carcinomas: an immunohistochemical and chromogenic in situ hybridization study. Breast Cancer Res Treat 2003;80:363–7. https://doi.org/10.1023/A:1024929730165
Seung B-J, Cho S-H, Kim S-H, Lim H-Y, Sur J-H. Quantitative analysis of HER2 mRNA expression by RNA in situ hybridization in canine mammary gland tumors: comparison with immunohistochemistry analysis. PLoS One 2020;15:e0229031. https://doi.org/10.1371/journal.pone.0229031
Burrai GP, Tanca A, De Miglio MR, Abbondio M, Pisanu S, Polinas M, et al. Investigation of HER2 expression in canine mammary tumors by antibody-based, transcriptomic and mass spectrometry analysis: is the dog a suitable animal model for human breast cancer? Tumor Biol 2015;36:9083–91. https://doi.org/10.1007/s13277-015-3661-2
Ressel L, Puleio R, Ruggero Loria G, Vannozzi I, Millanta F, Caracappa S, et al. HER-2 expression in canine morphologically normal, hyperplastic and neoplastic mammary tissues and its correlation with the clinical outcome. Res Vet Sci 2013;94:299–305. https://doi.org/10.1016/j.rvsc.2012.09.016
Dutra AP, Granja NVM, Schmitt FC, Cassali GD. c-erbB-2 expression and nuclear pleomorphism in canine mammary tumors. Brazilian J Med Biol Res 2004;37:1673–81. https://doi.org/10.1590/S0100-879X2004001100013
Shinoda H, Legare ME, Mason GL, Berkbigler JL, Afzali MF, Flint AF, et al. Significance of ERα, HER2, and CAV1 expression and molecular subtype classification to canine mammary gland tumor. J Vet Diagnostic Investig 2014;26:390–403. https://doi.org/10.1177/1040638714527289
Hsu WL, Huang HM, Liao JW, Wong ML, Chang SC. Increased survival in dogs with malignant mammary tumours overexpressing HER-2 protein and detection of a silent single nucleotide polymorphism in the canine HER-2 gene. Vet J 2009;180:116–23. https://doi.org/10.1016/j.tvjl.2007.10.013
Damasceno KA, Ferreira E, Estrela-Lima A, Bosco Y, Silva LP, Barros ALB, et al. Relationship between the expression of versican and EGFR, HER-2, HER-3 and CD44 in matrixproducing tumours in the canine mammary gland. Histol Histopathol 2016;31:675–88. https://doi.org/10.14670/HH-11-705
De Maria R, Olivero M, Iussich S, Nakaichi M, Murata T, Biolatti B, et al. Spontaneous feline mammary carcinoma is a model of HER2 overexpressing poor prognosis human breast cancer. Cancer Res 2005;65:907–12.
Ordás J, Millán Y, Dios R, Reymundo C, de las Mulas JM. Proto-oncogene HER-2 in normal, dysplastic and tumorous feline mammary glands: an immunohistochemical and chromogenic in situ hybridization study. BMC Cancer 2007;7:179. https://doi.org/10.1186/1471-2407-7-179
Ferreira D, Soares M, Correia J, Adega F, Ferreira F, Chaves R. Assessment of ERBB2 and TOP2a gene status and expression profile in feline mammary tumors: findings and guidelines. Aging (Albany NY) 2019;11:4688–705. https://doi.org/10.18632/aging.102079
Millanta F, Calandrella M, Citi S, Della Santa D, Poli A. Overexpression of HER-2 in feline invasive mammary carcinomas: an immunohistochemical survey and evaluation of its prognostic potential. Vet Pathol 2005;42:30–4. https://doi.org/10.1354/vp.42-1-30
Muscatello LV, Di Oto E, Sarli G, Monti V, Foschini MP, Benazzi C, et al. HER2 amplification status in feline mammary carcinoma: a tissue microarray-fluorescence in situ hydridization-based study. Vet Pathol 2019;56:230–8. https://doi.org/10.1177/0300985818808531
Soares M, Correia J, Rodrigues P, Simões M, De Matos A, Ferreira F. Feline HER2 protein expression levels and gene status in feline mammary carcinoma: Optimization of immunohistochemistry (IHC) and in situ hybridization (ISH) techniques. Microsc. Microanal., vol. 19, Cambridge University Press; 2013, p. 876–82. https://doi.org/10.1017/S1431927613001529
Wang Editor Z. ErbB Receptor Signaling Methods and Protocols Methods in Molecular Biology 1652, n.d.
Masuda H, Zhang D, Bartholomeusz C, Doihara H, Hortobagyi GN, Ueno NT. Role of epidermal growth factor receptor in breast cancer. Breast Cancer Res Treat 2012;136:331–45. https://doi.org/10.1007/s10549-012-2289-9
Meseure D, Vacher S, Drak Alsibai K, Trassard M, Susini A, Le Ray C, et al. Profiling of EGFR mRNA and protein expression in 471 breast cancers compared with 10 normal tissues: a candidate biomarker to predict EGFR inhibitor effectiveness. Int J Cancer 2012;131:1009–10. https://doi.org/10.1002/IJC.26434
Viale G, Rotmensz N, Maisonneuve P, Bottiglieri L, Montagna E, Luini A, et al. Invasive ductal carcinoma of the breast with the “triple-negative” phenotype: prognostic implications of EGFR immunoreactivity. Breast Cancer Res Treat 2009;116:317–28. https://doi.org/10.1007/S10549-008-0206-Z
Bose R, Kavuri SM, Searleman AC, Shen W, Shen D, Koboldt DC, et al. Activating HER2 mutations in HER2 gene amplification negative breast cancer. Cancer Discov 2013;3:224–37. https://doi.org/10.1158/2159-8290.CD-12-0349
Ma J, Lyu H, Huang J, Liu B. Targeting of erbB3 receptor to overcome resistance in cancer treatment. Mol Cancer 2014;13:1–9. https://doi.org/10.1186/1476-4598-13-105/FIGURES/2
Mishra R, Patel H, Alanazi S, Yuan L, Garrett JT. HER3 signaling and targeted therapy in cancer. Oncol Rev 2018;12:45–62. https://doi.org/10.4081/ONCOL.2018.355
Jones FE. HER4 intracellular domain (4ICD) activity in the developing mammary gland and breast cancer. J Mammary Gland Biol Neoplasia 2008;13:247–58. https://doi.org/10.1007/s10911-008-9076-6
Bates SE, Valverius EM, Ennis BW, Bronzert DA, Sheridan JP, Stampfer MR, et al. Expression of the transforming growth factor-/epidermal growth factor receptor pathway in normal human breast epithelial cells. Endocrinology 1990;126:596–607. https://doi.org/10.1210/endo-126-1-596
Valverius EM, Bates SE, Stampfer MR, Clark R, McCormick F, Salomon DS, et al. Transforming growth factor α production and epidermal growth factor receptor expression in normal and oncogene transformed human mammary epithelial cells. Mol Endocrinol 1989;3:203–14. https://doi.org/10.1210/mend-3-1-203
Santini D, Ceccarelli C, Tardio ML, Taffurelli M, Marrano D. Immunocytochemical expression of epidermal growth factor receptor in myoepithelial cells of the breast. Appl Immunohistochem Mol Morphol 2002;10:29–33. https://doi.org/10.1097/00129039-200203000-00005
Tsutsumi Y, Naber SP, DeLellis RA, Wolfe HJ, Marks PJ, McKenzie SJ, et al. Neu oncogene protein and epidermal growth factor receptor are independently expressed in benign and malignant breast tissues. Hum Pathol 1990;21:750–8. https://doi.org/10.1016/0046-8177(90)90035-4
Damjanov I, Mildner B, Knowles BB. Immunohistochemical localization of the epidermal growth factor receptor in normal human tissues. Lab Invest 1986;55:588–92.
Li S, Plowman GD, Buckley SD, Shipley GD. Heparin inhibition of autonomous growth implicates amphiregulin as an autocrine growth factor for normal human mammary epithelial cells. J Cell Physiol 1992;111:103–11. DOI: 10.1002/jcp.1041530114.
Pardo I, Lillemoe HA, Blosser RJ, Choi MR, Sauder CAM, Doxey DK, et al. Next-generation transcriptome sequencing of the premenopausal breast epithelium using specimens from a normal human breast tissue bank. Breast Cancer Res 2014;16:R26–R26. https://doi.org/10.1186/BCR3627
Mukhopadhyay C, Zhao X, Maroni D, Band V, Naramura M. Distinct effects of EGFR ligands on human mammary epithelial cell differentiation. PLoS One 2013;8:e75907. https://doi.org/10.1371/journal.pone.0075907
Pasic L, Eisinger-Mathason TSK, Velayudhan BT, Moskaluk CA, Brenin DR, Macara IG, et al. Sustained activation of the HER1-ERK1/2-RSK signaling pathway controls myoepithelial cell fate in human mammary tissue. Genes Dev 2011;25:1641–53. https://doi.org/10.1101/gad.2025611
Dvorak B. Milk epidermal growth factor and Gut Protection. J Pediatr 2010;156:31–5. https://doi.org/10.1016/j.jpeds.2009.11.018
Cardy RH. Sexual dimorphism of the normal rat mammary gland. Vet Pathol 1991;28:139–45. https://doi.org/10.1177/030098589102800206
Pompei LM, Carvalho FM, Ortiz SCBC, Motta MC, Cruz RJ, Melo NR. Morphometric evaluation of effects of two sex steroids on mammary gland of female rats. Maturitas 2005;51:370–9. https://doi.org/10.1016/J.MATURITAS.2004.09.007
Jenkins EC, Debnath S, Gundry S, Gundry S, Uyar U, Fata JE. Intracellular pH regulation by Na+/H + exchanger-1 (NHE1) is required for growth factor-induced mammary branching morphogenesis. Dev Biol 2012;365:71–81. https://doi.org/10.1016/j.ydbio.2012.02.010
Camacho Leal M del P, Pincini A, Tornillo G, Fiorito E, Bisaro B, Di Luca E, et al. p130Cas over-expression impairs mammary branching morphogenesis in response to estrogen and EGF. PLoS One 2012;7:e49817. https://doi.org/10.1371/journal.pone.0049817
Imagawa W, Tomooka Y, Hamamoto S, Nandi S. Stimulation of mammary epithelial cell growth in vitro: interaction of epidermal growth factor and mammogenic hormones. Endocrinology 1985;116:1514–24. https://doi.org/10.1210/endo-116-4-1514
Mohanam S, Salomon DS, Kidwell WR. Substratum modulation of epidermal growth factor receptor expression by normal mouse mammary cells. J Dairy Sci 1988;71:1507–14. https://doi.org/10.3168/jds.S0022-0302(88)79714-3
Xie W, Paterson AJ, Chin E, Nabell LM, Kudlow JE. Targeted expression of a dominant negative epidermal growth factor receptor in the mammary gland of transgenic mice inhibits pubertal mammary duct development. Mol Endocrinol 1997;11:1766–81. https://doi.org/10.1210/mend.11.12.0019
Wiesen JF, Young P, Werb Z, Cunha GR, Mandel R, Krajewski S, et al. Signaling through the stromal epidermal growth factor receptor is necessary for mammary ductal development. Development 1999;126:335–44. https://doi.org/10.1242/dev.126.2.335
Aupperlee MD, Leipprandt JR, Bennett JM, Schwartz RC, Haslam SZ. Amphiregulin mediates progesterone-induced mammary ductal development during puberty. Breast Cancer Res 2013;15. https://doi.org/10.1186/bcr3431
Aupperlee MD, Zhao Y, Tan YS, Leipprandt JR, Bennett J, Haslam SZ, et al. Epidermal growth factor receptor (EGFR) signaling is a key mediator of Hormone-Induced leukocyte infiltration in the Pubertal Female Mammary Gland. Endocrinology 2014;155:2301–13. https://doi.org/10.1210/en.2013-1933
Jackson-Fisher AJ, Bellinger G, Ramabhadran R, Morris JK, Lee K-F, Stern DF. ErbB2 is required for ductal morphogenesis of the mammary gland. Proc Natl Acad Sci 2004;101:17138–43. https://doi.org/10.1073/pnas.0407057101
Andrechek ER, White D, Muller WJ. Targeted disruption of ErbB2/Neu in the mammary epithelium results in impaired ductal outgrowth. Oncogene 2005;24:932–7. https://doi.org/10.1038/sj.onc.1208230
Jackson-Fisher AJ, Bellinger G, Breindel JL, Tavassoli FA, Booth CJ, Duong JK, et al. ErbB3 is required for ductal morphogenesis in the mouse mammary gland. Breast Cancer Res 2008;10. https://doi.org/10.1186/bcr2198
Lahlou H, Müller T, Sanguin-Gendreau V, Birchmeier C, Muller WJ. Uncoupling of PI3K from ErbB3 impairs mammary gland development but does not impact on ErbB2-induced mammary tumorigenesis. Cancer Res 2012;72:3080–90. https://doi.org/10.1158/0008-5472.CAN-11-3513
Jones FE, Jerry DJ, Guarino BC, Andrews GC SD. Heregulin induces in vivo proliferation and differentiation of mammary epithelium into secretory lobuloalveoli. Cell Growth Differ 1996;7:1031–8.
Wali VB, Gilmore-Hebert M, Mamillapalli R, Haskins JW, Kurppa KJ, Elenius K, et al. Overexpression of ERBB4 JM-a CYT-1 and CYT-2 isoforms in transgenic mice reveals isoformspecific roles in mammary gland development and carcinogenesis. Breast Cancer Res 2014;16:501. https://doi.org/10.1186/s13058-014-0501-z
Muraoka-Cook RS, Sandahl MA, Strunk KE, Miraglia LC, Husted C, Hunter DM, et al. ErbB4 splice variants Cyt1 and Cyt2 differ by 16 amino acids and exert opposing effects on the mammary epithelium in vivo. Mol Cell Biol 2009;29:4935–48. https://doi.org/10.1128/mcb.01705-08
Hilakivi-Clarke L, Cho E, Raygada M, Kenney N. Alterations in mammary gland development following neonatal exposure to estradiol, transforming growth factor α, and estrogen receptor antagonist ICI 182,780. J Cell Physiol 1997;170:279–89. https://doi.org/10.1002/(SICI)1097-4652(199703)170:3%3C279::AID-JCP9%3E3.0.CO;2-L
Berryhill GE, Lemay DG, Trott JF, Aimo L, Lock AL, Hovey RC. The transcriptome of estrogen-independent mammary growth in female mice reveals that not all mammary glands are created equally. Endocrinology 2017;158:3126–39. https://doi.org/10.1210/en.2017-00395
Sinha YN, Tucker HA. Mammary development and pituitary prolactin level of heifers from birth through puberty and during the estrous cycle. J Dairy Sci 1969;52:507–12. https://doi.org/10.3168/jds.S0022-0302(69)86595-1
Meyer MJ, Capuco A V., Ross DA, Lintault LM, Van Amburgh ME. Developmental and nutritional regulation of the prepubertal heifer mammary gland: I. Parenchyma and fat pad mass and composition. J Dairy Sci 2006;89:4289–97. https://doi.org/10.3168/jds.S0022-0302(06)72475-4
Johnsson ID, Hart IC. Pre-pubertal mammogenesis in the sheep 1. The effects of level of nutrition on growth and mammary development in female lambs. Anim Prod 1985;41:323–32. https://doi.org/10.1017/S0003356100036370
Capuco A V., Ellis S, Wood DL, Akers RM, Garrett W. Postnatal mammary ductal growth: three-dimensional imaging of cell proliferation, effects of estrogen treatment, and expression of steroid receptors in prepubertal calves. Tissue Cell 2002;34:143–54. https://doi.org/10.1016/S0040-8166(02)00024-1
Purup S, Vestergaard M, Sejrsen K. Involvement of growth factors in the regulation of pubertal mammary growth in cattle. Adv Exp Med Biol 2000;480:27–43. https://doi.org/10.1007/0-306-46832-8_4
Forsyth IA, Taylor JA, Moorby CD. DNA synthesis by ovine mammary alveolar epithelial cells: Effects of heparin, epidermal growth factor-related peptides and interaction with stage of pregnancy. J Endocrinol 1998;156:283–90. https://doi.org/10.1677/joe.0.1560283
Tou JS, McGrath MF, Zupec ME, Byatt JC, Violand BN, Kaempfe LA, et al. Chemical synthesis of bovine transforming growth factor-α: synthesis, characterization and biological activity. Biochem Biophys Res Commun 1990;167:484–91. https://doi.org/10.1016/0006-291X(90)92049-6
Collier RJ, Mcgrath MF, Byatt JC, Zurfluh LL. Regulation of bovine mammary growth by peptide hormones: involvement of receptors, growth factors and binding proteins. Livest Prod Sci 1993;35:21–33. https://doi.org/10.1016/0301-6226(93)90179-L
Peri I, Shamay A, McGrath MF, Collier RJ, Gertler A. Comparative mitogenic and galactopoietic effects of IGF-I, IGF-II and DES-3-IGF-I in bovine mammary gland in vitro. Cell Biol Int Rep 1992;16:359–68. https://doi.org/10.1016/S0309-1651(06)80141-4
Waksman M, Shamay A, Gertler A. Bovine pituitary, kidney, uterine and mammary gland extracts contain bovine mammary epithelium growth factors that synergise with IGF-I and fetal calf serum: indication for involvement of GTP-binding proteins. Domest Anim Endocrinol 1991;8:271–80. https://doi.org/10.1016/0739-7240(91)90063-p
Sheffield LG, In Suh Yuh. Influence of epidermal growth factor on growth of bovine mammary tissue in athymic nude mice. Domest Anim Endocrinol 1988;5:141–7. https://doi.org/10.1016/0739-7240(88)90014-8
Tonelli QJ, Sorof S. Epidermal growth factor requirement for development of cultured mammary gland. Nature 1980;285:250–2. https://doi.org/10.1038/285250a0
Fenton SE, Sheffield LG. Prolactin inhibits EGF-induced DNA synthesis in mammary epithelium via early signaling mechanisms: possible involvement of protein kinase C. Exp Cell Res 1997;236:285–93. https://doi.org/10.1006/excr.1997.3727
Taketani Y, Oka T. Epidermal growth factor stimulates cell proliferation and inhibits functional differentiation of mouse mammary epithelial cells in culture. Endocrinology 1983;113:871–7. https://doi.org/10.1210/endo-113-3-871
Vonderhaar BK, Nakhasi HL. Bifunctional activity of epidermal growth factor on α- and κ-Casein gene expression in rodent mammary glands in vitro. Endocrinology 1986;119:1178–84. https://doi.org/10.1210/endo-119-3-1178
Sankaran L, Topper YJ. Is EGF a physiological inhibitor of mouse mammary casein synthesis? Unphysiological responses to pharmacological levels of hormones. Biochem Biophys Res Commun 1987;146:121–5. https://doi.org/10.1016/0006-291X(87)90699-1
Kobayashi K, Oyama S, Kuki C, Tsugami Y, Matsunaga K, Suzuki T, et al. Distinct roles of prolactin, epidermal growth factor, and glucocorticoids in β-casein secretion pathway in lactating mammary epithelial cells. Mol Cell Endocrinol 2017;440:16–24. https://doi.org/10.1016/j.mce.2016.11.006
Tidcombe H, Jackson-fisher A, Mathers K, Stern DF, Gassmann M, Golding JP. Neural and mammary gland defects in ErbB4 knockout mice genetically rescued from embryonic lethality. Proc Natl Acad Sci 2003;100:8281–6.
Mapes J, Li Q, Kannan A, Anandan L, Laws M, Lydon JP, et al. CUZD1 is a critical mediator of the JAK/STAT5 signaling pathway that controls mammary gland development during pregnancy. PLOS Genet 2017;13:e1006654. https://doi.org/10.1371/journal.pgen.1006654
Muraoka-Cook RS, Sandahl M, Hunter D, Miraglia L, Earp HS. Prolactin and ErbB4/HER4 signaling interact via janus kinase 2 to induce mammary epithelial cell gene expression differentiation. Mol Endocrinol 2008;22:2307–21. https://doi.org/10.1210/me.2008-0055
Li L, Cleary S, Mandarano MA, Long W, Birchmeier C, Jones FE. The breast proto-oncogene, HRGα regulates epithelial proliferation and lobuloalveolar development in the mouse mammary gland. Oncogene 2002;21:4900–7. https://doi.org/10.1038/sj.onc.1205634
Swanson EW, Poffenbarger JI. Mammary gland development of dairy heifers during their first gestation. J Dairy Sci 1979;62:702–14. https://doi.org/10.3168/jds.S0022-0302(79)83313-5
Anderson RR, Harness JR, Snead AF, Salah MS. Mammary growth pattern in goats during pregnancy and lactation. J Dairy Sci 1981;64:427–32. https://doi.org/10.3168/jds.S0022-0302(81)82589-1
Anderson RR. Mammary gland growth in sheep. J Anim Sci 1975;41:118–23. https://doi.org/10.2527/jas1975.411118x
Sheffield LG. Hormonal regulation of epidermal growth factor receptor content and signaling in bovine mammary tissue. Endocrinology 1998;139:4568–75. https://doi.org/10.1210/endo.139.11.6318
Sheffield LG, Welsch CW. Cyclic nucleotide concentrations and protein kinase activities of bovine mammary tissue maintained in athymic nude mice: effects of mammogenic and lactogenic hormones. J Dairy Sci 1988;71:75–83. https://doi.org/10.3168/jds.S0022-0302(88)79527-2
Sheffield LG, Eppler CM, Tucker HA, Welsch CW. Influence of recombinant deoxyribonucleic acid-derived bovine growth hormone on α-lactalbumin production by bovine mammary tissue maintained in athymic nude mice. J Dairy Sci 1988;71:68–74. https://doi.org/10.3168/jds.S0022-0302(88)79526-0
Fowler KJ, Walker F, Alexander W, Hibbs ML, Nice EC, Bohmer RM, et al. A mutation in the epidermal growth factor receptor in waved-2 mice has a profound effect on receptor biochemistry that results in impaired lactation. Proc Natl Acad Sci 1995;92:1465–9. https://doi.org/10.1073/pnas.92.5.1465
Miettinen PJ, Berger JE, Meneses J, Phung Y, Pedersen RA, Werb Z, et al. Epithelial immaturity and multiorgan failure in mice lacking epidermal growth factor receptor. Nature 1995;376:337–41. https://doi.org/10.1038/376337A0
Dvorak B, Halpern MD, Holubec H, Williams CS, Mcwilliam DL, Dominguez JA, et al. Epidermal growth factor reduces the development of necrotizing enterocolitis in a neonatal rat model. Am J Physiol - Gastrointest Liver Physiol 2002;282:156–64. https://doi.org/10.1152/ajpgi.00196.2001
Schroeder JA, Lee DC. Transgenic mice reveal roles for TGFα and EGF receptor in mammary gland development and neoplasia. J Mammary Gland Biol Neoplasia 1997;2:119–29. https://doi.org/10.1023/A:1026347629876
Casey TM, Mulvey TM, Patnode TA, Dean A, Zakrzewska E, Plaut K. Mammary epithelial cells treated concurrently with TGF-α and TGF-β exhibit enhanced proliferation and death. Exp Biol Med 2007;232:1027–40. https://doi.org/10.3181/0609-RM-218
Sakai S, Mizuno M, Harigaya T, Yamamoto K, Mori T, Coffey RJ, et al. Cause of failure of lactation in mouse mammary tumor virus/human transforming growth factor α transgenic mice. Proc Soc Exp Biol Med 1994;205:236–42. https://doi.org/10.3181/00379727-205-43702
Booth BW, Jhappan C, Merlino G, Smith GH. TGFβ1 and TGFα contrarily affect alveolar survival and tumorigenesis in mouse mammary epithelium. Int J Cancer 2007;120:493–9. https://doi.org/10.1002/ijc.22310
Kenney NJ, Bowman A, Korach KS, Barrett JC, David S. Effect of exogenous epidermal-like growth factors on mammary gland development and differentiation in the estrogen receptor-alpha knockout (ERKO) mouse. Breast Cancer Res Treat 2003;79:161–73. https://doi.org/10.1023/A:1023938510508
Moore GPM, Silvapulle MJ. Lack of effect of epidermal growth factor treatment in late-pregnant ewes on subsequent lactation. J Dairy Res 1991;58:1–11. https://doi.org/10.1017/S0022029900033458
Pollard JW. Macrophages define the invasive microenvironment in breast cancer. J Leukoc Biol 2008;84:623–30. https://doi.org/10.1189/jlb.1107762
Booth BW, Smith GH, Booth BW, Smith GH. Roles of transforming growth factor- α in mammary development and disease. Growth Factors 2009;25:227–35. https://doi.org/10.1080/08977190701750698
Yallowitz AR, Alexandrova EM, Talos F, Xu S, Marchenko ND, Moll UM. P63 is a prosurvival factor in the adult mammary gland during post-lactational involution, affecting PI-MECs and ErbB2 tumorigenesis. Cell Death Differ 2014;21:645–54. https://doi.org/10.1038/cdd.2013.199
Capuco A V., Akers RM. Mammary involution in dairy animals. J Mammary Gland Biol Neoplasia 1999;4:137–44. https://doi.org/10.1023/A:1018769022990
Sobolewska A, Gajewska M, Zarzy J, Gajkowska B, Motyl T. IGF-I, EGF, and sex steroids regulate autophagy in bovine mammary epithelial cells via the mTOR pathway. Eur J Biol 2009;88:117–30. https://doi.org/10.1016/j.ejcb.2008.09.004
Martin PM, Cotard M, Mialot JP, André F, Raynaud JP. Animal models for hormone-dependent human breast cancer - relationship between steroid receptor profiles in canine and feline mammary tumors and survival rate. Cancer Chemother Pharmacol 1984;12:13–7. https://doi.org/10.1007/BF00255902
Beauvais W, Cardwell JM, Brodbelt DC. The effect of neutering on the risk of mammary tumours in dogs - a systematic review. J Small Anim Pract 2012;53:314–22. https://doi.org/10.1111/j.1748-5827.2011.01220.x
Whyte J, Begin O, Bianchi A, McNally S, Martin F. Key signalling nodes in mammary gland development and cancer mitogen-activated protein kinase signalling in experimental models of breast cancer progression and in mammary gland development. Breast Cancer Res 2009;11:209. https://doi.org/10.1186/bcr2361
Booth BW, Boulanger CA, Anderson LH, Jimenez-Rojo L, Brisken C, Smith GH. Amphiregulin mediates self-renewal in an immortal mammary epithelial cell line with stem cell characteristics. Exp Cell Res 2010;316:422–32. https://doi.org/10.1016/j.yexcr.2009.11.006
Fata JE, Mori H, Ewald AJ, Zhang H, Yao E, Werb Z, et al. The MAPK ERK-1,2 pathway integrates distinct and antagonistic signals from TGFα and FGF7 in morphogenesis of mouse mammary epithelium. Dev Biol 2007;306:193–207. https://doi.org/10.1016/j.ydbio.2007.03.013
Arendt LM, Grafwallner-Huseth TL, Schuler LA. Prolactin-growth factor crosstalk reduces mammary estrogen responsiveness despite elevated ERα expression. Am J Pathol 2009;174:1065–74. https://doi.org/10.2353/ajpath.2009.080719
Arendt LM, Rose-Hellekant TA, Sandgren EP, Schuler LA. Prolactin potentiates transforming growth factor induction of mammary neoplasia in transgenic mice. Am J Pathol 2006;168:1365–74. https://doi.org/10.2353/ajpath.2006.050861
Watson JAW and CJ. Signalling downstream of PI3 kinase in mammary epithelium: a play in 3 akts. Breast Cancer Res 2010;12:202. https://doi.org/10.1186/bcr2558
Maroulakou IG, Oemler W, Naber SP, Klebba I, Kuperwasser C, Tsichlis PN. Distinct roles of the three akt isoforms in lactogenic differentiation and involution. J Cell Physiol 2008;217:468–77. https://doi.org/10.1002/jcp.21518
Watson CJ, Neoh K. The Stat family of transcription factors have diverse roles in mammary gland development. Semin Cell Dev Biol 2008;19:401–6. https://doi.org/10.1016/J.SEMCDB.2008.07.021
Cui Y, Riedlinger G, Miyoshi K, Tang W, Li C, Deng C-X, et al. Inactivation of Stat5 in mouse mammary epithelium during pregnancy reveals distinct functions in cell proliferation, survival, and differentiation. Mol Cell Biol 2004;24:8037–47. https://doi.org/10.1128/mcb.24.18.8037-8047.2004
Li S, Rosen JM. Nuclear factor I and mammary gland factor (STAT5) play a critical role in regulating rat whey acidic protein gene expression in transgenic mice. Mol Cell Biol 1995;15:2063–70. https://doi.org/10.1128/mcb.15.4.2063
Kavarthapu R, Dufau ML. Role of EGF/ERBB1 in the transcriptional regulation of the prolactin receptor independent of estrogen and prolactin in breast cancer cells. Oncotarget 2016;7:65602. https://doi.org/10.18632/ONCOTARGET.11579
Chapman RS, Lourenco PC, Tonner E, Flint DJ, Selbert S, Takeda K, et al. Suppression of epithelial apoptosis and delayed mammary gland involution in mice with a conditional knockout of Stat3. Genes Dev 1999;13:2604–16. https://doi.org/10.1101/gad.13.19.2604
Proietti CJ, Rosemblit C, Beguelin W, Rivas MA, Celeste M, Flaqué D, et al. Activation of Stat3 by heregulin/ErbB-2 through the co-option of progesterone receptor signaling drives breast cancer growth. Mol Cell Biol 2009;29:1249–65. https://doi.org/10.1128/MCB.00853-08
Philp JAC, Burdon TG, Watson CJ. Differential activation of STATs 3 and 5 during mammary gland development. FEBS Lett 1996;396:77–80. https://doi.org/10.1016/0014-5793(96)01069-1
Khaled WT, Read EKC, Nicholson SE, Baxter FO, Brennan AJ, Came PJ, et al. The IL-4/IL-13/stat6 signalling pathway promotes luminal mammary epithelial cell development. Development 2007;134:2739–50. https://doi.org/10.1242/dev.003194
Zhao G, Liu L, Peek RM, Hao X, Polk DB, Li H, et al. Activation of epidermal growth factor receptor in macrophages mediates feedback inhibition of M2 polarization and gastrointestinal tumor cell growth. J Biol Chem 2016;291:20462. https://doi.org/10.1074/JBC.M116.750182
Cline JM. Assessing the mammary gland of nonhuman primates: effects of endogenous hormones and exogenous hormonal agents and growth factors. Birth Defects Res Part B Dev Reprod Toxicol 2007;80:126–46. https://doi.org/10.1002/BDRB.20112
Schams D, Russe I, Schallenberger E. The role of steroid hormones, prolactin and placental lactogen on mammary gland development in ewes and heifers. J Endocrinol 1984;102:121–30. https://doi.org/10.1677/joe.0.1020121
Graham JD, Mote PA, Salagame U, Van Dijk JH, Balleine RL, Huschtscha LI, et al. DNA replication licensing and progenitor numbers are increased by progesterone in normal human breast. Endocrinology 2009;150:3318–26. https://doi.org/10.1210/EN.2008-1630
Mcandrew J, Rudland PS, Platt-Higgins AM, Smith JA. Immunolocalization of alpha-transforming growth factor in the developing rat mammary gland in vivo, rat mammary cells in vitro and in human breast diseases. Histochem J 1994;26:355–66. https://doi.org/10.1007/BF00157769
Yang J, Guzman R, Richards J, Imagawa W, McCormick K, Nandi S. Growth factor- and cyclic nucleotide-induced proliferation of normal and malignant mammary epithelial cells in primary culture. Endocrinology 1980;107:35–41. https://doi.org/10.1210/endo-107-1-35
Vonderhaar BK. Local effects of EGF, α-TGF, and EGF-like growth factors on lobuloalveolar development of the mouse mammary gland in vivo. J Cell Physiol 1987;132:581–4. https://doi.org/10.1002/jcp.1041320324
Smith JA, Barraclough R, Fernig DG, Rudland PS. Identification of alpha transforming growth factor as a possible local trophic agent for the mammary gland. J Cell Physiol 1989;141:362–70. https://doi.org/10.1002/jcp.1041410218
Funding
This work was supported in part by funding from the National Institute of Food and Agriculture, Award 2020-67015-31176, by national grants from the Italian Ministry of Instruction, University and Research, and by the University of Turin, grant ACCP_AUTOF_21_01.
Author information
Authors and Affiliations
Contributions
Alessia Morato – conception, review of the literature, draft preparation, editing. Paolo Accornero – figure preparation, editing. Russell Hovey – conception, editing.
Corresponding author
Ethics declarations
Conflicts of Interest
Alessia Morato – No conflicts. Paolo Accornero – No conflicts. Russell Hovey is an Editorial Board Member for the Journal of Mammary Gland Biology and Neoplasia.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Morato, A., Accornero, P. & Hovey, R.C. ERBB Receptors and Their Ligands in the Developing Mammary Glands of Different Species: Fifteen Characters in Search of an Author. J Mammary Gland Biol Neoplasia 28, 10 (2023). https://doi.org/10.1007/s10911-023-09538-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10911-023-09538-w