Abstract
Macrophage influx is associated with negative outcomes for women with breast cancer and has been demonstrated to be required for metastasis of mammary tumors in mouse models. Pregnancy-associated breast cancer is characterized by particularly poor outcomes, however the reasons remain obscure. Recently, post-pregnancy mammary involution has been characterized as having a wound healing signature. We have proposed the involution-hypothesis, which states that the wound healing microenvironment of the involuting gland is tumor promotional. Macrophage influx is one of the prominent features of the involuting gland, identifying the macrophage a potential instigator of tumor progression and a novel target for breast cancer treatment and prevention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Components of chronic inflammation are common in the microenvironment of many cancers and further, inflammation is associated with initiation and promotion of specific cancers, such as colorectal, gastric, liver and breast [1]. In cancer-related inflammation, immune cell infiltration is associated with rampant cytokine/chemokine signaling, protease-mediated tissue remodeling and angiogenesis, constituents known to accelerate tumor progression [2, 3]. While the cellular milieu of cancer-related inflammation is complex, in breast cancer the presence of macrophages specifically predicts poor prognosis [4]. Importantly, data from several studies suggest that macrophages and associated wound healing programs may be integral to weaning-induced mammary gland involution [5–10]. Thus, physiologic gland regression after pregnancy is implicated in tumor promotion [11]. Consistent with this hypothesis, a subset of breast cancer defined by patients diagnosed within 5 years of a recent pregnancy is associated with poor prognosis [11]. This subset is referred to as pregnancy-associated breast cancer or PABC. We propose the involution-hypothesis to account for the highly metastatic nature of PABC. The involution-hypothesis predicts that the wound healing attributes of mammary involution contribute to a tumor promotional microenvironment, characterized by increases in protease activity, release of bioactive fragments of extracellular matrix, and accumulation of fibrillar and proteolyzed collagen [11]. These involution-associated changes in the mammary microenvironment are consistent with macrophage function and implicate macrophage involvement in promotion of PABC metastasis. In support of the involution-hypothesis, numerous attributes of the actively involuting mammary microenvironment have been demonstrated in preclinical models to induce metastatic phenotypes in tumor cells [5–7, 12]. The focus of this review is to explore the possible roles for involution macrophages in promoting breast cancer.
Evidence for Macrophage Involvement in Breast Cancer
Macrophages have been positively correlated with poor prognosis of breast cancer in multiple studies. The subset of macrophages found in close proximity to tumors have been referred to as tumor-associated or TAM. Importantly, TAM can contribute significantly to the cellular bulk of the tumor, implicating these cells in dictating tumor biology. In some cases, TAM have been reported to account for as much as 50% of the tumor mass [13]. Tumor macrophage infiltration is linked to significant decreased relapse-free survival (Hazard Ratio = 2.79) and overall survival (Hazard Ratio = 9.43) [14]. For comparison, hazard ratios for tumor size, a known negative prognostic indicator, were 2.48 for relapse-free survival and 1.09 for overall survival in the same cohort [14]. Increased TAM density is also associated with early establishment of breast cancer metastases [14, 15]. Consistent with these observations, in a meta-analysis of 15 studies that correlate TAM with cancer prognosis, 80% correlated high levels of TAM with negative outcomes for cancer patients, with all four of the included breast cancer studies demonstrating this relationship [4]. Given that distinct cytokine milieus can elicit macrophages with either tumor suppressive or tumor promotional activities; this may explain why tumor macrophage number does not always correlate with negative outcome. The concept that macrophages can be either tumor suppressive or promotional will be explored later in this review.
Specific proteins involved in macrophage growth and recruitment have also been implicated as predictors of poor prognosis in breast cancer. Colony stimulating factor 1 (CSF-1) is a growth factor that stimulates macrophage proliferation and maturation and is also chemotactic for macrophages [16, 17]. Overexpression of CSF-1 at the RNA and protein levels has been observed at sites of primary breast cancer [18]. In addition, mean plasma CSF-1 levels were 8% to 24% higher in breast cancer patients with locally advanced (388.3 pg/ml) or metastatic disease (446.1 pg/ml) as compared to those with in situ carcinoma (358.6 pg/ml), implicating macrophages in the transition from non-invasive to invasive disease [19]. High circulating CSF-1 levels also correlate strongly with rapid progression of metastatic disease and CSF-1 continues to be expressed higher at locations of metastatic recurrence [20]. In five breast cancer expression data sets, a CSF-1 response signature was found to correlate with other predictors of poor prognosis including estrogen and progesterone receptor negative status, higher tumor grade and larger size [21]. Monocyte chemotactic protein 1 (MCP-1), another protein known to attract macrophages, correlates both with macrophage accumulation in breast tumors and early relapse in patients [22]. These studies indicate that macrophages stimulate tumor cells directly, and/or tumor cells are responsive to these same stimuli as macrophages.
A survey of the available human breast cancer data reveals direct paracrine signaling between macrophages and tumor cells. Macrophages isolated from human breast tumors have been shown to release epithelial growth factor (EGF), and tumors that express high EGF receptor (EGFR) protein levels have increased macrophage infiltration [23, 24]. High tumor expression of EGFR is an independent predictor of negative prognosis in women, suggesting the importance of this paracrine signaling pathway in breast cancer [25]. A prediction of this model would be increased tumor cell proliferation with macrophage infiltrate. Indeed, macrophage infiltration does correlate with tumor cell proliferation in breast cancer, as assessed by Ki-67 levels [15]. This EGF/EGFR paracrine signaling is complimented with tumor cell production of CSF-1, which directly stimulates macrophages via the macrophage receptor CSF-1R [26]. The result is an apparent complete paracrine signaling loop between macrophages and tumor cells. Suitably, overexpression of CSF-1 in tumor cells independently indicates poor outcomes in breast cancer [27]. Further, CSF-1R has been reported to be expressed in 58% of all and 85% of invasive breast cancers, where both stromal macrophages and neoplastic epithelial cells stain positive [28]. An antibody to activated CSF-1R shows that 52% of CSF-1R positive breast carcinomas expressed the activated form of the receptor [29]. In addition, in preclinical models, it has been shown that tumor cells can commandeer the production of EGF leading to autocrine stimulation of EGFR [30]. Thus, it appears that not only is there evidence for direct macrophage-cancer cell paracrine signaling interactions in human breast cancer, but for autocrine CSF-1/CSF-1R and EGF/EGFR signaling in breast tumor cells as well. Altogether, macrophage infiltration, macrophage growth and chemotactic factors, and macrophage signaling pathways are all correlated with negative outcome for breast cancer patients.
Mouse Models: Interaction of Macrophages and Breast Cancer
Although clinical studies strongly implicate a relationship between macrophages and breast cancer progression, this interaction has been thoroughly established by J. Pollard and colleagues using mouse models. The mouse model of breast cancer in these studies is induced by polyoma middle T oncoprotein driven by the mammary specific MMTV promoter (MMTV-PyMT). In this model, the depletion of macrophages though a homozygous null germline mutation for CSF-1 resulted in decreased rates of tumor progression and an almost complete reduction in tumor metastasis [31]. When CSF-1 was transgenically re-expressed in the mammary epithelium of the CSF-1 null/MMTV-PyMT mice, both tumor growth and metastasis were restored [31]. Consistent with these observations, when CSF-1 was overexpressed in MMTV-PyMT mice tumor progression and metastasis were significantly accelerated [31]. To determine whether human breast tumor cells were similarly responsive to macrophages, human tumor cells were injected into mouse mammary glands and CSF-1/CSF-1R signaling blocked using antisense oligonucleotides, siRNAs and antibody against CSF-1. All three techniques for CSF-1 ablation lead to reduced macrophage recruitment to the tumor microenvironment, and a decrease in tumor growth and metastasis [32, 33]. Finally, micro-needle manipulation in conjunction with intravital imaging of fluorescently labeled cells in these models have provided additional evidence for a paracrine EGF/CSF-1 loop between macrophages and mammary tumor cells [26, 34]. Cumulatively, these preclinical studies in multiple models of breast cancer highlight a promotional role for macrophage growth factor CSF-1, and provide a plausible explanation for the clinical correlation between breast cancer prognosis and tumor associated macrophages.
Macrophages in the Pubertal and Pregnancy Stages of Mammary Gland Development
Due to the accumulating evidence that macrophages promote breast cancer, it is natural to evaluate the involvement of macrophages in the mammary gland in the absence of cancer. Specifically, it is of interest to know whether macrophage number is regulated during key developmental windows. During post-natal development of the gland, macrophages are recruited to growing terminal end buds (TEBs) [35]. To address their function during TEB outgrowth, leukocytes were depleted by sub-lethal γ irradiation, or macrophages were selectively eliminated using the CSF-1 homozygous null mice. These studies show that leukocytes, and specifically macrophages, are necessary for proper development and outgrowth of TEBs into the mammary fat pad and for subsequent TEB bifurcation [35]. It is speculated that that these macrophages contribute to ductal invasion through release of factors that promote growth, angiogenesis and extracellular matrix (ECM) breakdown, however, the mechanism remains undefined [35, 36]. Macrophages are also present during pregnancy, another period of epithelial expansion in the mammary gland [37]. Again, as in puberty, CSF-1 knockout results in reduced ductal growth and decreased branching in the mammary glands of pregnant mice [38]. However, these mice also have precocious lobulo-alveolar development, implicating macrophages as inhibitory to alveolar expansion in mice. Clearly these studies demonstrate that macrophage function contributes to mammary morphogenesis during key windows of differentiation, but much remains to be determined regarding their specific functions and mechanisms of action.
The Involuting Mammary Gland
Our understanding of macrophage function during weaning-induced mammary involution is poorly developed. With cessation of milk secretion, the mammary gland resorbs the elaborate milk-producing lobulo-alveolar structures of pregnancy and returns to a simpler ductal network poised to respond to another round of pregnancy hormones [39, 40]. The magnitude and speed of this tissue deconstruction is dramatic and considered unique to the mammary gland, as this physiologic tissue remodeling exceeds that which occurs under many pathological conditions. In rodents, where mammary involution has been extensively characterized, a full 50–80% of the secretory epithelium is eliminated by apoptosis and clearance within one week of weaning [41]. By 10 days post weaning, the gland is largely devoid of alveolar structures, and is dominated by a ductal epithelium embedded in an adipocyte rich stroma.
The process of mammary gland involution is an intrinsically regulated developmental program consisting of several ordered events. Early on it was recognized that involution could be separated into reversible and irreversible phases based on the ability of dams to resume nursing after pup removal [42, 43]. The reversible phase was characterized as proteinase-independent and corresponds with the early wave of secretory epithelial cell death prior to histological evidence of alveolar collapse [44]. The irreversible phase or proteinase-dependent phase, correlates with histological evidence of alveolar destruction, and was determined to involve the matrix metalloproteinases gelatinase A (MMP-2), stromelysin 1 (MMP-3) and the serine protease urokinase-type plasminogen activator (uPA) [44]. In this study, macrophages detected by Mac-2 immunohistochemical (IHC) stain were found to be rare during the protease-independent phase, but present at high levels during the protease-dependent phase [44]. By in situ hybridization, the macrophages did not appear to be major producers of MMP-2,−9 or uPA, and thus it was speculated that the macrophages were not involved in induction of mammary epithelial cell apoptosis, but rather in scavenging apoptotic debris [44]. Cluster analyses of microarray studies have provided evidence that involution is more complex than the two stage model, with gene expression patterns consistent with a multi-step process [45]. Some additional events associated with involution, of which potential roles of macrophages are currently unknown include adipocyte repopulation [46] and the transient ECM changes that occur during late involution and which may be associated with ductal stabilization [6].
Given that macrophages are professional phagocytes, it was anticipated that their role in involution would include the clearance of the dying alveolar cells. However, macrophages and other immune cells were found to be at very low levels during the peak window of apoptotic cell and milk clearance in C57BL/6 mice [47, 48]. Instead, as shown by elegant studies performed in the labs of Fadok and Henson, mammary epithelial cells were discovered to become amateur phagocytes responsible for cell and milk clearance, leaving the role of the involution-associated macrophages undefined (see Monks and Henson review, this issue) [47, 48]. The observation that mammary epithelial cells appear to fulfill the role of phagocytes during involution helped solidify the view that developmentally regulated mammary involution is not immune cell mediated.
Evidence of Immune Cell Involvement in Involution
Based on the considerable evidence for involution being a non-inflammatory process, it was surprising when gene expression data obtained from mouse studies identified numerous immune-related genes upregulated in the involuting mammary gland [9, 10]. In these studies, acute phase response genes, as well as gene profiles associated with innate and adaptive cellular immunity, increased with involution [9, 10]. An early gene set, upregulated within 12 h post-weaning, included genes for the inflammatory mediators interleukin-1α, interleukin-1β, and interleukin-13, which are associated with macrophage, T cell and B cell activation [9, 10]. Consistent with these gene expression profiles, the presence of plasma cells was reported to increase over 20 fold by involution day 4 [10]. Bacteria would be anticipated to trigger such a robust humoral microbial response, however there was no evidence for bacteria by Gram staining, suggesting a possible role for sterile inflammation in physiologic mammary involution.
In addition to implications for T and B cell involvement, innate immune cell genes were upregulated during involution [10]. Neutrophil chemoattractant gene growth-related oncogene 1 (GRO-1) expression was increased ~5 fold within the first 24 h post-weaning, with concurrent increased expression of neutrophilic granulocyte marker leucine-rich α 2 -glycoprotein (LRG) at 24–48 h, prior to macrophage influx at 72 h [44]. Histochemical analysis confirmed an increase in number of neutrophils as early as 24 h post weaning, with numbers steadily increasing through day 4 [10].
Many genes involved in macrophage recruitment and activation are upregulated at the RNA and protein levels during involution. A wave of gene expression that increases early at 24 h post-weaning consisted of several chemoattractants for monocytes and macrophages including CCL6, CCL7, CCL8, and CXCL14 [9, 10]. Several monocyte attracting cytokines, including CCL6 and macrophage inflammatory protein-1α (MIP-1α) have been shown to be secreted by neutrophils, so it is noteworthy that neutrophils have been reported to populate the involuting gland prior to macrophages [49, 50]. Next, the monocyte/macrophage specific antigens CSF-1R, CD68, low density lipoprotein-related protein 1 (LRP-1) and CD14 were found at high levels at 72 and 96 h post-weaning, consistent with macrophage influx [9, 10]. These results corroborated previous RNA expression data for macrophage markers F4/80 and Mac-2 [51]. IHC analyses for several of these macrophage associated proteins have validated the RNA expression data and demonstrate presence of macrophages in the late involuting mouse mammary gland (Table 1) [10, 44, 47, 52–54]. These observations have been extended to the rat model [8], and further, preliminary data from T. Lyons and Schedin demonstrate infiltration of CD45+ leukocytes into the involuting lobules of the human breast as well (Fig. 1). While the exact roles of the macrophages during involution are currently unknown, cumulatively the data support the hypothesis that mammary gland involution utilizes macrophages in a remodeling process that is distinct from pathologic tissue remodeling such as occurs with microbial stimuli or wound healing.
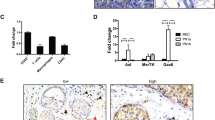
CD45 positive cell number increases in actively involuting lobules compared to adjacent lactating lobules in human breast. Immunohistochemical stain for CD45 (common leukocyte antigen) in breast tissue at time of weaning. CD45+ cells, detected by brown stain, are present at higher levels in actively involuting lobule identified by collapsed alveoli compared to actively lactating lobule identified by extended milk-filled alveoli. Scale bar represents 100 µm.
A Macrophage is not a Macrophage is not a Macrophage…
Monocytes are a dynamic group of cells that can mature across a spectrum of phenotypes depending on what signals are found in their environment (Table 2). Further, this maturation is thought to be reversible, permitting the tissue macrophage to respond appropriately to new stimuli. Classically, monocytes respond to stimuli involved in eliciting an immune response to intracellular pathogens including bacterial wall protein lipopolysaccharide (LPS), and the cytokines interferon-γ (IFN-γ), interleukin-1β (IFN-β), and tumor necrosis factor-α (TNF-α) [55]. These are the same signals involved in inducing a Th1-response in T cells and thus monocytes stimulated by these activators have been referred to as M1 macrophages. This Th1/M1 cellular immune reaction is characterized by activated cytotoxic T-lymphocytes and macrophages that target infected tissues. Classically activated, or M1-type, macrophages, typically release high levels of interleukin-12 (IL-12), interleukin-23 (IL-23), interleukin-1 (IL-1) and interlekin-6 (IL-6), cytokines known to enforce the Th1 response [55, 56]. Activities associated with an M1 macrophage include antigen presentation, killing intracellular pathogens, and promotion of cytotoxicity [55]. Importantly, these M1 activities set up an anti-tumoral environment [57, 58].
The nomenclature for macrophage polarization described above, has been proposed by Mantovani and colleagues to define classes of macrophages other than the M1 type. Their focus has been on macrophages with phenotypes distinct from classical M1-type, which are referred to as M2-type or alternatively activated. This alternative activation pathway was originally described as macrophages stimulated by pathogens presented by extracellular pathways, including parasitic and allergic responses. The broad M2 category originally included macrophages activated by interleukin-4 (IL-4), interleukin-13 (IL-13), and interleukin-10 (IL-10). Gordon et al. 2003 voiced a preference that only the IL-4/IL-13 stimulated macrophages be referred to as alternatively activated. These IL-4/IL-13 stimulated macrophages, further categorized as M2a by Mantovani et al. 2004, fit in the group of immune cells that respond to Th2 cytokines. IL-4 and IL-13, in a Th2-response, elevate humoral immunity through increased proliferation and activation of B cells into plasma cells that secrete high levels of antibody. Alternatively activated macrophages are involved in the killing and encapsulation of parasites, in allergic reactions, and in tissue repair associated with wound healing [55, 56, 59].
Two additional categories of macrophages have now been delineated, both involved in immunoregulation. M2b macrophages are stimulated by a combination of immune complexes and toll-like receptors (TLR) [55]. These macrophages promote Th2 activation yet secrete a combination of M2 and M1 cytokines including IL-10, IL-1, IL-6 and TNF-α [55]. In contrast, M2c macrophages are activated by and stimulated to produce IL-10 and tumor growth factor-β (TGF-β), both immunosuppressive cytokines [55, 59]. A primary action of M2c macrophages is inhibition of the Th1/M1 response program [55, 60]. M2c macrophages also down-regulate MHC Class II molecules used for antigen presentation, and are therefore additionally distinct from the alternatively activated M2a and M2b categories, which can present antigen [55, 59]. Further, M2c macrophages can contribute to matrix deposition and tissue remodeling, which is likely mediated through their upregulation of TGF-β [55, 61]. Recent reviews of the M1/M2 continuum show that M2 macrophages share similar cytokine profiles and activities with tumor associated macrophages (TAM), suggesting that M2 macrophages are tumor promotional [57, 58]. TAM and M2 macrophages both secrete IL-10, TGF-β, MMPs, and growth factors that collectively can lead to direct immunosuppression via IL-10, matrix remodeling, angiogenesis, and even tumor growth, invasion, and metastasis.
Other researchers have recently modeled macrophage sub-types into a non-linear spectrum [56]. The functional attributes of the three categories of macrophages they define; host defense, wound healing and immune regulation, do not directly align with the classification described above. Given macrophage plasticity, these classification schemas are designed to organize the widely diverse set of functional phenotypes that macrophages display as best as possible using current knowledge. Therefore, differences between classification schemes are to be expected in this rapidly progressing field. Even the proteins commonly used to distinguish all monocytes and macrophages from other leukocyte lineages need to be evaluated with caution. New flow cytometry data show that ‘pan’ macrophage markers CSF-1R and F4/80 identify two distinct, minimally overlapping macrophage populations (M. Pillai, unpublished data). Work by others have shown distinct populations of macrophages identified by non-overlapping expression of CD68, CD11b, and F4/80 [62–65]. In addition, we have observed in the rat mammary gland that CSF1-R and CD68 recognize different macrophage populations (O’Brien & Schedin, unpublished data). Whether these provocative data suggest that these pan-macrophage markers coincide with the M1/M2 sub-categories is an interesting, but unresolved question. Overall, these macrophage classification schemas can be well used as intellectual constructs with which to test specific hypotheses.
Are Involution Macrophages a Specific Subtype?
While numerous studies have now confirmed the influx of macrophages during mammary involution, the question of whether these macrophages are ‘polarized’ into distinct sub-types has not been addressed. Given the relationship between macrophage polarization and tumor cell surveillance, it is important to determine functional attributes of involution macrophages. Our lab has started to investigate the functional phenotype of involution macrophages by assessing for traditional M1/M2 markers [60]. Using iNOS (inducible nitric oxide synthase) as an M1 marker and Arginase-1 as an M2 marker, our IHC analyses show that while M1 macrophage levels stay consistently low across the pregnancy/lactation/involution cycle, M2-macrophage number increases 4–6 fold above nulliparous levels during mammary gland involution in both mouse and rat models (Fig. 2 and O’Brien & Schedin, unpublished data). Comprehensively, these data indicate that macrophages are not only present during the physiologically normal period of mammary involution, but have an M2-like phenotype that could exhibit pro-cancer attributes. What follows is a discussion of specific attributes of mammary gland involution that macrophages may facilitate, which are anticipated to promote cancer.
Use of M1/M2-specific macrophage markers by IHC identifies involution macrophages as M2-like. Immunohistochemical stain and quantification for iNOS (M1) or Arginase-1 (M2) in rat mammary tissue at Involution Day 6. Arginase-1 positive (M2) macrophage number is high during involution whereas iNOS positive (M1) macrophage number remains low in rat mammary tissue, *p< 0.01. Scale bars represent 50 µm.
Production and Release of Proteolytic Enzymes
Dramatic tissue remodeling occurs during mammary gland involution, with breakdown of alveolar structures and their surrounding extracellular matrix (ECM) [66, 67]. Stromal matrix metalloproteinases (MMP) levels and activity increase during involution, including MMP-2, −3 and −9 in the rat and 130K and 60K gelatinases in the mouse [6, 66, 68]. These observations are consistent with known functions of macrophages, where upregulation of several proteolytic enzymes including collagenases and serine proteases occur in response to endotoxin, thioglycollate, and CCL5 stimulation [69–75]. ECM proteins proteolyzed during involution include fibronectin, laminin, entactin/nidogen and collagen [6, 76] (O’Brien & Schedin, unpublished data). Laminin peptides are chemotactic to macrophages both in vitro and in vivo, and can promote expression of uPA and MMP-9, whereas fibronectin fragments trigger monocyte/macrophage secretion of MMP-2, −9 and −12 [77–81]. Thus, a positive feedback loop may exist between resident macrophages stimulated to secrete matrix proteinases resulting in ECM fragments that subsequently recruit and stimulate additional macrophages. Given the putative roles of MMPs in breast cancer progression, the secretion of proteases by macrophages during the involution period is likely tumor promotional [82]. Consistent with this role, macrophages co-cultured with breast tumor cells increase expression and activity of MMP-2, −3, −7 and −9 [83]. Further, macrophages located at the invasive front of breast tumors show positive IHC stain for Type IV collagenases and cathepsin B [84, 85]. Since many ECM fragments promote tumor cell motility and invasion in vitro, the production of ECM fragments via macrophage protease activity during involution is consistent with a similar role in breast cancer progression [5, 86].
Breakdown of Basement Membrane
The myoepithelial cell and basement membrane barrier that surrounds the mammary epithelium throughout pregnancy and lactation has been reported to be compromised during mammary gland involution, an event in which macrophages could be involved and of which tumor cells could take advantage. Electron microscopy (EM) of involuting rat mammary glands has shown that myoepithelial cells are not always in a continuous layer but can interdigitate with nearby epithelial cells [87]. During involution in the rat and human, EM analysis reveals the basement membrane as convoluted with variable thickness, while IHC analyses of basement membrane proteins laminin and type IV collagen show both loose structure and discontinuous areas [67, 87, 88]. Further, a diffuse stain of these proteins is observed throughout the tissue suggesting basement membrane degradation [67]. These data provide indirect evidence for interruption of this functional barrier during mammary gland involution.
Breakdown of the myoepithelial and basement membrane layers is the hallmark of local invasion from ductal carcinoma in situ (DCIS) to invasive breast carcinoma. Macrophages are implicated in promoting this proteolysis due to both their location at points of basement membrane breakdown early in tumorigenesis and their release of proteases that digest basement membrane proteins [69–75, 89]. Further, macrophages are enriched at the invasive fronts of mouse tumors, implicating a role in tumor cell invasion [90]. We propose that further investigation may reveal a role for macrophages in basement membrane breakdown during involution, and that in the presence of DCIS during involution, macrophages could foster the activation and dissemination of previously quiescent tumor cells.
Cell Movement along Collagen Fibers
The increase in fibrillar collagen with involution could serve as a means of transportation for the dissemination of macrophages and associated tumor cells. Collagen content, as assessed by picro-sirius red stain, increases in the rat mammary gland during involution compared to nulliparous controls [7]. Collagen fibers can be imaged due to the resonant emission of polarized light from triple α-helical structures called second harmonic generation (SHG) using multi-photon microscopy [91, 92]. With SHG visualization and intravital imaging techniques, eGFP expressing macrophages have been observed to co-localize and move along collagen fibers [93]. In the MMTV-PyMT model, macrophages are associated with the dense collagen fibers found at the mammary/tumor margin [94]. Intravital imaging revealed that ~90 % of motile tumor cells associate with macrophages whereas only ~10% of the tumor cells were found to be motile in the absence of macrophages. Importantly, these macrophages were often perivascular, suggesting a mechanisms for tumor cell intravasation [94]. With increased levels of collagen and macrophages in the involuting microenvironment, the stage is set for macrophage-promoted tumor cell invasion.
Angiogenesis
Another route through which involution macrophages could contribute to tumor progression is by promoting angiogenesis, the formation of new capillary networks from pre-existing blood vessels. Both wound healing and tumor associated macrophages have been implicated in angiogenesis [95]. Wound-derived macrophages have been shown in vivo to stimulate neovascularization in corneal and rabbit ear chamber angiogenesis assays [96–98]. The production of several pro-angiogenic factors by wound macrophages has also been demonstrated, including IL-1, TGF-α, TGF-β, insulin-like growth factor (IGF-1), platelet-derived growth factor (PDGF) and vascular endothelial growth factor (VEGF) [99, 100]. Wound-derived macrophages can be involved in many of the steps of angiogenesis including induction of endothelial cell chemotaxis, proliferation and matrix synthesis [101]. TAM share many of the pro-angiogenic abilities of wound-derived macrophages. The pro-angiogenic cytokines VEGF, TGF-α, and PDGF are also released by TAM when in hypoxic environments, as well as IL-8, basic FGF (bFGF), and prostaglandin E2 (PGE2) [102]. Several mouse models have demonstrated a distinct role for macrophages in the ‘angiogenic switch’ required for malignant progression. In a human xenograft model of breast cancer, the depletion of CSF-1 by anti-sense oligonucleotides, siRNAs or antibodies resulted in reduced angiogenesis as well as decreased tumor progression [32, 33]. Direct evidence for macrophage-induced tumor angiogenesis comes from a model where Tie2-expressing macrophages are recruited to tumors [103]. Ablation of these macrophages reduces both tumor angiogenesis and tumor growth [103]. Consistent with this study, F4/80 positive macrophage infiltration occurs just before increased tumor vessel density in the MMTV-PyMT model [104]. When the macrophages were depleted by genetic cross into the CSF-1 null background, the angiogenic switch was significantly delayed, and a 50% decrease in vascular density occurred [104]. In another study, tet-inducible MMTV-VEGF-A mice were crossed with the PyMT/CSF-1 null mice to determine whether the loss of macrophage angiogenic function could be restored by VEGF-A alone. The angiogenic switch was restored as well as tumor progression [105]. As the primary cellular source for pro-angiogenic VEGF-A in the PyMT model is TAM, this study highlights the promotional role macrophages have in tumor angiogenesis [104]. Clinical breast cancer data also support a relationship between macrophages and angiogenesis, as increased TAM number correlates with high vascular grades of breast tumors and with poor prognosis in multiple studies [4, 14, 99].
Under non-cancer conditions, involution macrophages are not likely to be pro-angiogenic, but could be promoted to this state by the presence of tumor cells. While angiogenesis is highly upregulated during the pregnant and lactational periods of mammary gland development, during involution the intricate capillary networks required for lactation regress through currently unknown mechanisms [106]. While it is difficult to detect apoptotic endothelial cells during involution, the vessel organization returns to the simple, pre-pregnant network within 10 days post-weaning [107]. Concurrently, there is a progressive decrease in VEGF and VEGF-receptor RNA levels [108]. However, based on the known plastic response of macrophages to various environmental cues, we propose that involution macrophages are poised to respond to the presence of cancer cells by switching to an angiogenic phenotype.
Targeting Macrophages for Prevention of PABC-associated Metastasis?
Cumulatively, the data implicating macrophages in breast cancer progression are highly compelling and identify involution macrophages as a novel target for breast cancer treatment and prevention. One potential direction would be to inhibit or eliminate macrophage function during involution. In order to pursue this approach it would be essential that involution macrophages be dispensable to gland regression following pregnancy. Thus key unanswered questions in this pursuit include determining whether the intrinsic program of epithelial cell death and the macrophage-associated tissue remodeling program during involution are causally linked, whether these processes can be separated and whether involution can proceed in the absence of macrophages. As previously noted, the function of the macrophage during gland involution is undefined. However, there are many roles by which macrophages may facilitate the involution process. As already discussed, one putative role is in apoptotic cell clearance. While early apoptotic cell clearance appears to be delegated to the phagocytic mammary epithelial cells, the question of whether macrophages contribute to clearance at later stages is unresolved. Since a primary function of macrophages is microbial clearance, it may be that they participate similarly in the protection of the involuting gland, as involution has been characterized by increased risk for mammary infection and mastitis [109, 110]. Supportive of this role, genes involved in the acute phase response, are upregulated during involution [9, 10]. One key control gene significantly upregulated during involution is signal transducer and activator of transcription 3 (STAT3), which has been shown to be essential for expression of several acute phase response genes including serum amyloid P, fibrinogen-α and -γ [111] (see Watson review, this issue). In the background of a mammary epithelial cell specific conditional STAT3 deletion, mammary involution becomes susceptible to mastitis [112]. Alternatively or in addition, the presence of macrophages in involution could contribute to protection from autoimmune activation. Impaired clearance of apoptotic cells can result in release of auto-antigens and the production of auto-antibodies [113]. Therefore, during involution, which is a physiologic period defined by very high levels of apoptotic activity, it may be advantageous for the immune system to be prepared for potential misdirection and auto-antibody formation. Obviously, the contribution of macrophages to gland involution requires further examination before targeting involution macrophages for prevention or treatment of breast cancer can be explored. Another potential direction could be modifying the phenotype of involution macrophages to reduce their tumor promoting capabilities. Theoretically, it would be possible to redirect macrophage polarization to full M1/tumor suppressive phenotype. Again, the consequences to normal mammary gland involution and potential adverse effects of such treatment would need to be fully explored.
Currently, pregnant and lactating women are permitted to take general anti-inflammatory drugs, including ibuprofen. Further, many pregnant and lactating women are encouraged to increase their intake of omega-3 fatty acids, which have anti-inflammatory activities. The omega-3 fatty acids eicosapentaenioc acid (EPA) and docosahexaenoic acid (DHA) are found at high concentrations in fatty-fish. EPA and DHA directly inhibit arachidonic acid biosynthesis from linoleic acid by inhibiting delta 6 desaturase activity [114] and act as anti-inflammatory agents in part by directly blocking arachidonic acid synthesis; the parent molecule for many inflammatory cytokines [115]. For example, n-3 fatty acids have been shown to inhibit monocyte and macrophage IL-1β and TNFα expression [115, 116]. Fish oil has also been shown to decrease endotoxin-induced activation of NF-kB in monocytes and subsequent inflammatory gene expression driven by the NF-kB transcription factor [117]. Thus, it is reasonable to determine whether fish oil or other mild anti-inflammatory treatments targeted to involution prevent mammary cancer promotion and metastasis in preclinical models of PABC. These studies are currently underway in our laboratory.
Abbreviations
- PABC:
-
pregnancy-associated breast cancer
- TAM:
-
tumor-associated macrophages
- CSF-1:
-
colony stimulating factor-1
- MCP-1:
-
monocyte chemotactic protein-1
- EGF:
-
epithelial growth factor
- EGFR:
-
epithelial growth factor receptor
- CSF-1R:
-
colony stimulating factor-1 receptor
- MMTV:
-
mouse mammary tumor virus promoter
- PyMT:
-
polyoma middle-T oncoprotein
- TEB:
-
terminal end bud
- ECM:
-
extracellular matrix
- MMP:
-
matrix metalloproteinase
- IHC:
-
immunohistochemical
- uPA:
-
urokinase-type plasminogen activator
- GRO-1:
-
growth-related oncogene 1
- LRG:
-
leucine-rich α2-glycoprotein
- CCL:
-
chemokine C-C motif ligand
- CXCL:
-
chemokine C-X-C motif ligand
- MIP-1α:
-
macrophage inflammatory protein-1α
- LRP-1:
-
low density lipoprotein-related protein 1
- LPS:
-
lipopolysaccharide
- IFN-γ:
-
interferon-γ
- IFN-β:
-
interleukin-1β
- TNF-α:
-
tumor necrosis factor-α
- IL:
-
interleukin
- TLR:
-
toll-like receptors
- TGF-β:
-
tumor growth factor-β
- MHC:
-
major histocompatibility complex
- Th1:
-
T-helper 1
- Th2:
-
T-helper 2
- iNOS:
-
inducible nitric oxide synthase
- EM:
-
electron microscopy
- DCIS:
-
ductal carcinoma in situ
- SHG:
-
second harmonic generation
- eGFP:
-
enhanced green fluorescent protein
- IGF-1:
-
insulin-like growth factor
- PDGF:
-
platelet-derived growth factor
- VEGF:
-
vascular endothelial growth factor
- bFGF:
-
basic fibroblast growth factor
- PGE2 :
-
prostaglandin E2
- STAT3:
-
signal transducer and activator of transcription 3
- EPA:
-
eicosapentaenioc acid
- DHA:
-
docosahexaenoic acid
- NF-kB:
-
nuclear factor kappa-light-chain-enhancer of activated B-cells
References
Balkwill F, Charles KA, Mantovani A. Smoldering and polarized inflammation in the initiation and promotion of malignant disease. Cancer Cell. 2005;7(3):211–7. doi:10.1016/j.ccr.2005.02.013.
Mantovani A, Marchesi F, Porta C, Sica A, Allavena P. Inflammation and cancer: breast cancer as a prototype. Breast. 2007;16(Suppl 2):S27–33. doi:10.1016/j.breast.2007.07.013.
Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature. 2008;454(7203):436–44. doi:10.1038/nature07205.
Bingle L, Brown NJ, Lewis CE. The role of tumour-associated macrophages in tumour progression: implications for new anticancer therapies. J Pathol. 2002;196(3):254–65. doi:10.1002/path.1027.
Schedin P, Strange R, Mitrenga T, Wolfe P, Kaeck M. Fibronectin fragments induce MMP activity in mouse mammary epithelial cells: evidence for a role in mammary tissue remodeling. J Cell Sci. 2000;113(Pt 5):795–806.
Schedin P, Mitrenga T, McDaniel S, Kaeck M. Mammary ECM composition and function are altered by reproductive state. Mol Carcinog. 2004;41(4):207–20. doi:10.1002/mc.20058.
McDaniel SM, Rumer KK, Biroc SL, Metz RP, Singh M, Porter W, et al. Remodeling of the mammary microenvironment after lactation promotes breast tumor cell metastasis. Am J Pathol. 2006;168(2):608–20. doi:10.2353/ajpath.2006.050677.
Schedin P, O'Brien J, Rudolph M, Stein T, Borges V. Microenvironment of the involuting mammary gland mediates mammary cancer progression. J Mammary Gland Biol Neoplasia. 2007;12(1):71–82. doi:10.1007/s10911-007-9039-3.
Clarkson RW, Wayland MT, Lee J, Freeman T, Watson CJ. Gene expression profiling of mammary gland development reveals putative roles for death receptors and immune mediators in post-lactational regression. Breast Cancer Res. 2004;6(2):R92–109. doi:10.1186/bcr754.
Stein T, Morris JS, Davies CR, Weber-Hall SJ, Duffy MA, Heath VJ, et al. Involution of the mouse mammary gland is associated with an immune cascade and an acute-phase response, involving LBP, CD14 and STAT3. Breast Cancer Res. 2004;6(2):R75–91. doi:10.1186/bcr753.
Schedin P. Pregnancy-associated breast cancer and metastasis. Nat Rev Cancer. 2006;6(4):281–91. doi:10.1038/nrc1839.
Bemis LT, Schedin P. Reproductive state of rat mammary gland stroma modulates human breast cancer cell migration and invasion. Cancer Res. 2000;60(13):3414–8.
Kelly PM, Davison RS, Bliss E, McGee JO. Macrophages in human breast disease: a quantitative immunohistochemical study. Br J Cancer. 1988;57(2):174–7.
Leek RD, Lewis CE, Whitehouse R, Greenall M, Clarke J, Harris AL. Association of macrophage infiltration with angiogenesis and prognosis in invasive breast carcinoma. Cancer Res. 1996;56(20):4625–9.
Tsutsui S, Yasuda K, Suzuki K, Tahara K, Higashi H, Era S. Macrophage infiltration and its prognostic implications in breast cancer: the relationship with VEGF expression and microvessel density. Oncol Rep. 2005;14(2):425–31.
Stanley ER, Berg KL, Einstein DB, Lee PS, Yeung YG. The biology and action of colony stimulating factor-1. Stem Cells. 1994;12(Suppl 1):15–24. discussion 25.
Wang JM, Griffin JD, Rambaldi A, Chen ZG, Mantovani A. Induction of monocyte migration by recombinant macrophage colony-stimulating factor. J Immunol. 1988;141(2):575–9.
Kacinski BM, Scata KA, Carter D, Yee LD, Sapi E, King BL, et al. FMS (CSF-1 receptor) and CSF-1 transcripts and protein are expressed by human breast carcinomas in vivo and in vitro. Oncogene. 1991;6(6):941–52.
McDermott RS, Deneux L, Mosseri V, Vedrenne J, Clough K, Fourquet A, et al. Circulating macrophage colony stimulating factor as a marker of tumour progression. Eur Cytokine Netw. 2002;13(1):121–7.
Scholl SM, Crocker P, Tang R, Pouillart P, Pollard JW. Is colony-stimulating factor-1 a key mediator of breast cancer invasion and metastasis? Mol Carcinog. 1993;7(4):207–11. doi:10.1002/mc.2940070402.
Beck AH, Espinosa I, Edris B, Li R, Montgomery K, Zhu S, et al. The macrophage colony-stimulating factor 1 response signature in breast carcinoma. Clin Cancer Res. 2009;15((3):778–87. doi:10.1158/1078-0432.CCR-08-1283.
Ueno T, Toi M, Saji H, Muta M, Bando H, Kuroi K, et al. Significance of macrophage chemoattractant protein-1 in macrophage recruitment, angiogenesis, and survival in human breast cancer. Clin Cancer Res. 2000;6(8):3282–9.
O'Sullivan C, Lewis CE, Harris AL, McGee JO. Secretion of epidermal growth factor by macrophages associated with breast carcinoma. Lancet. 1993;342(8864):148–9. doi:10.1016/0140-6736(93)91348-P.
Leek RD, Hunt NC, Landers RJ, Lewis CE, Royds JA, Harris AL. Macrophage infiltration is associated with VEGF and EGFR expression in breast cancer. J Pathol. 2000;190(4):430–6. doi:10.1002/(SICI)1096-9896(200003)190:4<430::AID-PATH538>3.0.CO;2-6.
Sainsbury JR, Farndon JR, Needham GK, Malcolm AJ, Harris AL. Epidermal-growth-factor receptor status as predictor of early recurrence of and death from breast cancer. Lancet. 1987;1(8547):1398–402.
Wyckoff J, Wang W, Lin EY, Wang Y, Pixley F, Stanley ER, et al. A paracrine loop between tumor cells and macrophages is required for tumor cell migration in mammary tumors. Cancer Res. 2004;64(19):7022–9. doi:10.1158/0008-5472.CAN-04-1449.
Scholl SM, Pallud C, Beuvon F, Hacene K, Stanley ER, Rohrschneider L, et al. Anti-colony-stimulating factor-1 antibody staining in primary breast adenocarcinomas correlates with marked inflammatory cell infiltrates and prognosis. J Natl Cancer Inst. 1994;86(2):120–6. doi:10.1093/jnci/86.2.120.
Tang R, Beuvon F, Ojeda M, Mosseri V, Pouillart P, Scholl S. M-CSF (monocyte colony stimulating factor) and M-CSF receptor expression by breast tumour cells: M-CSF mediated recruitment of tumour infiltrating monocytes? J Cell Biochem. 1992;50(4):350–6. doi:10.1002/jcb.240500403.
Flick MB, Sapi E, Perrotta PL, Maher MG, Halaban R, Carter D, et al. Recognition of activated CSF-1 receptor in breast carcinomas by a tyrosine 723 phosphospecific antibody. Oncogene. 1997;14(21):2553–61. doi:10.1038/sj.onc.1201092.
Mizukami Y, Nonomura A, Noguchi M, Taniya T, Koyasaki N, Saito Y, et al. Immunohistochemical study of oncogene product ras p21, c-myc and growth factor EGF in breast carcinomas. Anticancer Res. 1991;11(4):1485–94.
Lin EY, Nguyen AV, Russell RG, Pollard JW. Colony-stimulating factor 1 promotes progression of mammary tumors to malignancy. J Exp Med. 2001;193(6):727–40. doi:10.1084/jem.193.6.727.
Aharinejad S, Paulus P, Sioud M, Hofmann M, Zins K, Schafer R, et al. Colony-stimulating factor-1 blockade by antisense oligonucleotides and small interfering RNAs suppresses growth of human mammary tumor xenografts in mice. Cancer Res. 2004;64(15):5378–84. doi:10.1158/0008-5472.CAN-04-0961.
Paulus P, Stanley ER, Schafer R, Abraham D, Aharinejad S. Colony-stimulating factor-1 antibody reverses chemoresistance in human MCF-7 breast cancer xenografts. Cancer Res. 2006;66(8):4349–56. doi:10.1158/0008-5472.CAN-05-3523.
Goswami S, Sahai E, Wyckoff JB, Cammer M, Cox D, Pixley FJ, et al. Macrophages promote the invasion of breast carcinoma cells via a colony-stimulating factor-1/epidermal growth factor paracrine loop. Cancer Res. 2005;65(12):5278–83. doi:10.1158/0008-5472.CAN-04-1853.
Gouon-Evans V, Rothenberg ME, Pollard JW. Postnatal mammary gland development requires macrophages and eosinophils. Development. 2000;127(11):2269–82.
Lanigan F, O'Connor D, Martin F, Gallagher WM. Molecular links between mammary gland development and breast cancer. Cell Mol Life Sci. 2007;64(24):3159–84. doi:10.1007/s00018-007-7386-2.
Schwertfeger KL, Rosen JM, Cohen DA. Mammary gland macrophages: pleiotropic functions in mammary development. J Mammary Gland Biol Neoplasia. 2006;11(3–4):229–38. doi:10.1007/s10911-006-9028-y.
Pollard JW, Hennighausen L. Colony stimulating factor 1 is required for mammary gland development during pregnancy. Proc Natl Acad Sci USA. 1994;91(20):9312–6. doi:10.1073/pnas.91.20.9312.
Strange R, Li F, Saurer S, Burkhardt A, Friis RR. Apoptotic cell death and tissue remodelling during mouse mammary gland involution. Development. 1992;115(1):49–58.
Schedin PJ, Thackray LB, Malone P, Fontaine SC, Friis RR, Strange R. Programmed cell death and mammary neoplasia. Cancer Treat Res. 1996;83:3–22.
Walker NI, Bennett RE, Kerr JF. Cell death by apoptosis during involution of the lactating breast in mice and rats. Am J Anat. 1989;185(1):19–32. doi:10.1002/aja.1001850104.
Li M, Liu X, Robinson G, Bar-Peled U, Wagner KU, Young WS, et al. Mammary-derived signals activate programmed cell death during the first stage of mammary gland involution. Proc Natl Acad Sci USA. 1997;94(7):3425–30. doi:10.1073/pnas.94.7.3425.
Sorenson A, Knight CH. Restoration of lactation in mice after litter removal for various lengths of time. J Reprod Fertil Abstr Ser. 1997;19:46.
Lund LR, Romer J, Thomasset N, Solberg H, Pyke C, Bissell MJ, et al. Two distinct phases of apoptosis in mammary gland involution: proteinase-independent and -dependent pathways. Development. 1996;122(1):181–93.
Stein T, Salomonis N, Gusterson BA. Mammary gland involution as a multi-step process. J Mammary Gland Biol Neoplasia. 2007;12(1):25–35. doi:10.1007/s10911-007-9035-7.
Alexander CM, Selvarajan S, Mudgett J, Werb Z. Stromelysin-1 regulates adipogenesis during mammary gland involution. J Cell Biol. 2001;152(4):693–703. doi:10.1083/jcb.152.4.693.
Monks J, Smith-Steinhart C, Kruk ER, Fadok VA, Henson PM. Epithelial cells remove apoptotic epithelial cells during post-lactation involution of the mouse mammary gland. Biol Reprod. 2008;78(4):586–94. doi:10.1095/biolreprod.107.065045.
Monks J, Rosner D, Geske FJ, Lehman L, Hanson L, Neville MC, et al. Epithelial cells as phagocytes: apoptotic epithelial cells are engulfed by mammary alveolar epithelial cells and repress inflammatory mediator release. Cell Death Differ. 2005;12(2):107–14. doi:10.1038/sj.cdd.4401517.
Orlofsky A, Berger MS, Prystowsky MB. Novel expression pattern of a new member of the MIP-1 family of cytokine-like genes. Cell Regul. 1991;2(5):403–12.
Kasama T, Strieter RM, Standiford TJ, Burdick MD, Kunkel SL. Expression and regulation of human neutrophil-derived macrophage inflammatory protein 1 alpha. J Exp Med. 1993;178(1):63–72. doi:10.1084/jem.178.1.63.
Master SR, Hartman JL, D'Cruz CM, Moody SE, Keiper EA, Ha SI, et al. Functional microarray analysis of mammary organogenesis reveals a developmental role in adaptive thermogenesis. Mol Endocrinol. 2002;16(6):1185–203. doi:10.1210/me.16.6.1185.
Hanayama R, Nagata S. Impaired involution of mammary glands in the absence of milk fat globule EGF factor 8. Proc Natl Acad Sci USA. 2005;102(46):16886–91. doi:10.1073/pnas.0508599102.
Atabai K, Fernandez R, Huang X, Ueki I, Kline A, Li Y, et al. Mfge8 is critical for mammary gland remodeling during involution. Mol Biol Cell. 2005;16(12):5528–37. doi:10.1091/mbc.E05-02-0128.
Gouon-Evans V, Lin EY, Pollard JW. Requirement of macrophages and eosinophils and their cytokines/chemokines for mammary gland development. Breast Cancer Res. 2002;4(4):155–64. doi:10.1186/bcr441.
Mantovani A, Sica A, Sozzani S, Allavena P, Vecchi A, Locati M. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol. 2004;25(12):677–86. doi:10.1016/j.it.2004.09.015.
Mosser DM, Edwards JP. Exploring the full spectrum of macrophage activation. Nat Rev Immunol. 2008;8(12):958–69. doi:10.1038/nri2448.
Sica A, Schioppa T, Mantovani A, Allavena P. Tumour-associated macrophages are a distinct M2 polarised population promoting tumour progression: potential targets of anti-cancer therapy. Eur J Cancer. 2006;42(6):717–27. doi:10.1016/j.ejca.2006.01.003.
Mantovani A, Sozzani S, Locati M, Allavena P, Sica A. Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 2002;23(11):549–55. doi:10.1016/S1471-4906(02)02302-5.
Gordon S. Alternative activation of macrophages. Nat Rev Immunol. 2003;3(1):23–35. doi:10.1038/nri978.
Van Ginderachter JA, Movahedi K, Hassanzadeh Ghassabeh G, Meerschaut S, Beschin A, Raes G, et al. Classical and alternative activation of mononuclear phagocytes: picking the best of both worlds for tumor promotion. Immunobiology. 2006;211(6–8):487–501. doi:10.1016/j.imbio.2006.06.002.
Verrecchia F, Mauviel A. Transforming growth factor-beta signaling through the Smad pathway: role in extracellular matrix gene expression and regulation. J Invest Dermatol. 2002;118(2):211–5. doi:10.1046/j.1523-1747.2002.01641.x.
Noel JG, Guo X, Wells-Byrum D, Schwemberger S, Caldwell CC, Ogle CK. Effect of thermal injury on splenic myelopoiesis. Shock. 2005;23(2):115–22. doi:10.1097/01.shk.0000154239.00887.18.
Rabinowitz SS, Gordon S. Macrosialin, a macrophage-restricted membrane sialoprotein differentially glycosylated in response to inflammatory stimuli. J Exp Med. 1991;174(4):827–36. doi:10.1084/jem.174.4.827.
Masaki T, Chow F, Nikolic-Paterson DJ, Atkins RC, Tesch GH. Heterogeneity of antigen expression explains controversy over glomerular macrophage accumulation in mouse glomerulonephritis. Nephrol Dial Transplant. 2003;18(1):178–81. doi:10.1093/ndt/18.1.178.
McKnight AJ, Gordon S. Membrane molecules as differentiation antigens of murine macrophages. Adv Immunol. 1998;68:271–314. doi:10.1016/S0065-2776(08)60562-3.
Dickson SR, Warburton MJ. Enhanced synthesis of gelatinase and stromelysin by myoepithelial cells during involution of the rat mammary gland. J Histochem Cytochem. 1992;40(5):697–703.
Warburton MJ, Mitchell D, Ormerod EJ, Rudland P. Distribution of myoepithelial cells and basement membrane proteins in the resting, pregnant, lactating, and involuting rat mammary gland. J Histochem Cytochem. 1982;30(7):667–76.
Talhouk RS, Chin JR, Unemori EN, Werb Z, Bissell MJ. Proteinases of the mammary gland: developmental regulation in vivo and vectorial secretion in culture. Development. 1991;112(2):439–49.
Wahl LM, Wahl SM, Mergenhagen SE, Martin GR. Collagenase production by endotoxin-activated macrophages. Proc Natl Acad Sci USA. 1974;71(9):3598–601. doi:10.1073/pnas.71.9.3598.
Werb Z, Gordon S. Secretion of a specific collagenase by stimulated macrophages. J Exp Med. 1975;142(2):346–60. doi:10.1084/jem.142.2.346.
Welgus HG, Campbell EJ, Bar-Shavit Z, Senior RM, Teitelbaum SL. Human alveolar macrophages produce a fibroblast-like collagenase and collagenase inhibitor. J Clin Invest. 1985;76(1):219–24. doi:10.1172/JCI111949.
Nathan CF. Secretory products of macrophages. J Clin Invest. 1987;79(2):319–26. doi:10.1172/JCI112815.
Klimetzek V, Sorg C. Lymphokine-induced secretion of plasminogen activator by murine macrophages. Eur J Immunol. 1977;7(3):185–7. doi:10.1002/eji.1830070314.
Locati M, Deuschle U, Massardi ML, Martinez FO, Sironi M, Sozzani S, et al. Analysis of the gene expression profile activated by the CC chemokine ligand 5/RANTES and by lipopolysaccharide in human monocytes. J Immunol. 2002;168(7):3557–62.
Adams D, Hamilton T. Macrophages as destructive cells in host defense. In: Gallin JI, Goldstein IM, Snyderman R, editors. Inflammation: basic principles and clinical correlates. 2nd ed. New York: Raven Press; 1992. p. 637–662.
Alexander CM, Howard EW, Bissell MJ, Werb Z. Rescue of mammary epithelial cell apoptosis and entactin degradation by a tissue inhibitor of metalloproteinases-1 transgene. J Cell Biol. 1996;135(6 Pt 1):1669–77. doi:10.1083/jcb.135.6.1669.
Adair-Kirk TL, Atkinson JJ, Broekelmann TJ, Doi M, Tryggvason K, Miner JH, et al. A site on laminin alpha 5, AQARSAASKVKVSMKF, induces inflammatory cell production of matrix metalloproteinase-9 and chemotaxis. J Immunol. 2003;171(1):398–406.
Faisal Khan KM, Laurie GW, McCaffrey TA, Falcone DJ. Exposure of cryptic domains in the alpha 1-chain of laminin-1 by elastase stimulates macrophages urokinase and matrix metalloproteinase-9 expression. J Biol Chem. 2002;277(16):13778–86. doi:10.1074/jbc.M111290200.
Marom B, Rahat MA, Lahat N, Weiss-Cerem L, Kinarty A, Bitterman H. Native and fragmented fibronectin oppositely modulate monocyte secretion of MMP-9. J Leukoc Biol. 2007;81(6):1466–76. doi:10.1189/jlb.0506328.
Xie DL, Meyers R, Homandberg GA. Release of elastase from monocytes adherent to a fibronectin-gelatin surface. Blood. 1993;81(1):186–92.
Sudhakaran PR, Radhika A, Jacob SS. Monocyte macrophage differentiation in vitro: Fibronectin-dependent upregulation of certain macrophage-specific activities. Glycoconj J. 2007;24(1):49–55. doi:10.1007/s10719-006-9011-2.
Rudolph-Owen LA, Matrisian LM. Matrix metalloproteinases in remodeling of the normal and neoplastic mammary gland. J Mammary Gland Biol Neoplasia. 1998;3(2):177–89. doi:10.1023/A:1018746923474.
Hagemann T, Robinson SC, Schulz M, Trumper L, Balkwill FR, Binder C. Enhanced invasiveness of breast cancer cell lines upon co-cultivation with macrophages is due to TNF-alpha dependent up-regulation of matrix metalloproteases. Carcinogenesis. 2004;25(8):1543–9. doi:10.1093/carcin/bgh146.
Tryggvason K, Hoyhtya M, Pyke C. Type IV collagenases in invasive tumors. Breast Cancer Res Treat. 1993;24(3):209–18. doi:10.1007/BF01833261.
Domagala W, Striker G, Szadowska A, Dukowicz A, Weber K, Osborn M. Cathepsin D in invasive ductal NOS breast carcinoma as defined by immunohistochemistry. No correlation with survival at 5 years. Am J Pathol. 1992;141(5):1003–12.
Giannelli G, Falk-Marzillier J, Schiraldi O, Stetler-Stevenson WG, Quaranta V. Induction of cell migration by matrix metalloprotease-2 cleavage of laminin-5. Science. 1997;277(5323):225–8. doi:10.1126/science.277.5323.225.
Radnor CJ. Myoepithelium in involuting mammary glands of the rat. J Anat. 1972;112(Pt 3):355–65.
Martinez-Hernandez A, Fink LM, Pierce GB. Removal of basement membrane in the involuting breast. Lab Invest. 1976;34(5):455–62.
Condeelis J, Pollard JW. Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell. 2006;124(2):263–6. doi:10.1016/j.cell.2006.01.007.
Pollard JW. Macrophages define the invasive microenvironment in breast cancer. J Leukoc Biol. 2008;84(3):623–30. doi:10.1189/jlb.1107762.
Stoller P, Reiser KM, Celliers PM, Rubenchik AM. Polarization-modulated second harmonic generation in collagen. Biophys J. 2002;82(6):3330–42. doi:10.1016/S0006-3495(02)75673-7.
Zipfel WR, Williams RM, Christie R, Nikitin AY, Hyman BT, Webb WW. Live tissue intrinsic emission microscopy using multiphoton-excited native fluorescence and second harmonic generation. Proc Natl Acad Sci USA. 2003;100(12):7075–80. doi:10.1073/pnas.0832308100.
Ingman WV, Wyckoff J, Gouon-Evans V, Condeelis J, Pollard JW. Macrophages promote collagen fibrillogenesis around terminal end buds of the developing mammary gland. Dev Dyn. 2006;235(12):3222–9. doi:10.1002/dvdy.20972.
Wyckoff JB, Wang Y, Lin EY, Li JF, Goswami S, Stanley ER, et al. Direct visualization of macrophage-assisted tumor cell intravasation in mammary tumors. Cancer Res. 2007;67(6):2649–56. doi:10.1158/0008-5472.CAN-06-1823.
Sunderkotter C, Steinbrink K, Goebeler M, Bhardwaj R, Sorg C. Macrophages and angiogenesis. J Leukoc Biol. 1994;55(3):410–22.
Clark RA, Stone RD, Leung DY, Silver I, Hohn DC, Hunt TK. Role of macrophages in would healing. Surg Forum. 1976;27(62):16–8.
Hunt TK, Knighton DR, Thakral KK, Goodson WH 3rd, Andrews WS. Studies on inflammation and wound healing: angiogenesis and collagen synthesis stimulated in vivo by resident and activated wound macrophages. Surgery. 1984;96(1):48–54.
Thakral KK, Goodson WH 3rd, Hunt TK. Stimulation of wound blood vessel growth by wound macrophages. J Surg Res. 1979;26(4):430–6. doi:10.1016/0022-4804(79)90031-3.
Crowther M, Brown NJ, Bishop ET, Lewis CE. Microenvironmental influence on macrophage regulation of angiogenesis in wounds and malignant tumors. J Leukoc Biol. 2001;70(4):478–90.
Brown LF, Yeo KT, Berse B, Yeo TK, Senger DR, Dvorak HF, et al. Expression of vascular permeability factor (vascular endothelial growth factor) by epidermal keratinocytes during wound healing. J Exp Med. 1992;176(5):1375–9. doi:10.1084/jem.176.5.1375.
Kovacs EJ, DiPietro LA. Fibrogenic cytokines and connective tissue production. FASEB J. 1994;8(11):854–61.
Lewis C, Murdoch C. Macrophage responses to hypoxia: implications for tumor progression and anti-cancer therapies. Am J Pathol. 2005;167(3):627–35.
De Palma M, Venneri MA, Galli R, Sergi Sergi L, Politi LS, Sampaolesi M, et al. Tie2 identifies a hematopoietic lineage of proangiogenic monocytes required for tumor vessel formation and a mesenchymal population of pericyte progenitors. Cancer Cell. 2005;8(3):211–26. doi:10.1016/j.ccr.2005.08.002.
Lin EY, Li JF, Gnatovskiy L, Deng Y, Zhu L, Grzesik DA, et al. Macrophages regulate the angiogenic switch in a mouse model of breast cancer. Cancer Res. 2006;66(23):11238–46. doi:10.1158/0008-5472.CAN-06-1278.
Lin EY, Li JF, Bricard G, Wang W, Deng Y, Sellers R, et al. VEGF Restores Delayed Tumor Progression in Tumors Depleted of Macrophages. Mol Oncol. 2007;1(3):288–302. doi:10.1016/j.molonc.2007.10.003.
Djonov V, Andres AC, Ziemiecki A. Vascular remodelling during the normal and malignant life cycle of the mammary gland. Microsc Res Tech. 2001;52(2):182–9. doi:10.1002/1097-0029(20010115)52:2<182::AID-JEMT1004>3.0.CO;2-M.
Matsumoto M, Nishinakagawa H, Kurohmaru M, Hayashi Y, Otsuka J. Pregnancy and lactation affect the microvasculature of the mammary gland in mice. J Vet Med Sci. 1992;54(5):937–43.
Pepper MS, Baetens D, Mandriota SJ, Di Sanza C, Oikemus S, Lane TF, et al. Regulation of VEGF and VEGF receptor expression in the rodent mammary gland during pregnancy, lactation, and involution. Dev Dyn. 2000;218(3):507–24. doi:10.1002/1097-0177(200007)218:3<507::AID-DVDY1012>3.0.CO;2-5.
Nickerson SC. Immunological aspects of mammary involution. J Dairy Sci. 1989;72(6):1665–78.
Oliver SP, Sordillo LM. Approaches to the manipulation of mammary involution. J Dairy Sci. 1989;72(6):1647–64.
Alonzi T, Maritano D, Gorgoni B, Rizzuto G, Libert C, Poli V. Essential role of STAT3 in the control of the acute-phase response as revealed by inducible gene inactivation [correction of activation] in the liver. Mol Cell Biol. 2001;21(5):1621–32. doi:10.1128/MCB.21.5.1621-1632.2001.
Chapman RS, Lourenco PC, Tonner E, Flint DJ, Selbert S, Takeda K, et al. Suppression of epithelial apoptosis and delayed mammary gland involution in mice with a conditional knockout of Stat3. Genes Dev. 1999;13(19):2604–16. doi:10.1101/gad.13.19.2604.
Hanayama R, Tanaka M, Miyasaka K, Aozasa K, Koike M, Uchiyama Y, et al. Autoimmune disease and impaired uptake of apoptotic cells in MFG-E8-deficient mice. Science. 2004;304(5674):1147–50. doi:10.1126/science.1094359.
Garg ML, Thomson AB, Clandinin MT. Interactions of saturated, n-6 and n-3 polyunsaturated fatty acids to modulate arachidonic acid metabolism. J Lipid Res. 1990;31(2):271–7.
Calder PC. Polyunsaturated fatty acids and inflammation. Prostaglandins Leukot Essent Fatty Acids. 2006;75(3):197–202. doi:10.1016/j.plefa.2006.05.012.
Kroman N, Mouridsen HT. Prognostic influence of pregnancy before, around, and after diagnosis of breast cancer. Breast. 2003;12(6):516–21. doi:10.1016/S0960-9776(03)00159-0.
Novak TE, Babcock TA, Jho DH, Helton WS, Espat NJ. NF-kappa B inhibition by omega -3 fatty acids modulates LPS-stimulated macrophage TNF-alpha transcription. Am J Physiol Lung Cell Mol Physiol. 2003;284(1):L84–9.
Acknowledgements
We would like to thank Douglas K. Graham, MD, PhD, and Manoj Pillai, MD, for critical review of the manuscript.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Financial Support: This work was supported in part by grants from The Avon and Mary Kay Ash Foundations, and DOD BC060531 to PS and a DOD Predoctoral Grant BC073482 to JO.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
O’Brien, J., Schedin, P. Macrophages in Breast Cancer: Do Involution Macrophages Account for the Poor Prognosis of Pregnancy-Associated Breast Cancer?. J Mammary Gland Biol Neoplasia 14, 145–157 (2009). https://doi.org/10.1007/s10911-009-9118-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10911-009-9118-8