Abstract
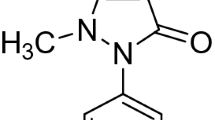
Two new thione-based nickel complexes, viz., [Ni(Tmipr)2Cl2]⋅2H2O 1 and [Ni(Ttxyly)2] ⋅H2O⋅2CH3OH 2 {Tmipr = 1-(2-isopropylphenyl)-1H-imidazole-2(3H)-thione; Ttxylyl− = hydrotris[1-(2,6-dimethylphenyl)-1H-imidazole-2(3H)-thione]borate anion} have been synthesized. Their structures and properties were characterized by several physicochemical methods, namely elemental and thermal analysis, FT-IR and Raman and UV–Vis spectroscopes, as well as electrical molar conductivity measurements. Structural determination of complex 2 showed that the coordination geometry around nickel atom is distorted tetrahedral with four thione sulfur donors from two molecules of the ligand KTtxylyl. Steric interactions between the xylyl rings appear to be responsible for the formation of the mononuclear nickel(II) complex 2. Molecules of complex 2 are connected via S–H···O hydrogen bonds, involving hydrogen atoms of the coordinated thiones and lattice water molecules. The superoxide dismutase biomimetic catalytic activities of both complexes were tested and found to be promising candidates as functional mimic enzyme to serve for complete superoxide radical’s detoxification. The observed IC50 values of these complexes are 8.38 and 7.4 mM for complexes 1 and 2, respectively.









Similar content being viewed by others
References
D. Fusco, G. Colloca, M.R.L. Monaco, M. Cesari, Clin. Interv. Aging 2, 377 (2007)
B. Poljsak, Oxidative Med. Cell. Longev. 2011, 1 (2011)
W. Dröge, Physiol. Rev. 82, 47 (2002)
B. Uttara, A.V. Singh, P. Zamboni, R.T. Mahajan, Curr. Neuropharmacol. 7, 65 (2009).
P. Pacherm, J.S. Beckman, L. Liaudet, Physiol. Rev. 87, 315 (2007)
J. Zhao, Plant Signal Behav. 2, 544 (2007)
V. Lobo, A. Patil, A. Phatak, N. Chandra, Pharmacogn. Rev. 4, 118 (2010).
S.R. Maxwell, Drugs 9, 345 (1995)
A. Camins, F. Junyent, E. Verdaguer, C. Beas-Zarate, A.E. Rojas-Mayorquín, D. Ortuño-Sahagún, M. Pallàs, Pharmaceuticals (Basel) 2, 194 (2009)
S.C. Shivhare, A.O. Patidar, K.G. Malviya, K.K. Shivhare-Malviya, Ayu 32, 388 (2011)
H.S. Buttar, T. Li, N. Ravi, Exp. Clin. Cardiol. 10, 229 (2005)
S.L. Mehta, Y. Lin, W. Chen, F. Yu, L. Cao, Q. He, P.H. Chan, P.A. Li, Transl. Stroke Res. 2, 42 (2011)
S.W. Ragsdale, J. Biol. Chem. 284, 18571 (1999)
K.C. Ryan, A.I. Guce, O.E. Johnson, T.C. Brunold, D.E. Cabelli, S.C. Garman, M.J. Maroney, Biochemistry 54, 1016 (2015)
B. Palenic, B. Brahamsha, F.W. Larimer, M. Land, L. Hauser, P. Chain, J. Lamerdin, W. Regala, E.E. Allen, J. McCarren, I. Paulsen, A. Dufresne, F. Partensky, E.A. Webb, J. Waterbury, Nature 424, 1037 (2003)
S.B. Choudhury, J.W. Lee, G. Davidson, Y.I. Yim, K. Bose, M.L. Sharma, S.O. Kang, D.E. Cabelli, M.J. Maroney, Biochemistry 38, 3744 (1999).
D.J. Wuerges, J.-W. Lee, Y.-I. Yim, H.-S. Yim, S.-O. Kang, K.D. Carugo, Proc. Natl. Acad. Sci. USA 101, 8569 (2004)
Z. Xu, S. Pan, G. Li, Y. He, R. Wang, J. Inorg. Organomet. Polym. 25, 1313 (2015)
G. Yan, Y. He, G. Li, Y. Xiong, P. Song, R. Wang, J. Chem. Sci. 11, 1783 (2016)
G. Li, H. Zhang, R. Wang, Y. Feng, H. Yu, B. Xiong, Chin. Sci. Bull. 58, 2956 (2013)
S.K. Chatterjee, R.C. Maji, S.K. Barman, M.M. Olmstead, A.K. Patra, Angew. Chem. Int. Ed. Engl. 53, 10184 (2014)
D. Nakane, Y. Wasada-Tsutsui, Y. Funahashi, T. Hatanaka, T. Ozawa, H. Masuda, Inorg. Chem. 53, 6512 (2014)
A. Prokop, J.M. Davidson, J. Pharm. Sci. 9, 3518 (2008)
I. Batinić-Haberle, J.S. Rebouças, I. Spasojević, Antioxid. Redox Signal 13, 877 (2010)
M.M. Ibrahim, A.M. Ramadan, G.A.M. Mersal, S.A. El-Shazly, J. Mol. Struct. 998, 1 (2011)
A.M. Ramadan, M.M. Ibrahim, S.Y. Shaban, J. Mol. Struct. 1006, 348 (2011)
S.Y. Shaban, A.M. Ramadan, M.M. Ibrahim, M.A. Mohamed, R. Van Eldik, Dalton Trans. 44, 14110 (2015)
M.M. Ibrahim, A.M. Ramadan, M.A. Mohamed, M. Soliman, S.I.M. Zayed, J. Coord. Chem. 18, 4296 (2015)
A.M. Ramadan, J. Mol. Struct. 1015, 56 (2012)
A.M. Ramadan, J. Coord. Chem. 65(8), 1417 (2012)
M.M.M. Ibrahim, S.Y. Shaban, Inorg. Chim. Acta 362, 1471 (2009)
M.M. Ibrahim, J. Mol. Struct. 937, 50 (2009)
M.M. Ibrahim, C.P. Olmo, T. Tekeste, J. Seebacher, G. He, J.A.M. Calvo, K. Böhmerle, G. Steinfeld, H. Brombacher, H. Vahrenkamp, Inorg. Chem. 45, 7493 (2006)
M.M. Ibrahim, J. Seebacher, G. Steinfeld, H. Vahrenkamp, Inorg. Chem. 44, 8531 (2005)
M.M. Ibrahim, G. He, J. Seebacher, B. Benkmil, H. Vahrenkamp, Eur. J. Inorg. Chem. 2005, 4070–4077 (2005)
SAINT, V6.02, BrukerAXS, Madison, WI (1999)
G.M. Sheldrick, SADABS, Area-Detector Absorption Correction, Göttingen, Germany (1996)
G.M. Sheldrick, SHELXL2013. University of Göttingen, Germany (2013)
L. Lutterotti, MAUD, CPD, Newsletter (IUCr) 24 (2000)
M. Nishikimi, N.A. Roa, K. Yogi, Biochem. Biophys. Res. Commun. 46, 849–854 (1972)
A.M. Ramadan, R.M. Issa, Trans. Met. Chem. 30, 529 (2005)
P.N. Patel, K.K. Shukla, A. Singh, D.K. Patel, J. Niclos-Gutierrez, D. Choquerillo-Lazarte, J. Coord. Chem. 63, 3648 (2010)
M. Chun, S. Lee, S. Yang, J. Colloid Interface Sci. 266, 120 (2003)
R. Shunmugam, D. Sathyanarayana, J. Coord. Chem. 12, 151 (1983)
E.S. Raper, J.L. Books, J. Inorg. Nucl. Chem. 39, 2163 (1977).
L.J. Bellamy, The infrared spectra of complex molecules, 3rd edn. (Chapman and Hall, London, 1975)
E.S. Raper, P.H. Crackett, Inorg. Chim. Acta 50, 159 (1981)
M.O. Albers, S.F.A. Crosby, D.C. Liles, D.J. Robinson, A. Shaver, E. Singleton, Organometallics 6, 2014 (1987)
J.C. Bommer, K.W. Morse, J. Am. Chem. Soc. 96, 6222 (1974)
J.C. Bommer, K.W. Morse, Inorg. Chem. 19, 587 (1980)
G.G. Lobbia, C. Pettinari, C. Santini, N. Somers, B.W. Skelton, A.H. White, Inorg. Chim. Acta 319, 15 (2001)
P.D. Bailey, A. Dawson, C. McCormack, S.A. Moggach, I.D.H. Oswald, D.W.H. Rankin, A. Turner, Inorg. Chem. 44, 8884 (2005)
D.V. Patel, J. Mihalcik, K.A. Kreisel, G.P.A. Yap, L.N. Zakharov, W.S. Kassel, A.L. Rheingold, D. Rabinovich, Dalton Trans. 14, 2410 (2005)
M. Careri, L. Elviri, M. Lanfranchi, L. Marchiò, C. Mora, M.A. Pellinghelli, Inorg. Chem. 42, 2109 (2003)
R. Cammi, M. Gennari, M. Giannetto, M. Lanfranchi, L. Marchiò, G. Morri, C. Paiola, M.A. Pellinghelli, Inorg. Chem. 44, 433 (2005)
S. Kiani, J.R. Long, P. Stavropoulos, Inorg. Chim. Acta 263, 357 (1997)
M. Gennari, M. Lanfranchi, L. Marchiò, M.A. Pellinghelli, M. Tegoni, R. Cammi, Inorg. Chem. 45, 3456 (2006)
H. Takeuchi, I. Harada, Spectrochim. Acta 49, 1069 (1986).
R.L. Dutta, A. Syamal, Elements of magnetochemistry, 2nd edn. (Affiliated East-West Press, Delhi, 2007)
B.N. Figgis, J. Lewis, Prog. Inorg. Chem. 6, 37 (1964)
A.B.P. Lever, Inorganic electronic spectroscopy, 2nd edn. (Elsevier, Amsterdam, 1984)
M. Amirnasr, A.H. Mahmoudkhani, A. Gorji, S. Dehghanpour, H.R. Bijanzadeh, Polyhedron 21, 2733 (2002)
H.A. Bayoumi, A.M.A. Alaghaz, M.S. Aljahdali, Int. J. Electrochem. Sci. 8, 9399 (2013)
B.M. Bridgewater, T. Fillebeen, R.A. Friesner, G. Parkin, J. Chem. Soc. Dalton Trans. 24, 4494 (2000)
C. Kimblin, B.M. Bridgewater, D.G. Churchill, T. Hascall, G. Parkin, Inorg. Chem. 39, 4240 (2000)
H. Vahrenkamp, Acc. Chem. Res. 32, 589 (1999)
M. Tesmer, M. Shu, H. Vahrenkamp, Inorg. Chem. 40, 4022 (2001)
M.M. Ibrahim, G.A.M. Mersal, S. Al-Juaid, S. El-Shazly, J. Mol. Struct. 166, 1056 (2014)
M.M. Ibrahim, S.S. Al-Juaid, Q. Mohsen, Phosphorus Sulfur Silicon 184, 2324 (2009)
G. Ischia, H.-R. Wenk, L. Lutterotti, F. Berberich, Quantitative Rietveld texture analysis from single synchrotron diffraction images. J. Appl. Cryst. 38, 377 (2005)
J.R.J. Sorenson (Ed.), Inflammatory diseases and copper, Humana Press, Clifton, 1982)
J.R.J. Sorenson, in Metal ions in biological systems, vol. 14, ed. by H. Sigel (Marcel Dekker, New York, 1982), p. 77
R.N. Patel, N. Singh, K.K. Shukla, V.L.N. Gundla, U.K. Chauhan, Spectrochim. Acta A 63, 21 (2006)
H.D. Youn, E.J. Kim, J.H. Roe, Y.C. Hah, S.O. Kang, Biochem. J. 318, 889 (1996)
S.L. Lippard, J. M. Berg, Principles of bioinorganic chemistry. (University Science Books, Mill Valley, 1994)
E.J. Baran, Quımica bioinorganica. (McGraw-Hill Interamericana de Espana S, McGraw-Hill Interamericana de Espana S.A, Madrid, 1995)
A.L. Abuhijleh, J. Khalaf, Eur. J. Med. Chem. 45, 3811 (2010)
Acknowledgements
This work was financially supported by Taif University, Saudi Arabia, Project No.: 1-437-5309.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ibrahim, M.M., Ramadan, A.EM.M., Shaban, S.Y. et al. Thione-Based Nickel(II) Complexes as Functional Antioxidant Mimics: Scavenging Activity of Reactive Oxygen Species O2 −· and X-Ray Crystal Structure of [Ni(Ttxyly)2]2 {Ttxylyl = Hydrotris(2-mercapto-1-xylyl-imidazolyl)borate}. J Inorg Organomet Polym 27, 1252–1263 (2017). https://doi.org/10.1007/s10904-017-0573-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-017-0573-1