Abstract
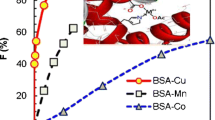
A kind of novel biopolymer antioxidant (BSA/HOSalenM, M=Co, Mn, Zn) is prepared with conjugation, which increases the antioxidant activity of the bovine serum albumin (BSA). The conjugations have been characterized by IR spectra, UV-Vis spectra, Fluorescence spectra, Circular dichroism (CD) spectra and Native-PAGE. The BSA is used as a biopolymer scaffold, and the insoluble Salen Schiff-base metal complexes HOSalenM make axial coordination with the amino acid residues of the BSA. The structure of the BSA is unchanged when the binding rate of HOSalenCo is less than 10. The HOSalenCo conjugations show an excellent hydroxyl radical (·OH) scavenging activity, and the activity (EC50) of BSA/HOSalenCo(10) (BSA:HOSalenCo=1:10) is improved by two orders of magnitude compared with the BSA, while the activity of the BSA/HOSalenMn is weak and the BSA/HOSalenZn shows no scavenging activity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Qi H M, Zhang Q B, Zhao T T, et al. Antioxidant activity of different sulfate content derivatives of polysaccharide extracted from Ulva pertusa (Chlorophyta) in vitro. Int J Biol Macromol, 2005, 37: 195–199
Koppenol W H. Names for inorganic radicals. Pure Appl Chem, 2000, 72: 437–446
Cadenas E, Davies K J A. Mitochondrial free radical generation, oxidative stress and aging. Free Radic Biol Med, 2000, 29: 222–230
Liu Y H, Lin Y Sh, Lee T L, et al. Semicarbazide-sensitive amine oxidase inhibitory and hydroxyl radical scavenging activities of aspartic acid β-hydroxamate and glutamic acid γ-monohydroxamate. Botan Stud, 2012, 53: 301–306
Nandita S, Rajini P S. Free radical scavenging activity of an aqueous extract of potato peel. Food Chem, 2004, 85: 611–616
Shibata T, Ishimaru K, Kawaguchi S, et al. Antioxidant activities of phlorotannins isolated from Japanese Laminariaceae. J Appl Phycol, 2008, 20: 705–711
Ohse T, Nagaoka S, Arakawa Y, et al. Cell death by reactive oxygen species generated from water-soluble cationic metalloporphyrins as superoxide dismutase mimics. J Inorg Biochem, 2001, 85: 201–208
Vanco J, Svajlenova O, Racanska E, et al. Antiradical activity of different copper (II) Schiff base complexes and their effect on alloxan-induced diabetes. J Trace Elem Med Biol, 2004, 18: 155–161
Lacikova L, Muselik J, Masterova I, et al. Antioxidant activity and phenols compounds in different extracts of four staphylea L. Species. Molecules, 2007, 12: 98–102
Santos M L P, Faljoni-Alario A, Mangrich A S, et al. Ferreira, antioxidant and pro-oxidant properties of some diimine-copper(II) complexes. J Inorg Biochem, 1998, 71: 71–78
Tarafder M T H, Kasbollah A, Crouse K A, et al. Synthesis and characterization of Zn(II) and Cd(II) complexes of S-benzyl-β-N-(2-pyridyl) methylenedithiocarbazate (HNNS): Bioactivity of the HNNS Schiff base and its Zn(II), Cu(II) and Cd(II) complexes and the X-ray structure of the [Zn(NNS)2] complex. Polyhedron, 2001, 20: 2363–2370
Peters T. All About Albumin: Biochemistry Genetics and Medical Applications. San Diego, CA: Academic Press Inc, 1995. 25
Peter K, Svend H, Ib J, et al. Albumin binding and time action of acylated insulins in various species. J Pharm Sci, 1995, 312: 725–731
Curry S, Madelkow H, Brick P, et al. Crystal structure of human serum albumin complexed with fatty acid reveals an asymmetric distribution of binding sites. Nat Struct Biol, 1998, 5: 827–835
Yin X C, Wang R M, He Y F, et al. Artifical oxygen carriers. Prog Chem, 2011, 23: 963–973
Gupta P K, Hung C T, Perrier D G. Albumin microspheres, I: Release characteristics of adriamycin. Int J Pharm, 1986: 33: 137–146
Brelle M C, Torres-Martinez C L, McNulty J C, et al. Synthesis and characterization of CuxS nanoparticles. Nature of the infrared band and charge-carrier dynamics. Pure Appl Chem, 2000, 72: 101–117
Sheffield W P, Eltringham-Smith L J, Gataiance S, et al. Addition of a sequence from α2-antiplasmin transforms human serum albumin into a blood clot component that speeds clot lysis. BMC Biotechnol, 2009, 9: 15–21
Wang R M, Komatsu T, Nakagawa A, et al. Human serum albumin bearing covalently attached iron(II) porphyrins as O2-coordination Sites. Bioconjugate Chem, 2005, 15: 23–25
Komatsu T, Wang R M, Zunszain P A, et al. Photosensitized reduction of water to hydrogen using human serum albumin complexed with zinc-protoporphyrin IX. J Am Chem Soc, 2006: 128: 16297–16301
Moran E C, Kamiguti A S, Cawley J C, et al. Cytoprotective antioxidant activity of serum albumin and autocrine catalase in chronic lymphocytic leukaemia. Br J Haematol, 2002, 116: 316–328
Pall T, Benjamin D, Kelly V. Well-defined protein-polymer conjugates—Synthesis and potential applications. Appl Microbiol Biotechnol, 2006, 73: 243–254
Pfeiffer P, Breith E, Lubbe E, et al. Tricyclic ortho-condensed partial valence rings. Justus, Liebigs. Ann Chem, 1993, 503: 84–130
Yin X C, Li X X, Wang R M, et al. Bifunctional antioxidant enzyme mimics of albumin-binding salphen Schiff-base metal complexes. Pure Appl Chem, 2012, 84: 2641–2652
Komatsu T, Ohmichi N, Nakagawa A, et al. O2 and CO binding properties of artificial hemoproteins formed by complexing iron protoporphyrin IX with human serum albumin mutants. J Am Chem Soc, 2005, 127: 15933–15942
Boelens R, Sheek R M, Dijkstra K, et al. Sequential assignment of imino- and amino-proton resonances in 1H NMR spectra of oligonucleotides by two-dimensional NMR spectroscopy. Application to a lac operator fragment. J Magn Reson, 1985, 62: 378–386
Quiming N S, Vergel R B, Nicolas M G, et al. Interaction of bovine serum albumin and metallothionin. J Health Sci, 2005, 51: 8–15
Shrivastava H Y, Kanthimathi M, Niar B U. Interaction of Schiff base with bovine serum albumin: Site-specific photocleavage. Biochem Biophys Res Commun, 1999, 265: 311–314
Venyaminov S Y, Vasilenko K S. Determination of protein tertiary structure class from circular dichroism spectra. Anal Biochem, 1994, 222: 176–184
Zhou Y H, Fu H, Zhao W X, et al. Synthesis, structure and activity of supramolecular mimics for the active site and arg141 residue of copper zinc-superoxide dismutase. Inorg Chem, 2007, 46: 734–739
Hart P J, Balbirnie M M, Ogihara N L, et al. Structure-based mechanism for copper-zinc superoxide dismutase. Biochemistry, 1999, 38: 2167–2168
Mahfuz E, Omer I, Ibrahim T, et al. Determination of antioxidant activity and antioxidant compounds in wild edible mushrooms. J Food Compos Anal, 2007, 203: 337–345
Cai Y Z, Sun M, Xing J, et al. Structure-radical scavenging activity relationships of phenolic compounds from traditional Chinese medicinal plants. Life Sci, 2006, 78: 2872–2888
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Li, G., Zhang, H., Wang, R. et al. Preparation and antioxidant activity of albumin binding Salen Schiff-base metal complexes. Chin. Sci. Bull. 58, 2956–2963 (2013). https://doi.org/10.1007/s11434-013-5787-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-013-5787-1