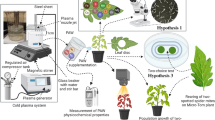
Abstract
Generalist insects show reduced selectivity when subjected to similar, but not identical, host plant chemical signatures. Here, we produced transgenic Arabidopsis thaliana plants that over-express genes regulating the aliphatic- and indolyl- glucosinolates biosynthetic pathways with either a constitutive (CaMV 35S) or a phloem-specific promoter (AtSUC2). This allowed us to examine how exposure to high levels of aliphatic- or indolyl-glucosinolates in homogenous habitats (leaf cage apparatus containing two wild-type or two transgenic leaves) and heterogeneous habitats (leaf cage apparatus containing one wild-type and one transgenic leaf) affects host selection and performance of Bemsia tabaci, a generalist phloem-feeding insect. Data from homogenous habitats indicated that exposure to A. thaliana plants accumulating high levels of aliphatic- or indolyl-glucosinolates negatively affected the performance of both adult females and nymphs of B. tabaci. Data from heterogeneous habitats indicated that B. tabaci adult females selected for oviposition plants on which their offspring perform better (preference-performance relationship). However, the combinations of wild-type and transgenic plants in heterogeneous habitats increased the period of time until the first choice was made and led to increased movement rate on transgenic plants, and reduced fecundity on wild-type plants. Overall, our findings are consistent with the view that both performance and selectivity of B. tabaci decrease in heterogeneous habitats that contain plants with closely-related chemical signatures.





Similar content being viewed by others
References
Agerbirk N, Vos M, Kim JH, Jander G (2009) Indole glucosinolate breakdown and its biological effects. Phytochem Rev 8:101–120
Bernays EA (1999) When host choice is a problem for a generalist herbivore: experiments with the whitefly, Bemisia tabaci. Ecol Entomol 24:260–267
Bernays EA (2001) Neural limitations in phytophagous insects: implications for diet breadth and evolution of host affiliation. Annu Rev Entomol 46:703–727
Bird TL, Krüger K (2006) Response of the polyphagous whitefly Bemisia tabaci B-biotype (Hemiptera: Aleyrodidae) to crop diversification—influence of multiple sensory stimuli on activity and fecundity. Bull Entomol Res 96:15–23
Bleeker PM, Diergaarde PJ, Ament K, Guerra J, Weidner M, Schütz S, de Both MTJ, Haring MA, Schuurink RC (2009) The role of specific tomato volatiles in tomato-whitefly interaction. Plant Physiol 151:925–935
Buckner JS, Freeman TP, Ruud RL, Chu C, Henneberry TJ (2002) Characterization and functions of the whitefly egg pedicel. Arch Insect Biochem 49:22–33
Burow M, Müller R, Gershenzon J, Wittstock U (2006) Altered glucosinolate hydrolysis in genetically engineered Arabidopsis thaliana and its influence on the larval development of Spodoptera littoralis. J Chem Ecol 32:2333–2349
Celenza JL, Quiel JA, Smolen GA, Merrikh H, Silvestro AR, Normanly J, Bender J (2005) The Arabidopsis ATR1 Myb transcription factor controls indolic glucosinolate homeostasis. Plant Physiol 37:253–262
Chen S, Petersen BL, Olsen CE, Schulz A, Halkier BA (2001) Long-distance phloem transport of glucosinolates in Arabidopsis. Plant Physiol 127:194–201
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Egan SP, Funk DJ (2006) Individual advantages to ecological specialization: insights on cognitive constraints from three conspecific taxa. Proc Roy Soc B Biol Sci 273:843–848
Elbaz M, Halon E, Malka O, Malitsky S, Blum E, Aharoni A, Morin S (2012) Asymmetric adaptation to indolic and aliphatic glucosinolates in the B and Q sibling species of Bemisia tabaci (Hemiptera: Aleyrodidae). Mol Ecol 21:4533–4546
Fox CW, Lalonde RG (1993) Host confusion and the evolution of insect diet breadths. Oikos 67:577
Frerigmann H, Böttcher C, Baatout D, Gigolashvili T (2012) Glucosinolates are produced in trichomes of Arabidopsis thaliana. Front Plant Sci 3:242
Gigolashvili T, Berger B, Mock HP, Müller C, Weisshaar B, Flügge UI (2007a) The transcription factor HIG1/MYB51 regulates indolic glucosinolate biosynthesis in Arabidopsis thaliana. Plant J 50:886–901
Gigolashvili T, Yatusevich R, Berger B, Müller C, Flügge UI (2007b) The R2R3-MYB transcription factor HAG1/MYB28 is a regulator of methionine-derived glucosinolate biosynthesis in Arabidopsis thaliana. Plant J 51:247–261
Gigolashvili T, Berger B, Flügge UI (2009) Specific and coordinated control of indolic and aliphatic glucosinolate biosynthesis by R2R3-MYB transcription factors in Arabidopsis thaliana. Phytochem Rev 8:3–13
Gill RJ (1990) The morphology of whiteflies. In: Gerling D (ed) Whiteflies: their bionomics, pest status and management. Intercept, Andover, pp 13–46
Halkier BA, Gershenzon J (2006) Biology and biochemistry of glucosinolates. Annu Rev Plant Biol 57:303–333
Hirai MY, Sugiyama K, Sawada Y, Tohge T, Obayashi T, Suzuki A, Araki R, Sakurai N, Suzuki H, Aoki K, Goda H, Nishizawa OI, Shibata D, Saito K (2007) Omics-based identification of Arabidopsis Myb transcription factors regulating aliphatic glucosinolate biosynthesis. Proc Natl Acad Sci U S A 104:6478–6483
Hraška M, Rakouský S, Čurn V (2008) Tracking of the CaMV-35S promoter performance in GFP transgenic tobacco, with a special emphasis on flowers and reproductive organs, confirmed its predominant activity in vascular tissues. Plant Cell Tissue Organ Cult 94:239–251
Isaacs R, Hardie J, Hick AJ, Pye BJ, Smart LE, Wadhams LJ, Woodcock CM (1993) Behavioural responses of Aphis fabae to isothiocyanates in the laboratory and field. Pestic Sci 39:349–355
Isaacs R, Cahill M, Byrne DN (1999) Host plant evaluation behaviour of Bemisia tabaci and its modification by external or internal uptake of imidacloprid. Physiol Entomol 24:101–108
Jander G, Norris SR, Joshi V, Fraga M, Rugg A, Yu S, Li L, Last RL (2004) Application of a high-throughput HPLC-MS/MS assay to Arabidopsis mutant screening; evidence that threonine aldolase plays a role in seed nutritional quality. Plant J 39:465–475
Janz N (2003) The cost of polyphagy: oviposition decision time vs error rate in a butterfly. Oikos 100:493–496
Jiang Y, Lei H, Collar J, Martin B, Muniz M, Fereres A (1999) Probing and feeding behavior of two distinct biotypes of Bemisia tabaci (Homoptera: Aleyrodidae) on tomato plants. J Econ Entomol 92:357–366
Jing Y, Huang J, Ma R, Han J (2003) Host plant preferences of Bemisia tabaci Gennadius. Entomol Sin 10:109–114
Johnson DD, Walker GP (1999) Intracellular punctures by the adult whitefly Bemisia argentifolii on DC and AC electronic feeding monitors. Entomol Exp Appl 92:257–270
Kawakishi S, Kaneko T (1987) Interaction of proteins with allyl isothiocyanate. J Agric Food Chem 35:85–88
Khasdan V, Levin I, Rosner A, Morin S, Kontsedalov S, Maslenin L, Horowitz AR (2005) DNA markers for identifying biotypes B and Q of Bemisia tabaci (Hemiptera: Aleyrodidae) and studying population dynamics. Bull Entomol Res 95:605–613
Kim JH, Jander G (2007) Myzus persicae (green peach aphid) feeding on Arabidopsis induces the formation of a deterrent indole glucosinolate. Plant J 49:1008–1019
Kim JH, Lee BW, Schroeder FC, Jander G (2008) Identification of indole glucosinolate breakdown products with antifeedant effects on Myzus persicae (green peach aphid). Plant J 54:1015–1026
Koroleva OA, Cramer R (2011) Single-cell proteomic analysis of glucosinolate-rich S-cells in Arabidopsis thaliana. Methods 54:413–423
Koroleva OA, Gibson TM, Cramer R, Stain C (2010) Glucosinolate-accumulating S-cells in Arabidopsis leaves and flower stalks undergo programmed cell death at early stages of differentiation. Plant J 64:456–469
Li X, Baudry J, Berenbaum MR, Schuler MA (2004) Structural and functional divergence of insect CYP6B proteins: from specialist to generalist cytochrome P450. Proc Natl Acad Sci U S A 101:2939–2944
Liu B, Yan F, Chu D, Pan H, Jiao X, Xie W, Wu Q, Wang S, Xu B, Zhou X, Zhang Y (2012) Difference in feeding behaviors of two invasive whiteflies on host plants with different suitability: implication for competitive displacement. Int J Biol Sci 8:697–706
Malitsky S, Blum E, Less H, Venger I, Elbaz M, Morin S, Eshed Y, Aharoni A (2008) The transcript and metabolite networks affected by the two clades of Arabidopsis glucosinolate biosynthesis regulators. Plant Physiol 148:2021–2049
Mellor HE, Anderson M (1995) Antennal sensilla of whiteflies: Trialeurodes vaporariorum (Westwood), the glasshouse whitefly, and Aleyrodes proletella (Linnaeus), the cabbage whitefly, (Homoptera: Aleyrodidae). Part 2: Ultrastructure. Int J Insect Morphol Embryol 24:145–160
Oliveira MRV, Henneberry TJ, Anderson P (2001) History, current status, and collaborative research projects for Bemisia tabaci. Crop Prot 20:709–723
Perring TM (2001) The Bemisia tabaci species complex. Crop Prot 20:725–737
Powel G (1991) Cell membrane punctures during epidermal penetrations by aphids: consequences for the transmission of two potyviruses. Ann Appl Biol 119:313–321
Powell G, Tosh CR, Hardie J (2006) Host plant selection by aphids: behavioral, evolutionary, and applied perspectives. Annu Rev Entomol 51:309–330
Redovniković IR, Textor S, Lisnić B, Gershenzon J (2012) Expression pattern of the glucosinolate side chain biosynthetic genes MAM1 and MAM3 of Arabidopsis thaliana in different organs and developmental stages. Plant Physiol Biochem 53:77–83
Sjolund RD (1997) The phloem sieve element: a river runs through it. Plant Cell 9:1137–1146
Sokal RR, Rohlf FJ (1995) Biometry, 3rd edn. WH Freeman and Co, New York
Sønderby IE, Hansen BG, Bjarnholt N, Ticconi C, Halkier BA, Kliebenstein DJ (2007) A systems biology approach identifies a R2R3 MYB gene subfamily with distinct and overlapping functions in regulation of aliphatic glucosinolates. PLoS One 2:e1322
Sunilkumar G, Mohr L, Lopata-Finch E, Emani C, Rathore KS (2002) Developmental and tissue-specific expression of CaMV 35S promoter in cotton as revealed by GFP. Plant Mol Biol 50:463–474
Textor S, Gershenzon J (2009) Herbivore induction of the glucosinolate–myrosinase defense system: major trends, biochemical bases and ecological significance. Phytochem Rev 8:149–170
Togni PHB, Laumann RA, Medeiros MA, Sujii ER (2010) Odour masking of tomato volatiles by coriander volatiles in host plant selection of Bemisia tabaci biotype B. Entomol Exp Appl 136:164–173
Tosh CR, Krause J, Ruxton GD (2009) Theoretical predictions strongly support decision accuracy as a major driver of ecological specialization. Proc Natl Acad Sci U S A 106:5698–5702
Truernit E, Sauer N (1995) The promoter of the Arabidopsis thaliana SUC2 sucrose-H + symporter gene directs expression of beta-glucuronidase to the phloem: evidence for phloem loading and unloading by SUC2. Planta 196:564–570
Walling LL (2008) Avoiding effective defenses: strategies employed by phloem-feeding insects. Plant Physiol 146:859–866
Winde I, Wittstock U (2011) Insect herbivore counteradaptations to the plant glucosinolate-myrosinase system. Phytochemistry 72:1566–1575
Wittstock U, Halkier BA (2002) Glucosinolate research in the Arabidopsis era. Trends Plant Sci 7:263–270
Acknowledgments
This work was supported by the Israel Science Foundation grant 848/08 and the Binational Agricultural Research and Development Fund (BARD) research grant No. IS-4169-08 R.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table S1
(DOC 31 kb)
Table S2
(DOC 63 kb)
Fig. S1
Specially designed two leaf-cage apparatus. In each replicate of the choice experiments, three adult pairs (male and female) were isolated at the late fourth instar stage and placed in the middle chamber of the specially designed two leaf-cage apparatus. After emergence, adults were allowed to choose between rosette sink leaves (placed in the left and right chambers) of OX-MYB and WT plants. Control (no-choice) treatments used two plants of the same type. In egg to adult development and survival experiments, 12 pairs (male and female) of Bemisia tabaci adults, 3 d old, were released in the middle chamber while two sink rosette leaves of the same plant type (WT, 35S-MYB28, 35S-MYB51, pSUC-MYB28, and pSUC-MYB51) were placed in the left and right chambers. (PPT 591 kb)
Fig. S2
Images of Bemisia tabaci behavior in selected homogeneous (WT/WT and 35S-MYB51/35S-MYB51) or heterogeneous (WT/35S-MYB51) habitats. Cages were prepared using small 35 mm Petri dishes. Two similar-sized holes were made on two sides of the lower and upper covers of the Petri dish to allow the insertion of non-detached leaves (A–B). A plastic film at the size of upper cover was prepared with two cuts of equal size, around 14 × 7 mm each (C). Sink rosette leaves were inserted through the two cuts in the Petri dish and kept between the plastic film and the upper cover (D). This set up prevented the insects from moving to the adaxial side of the leaf and in addition, provided equal leaf areas for the insects. Two different cameras were used to record B. tabaci behavior: a Sony Digital Handycam (DCR-PC 110E PAL) (E) and a Dino-Lite Plus Digital Microscope (AM313T+) (F). The behavior of newly emerged B. tabaci female individuals (in red cicrcle) was recorded for five days (G). (PPT 1631 kb)
Fig. S3
Analysis of AtMYB28/AtMYB51 gene expression by quantitative real-time PCR (qRT-PCR). 2−ΔΔCt data were log- transformed. Dunnett’s post-hoc test was used to compare means of WT plants to those of OX-MYB plants (T-20 for 35S-MYB51, T-9 for 35S-MYB28, T-45 for pSUC-MYB51, and T-29 for pSUC-MYB28). Asterisks indicate significant differences between the means (P ≤ 0.05). Error bars represent standard error of the means (N = 3). (PPT 80 kb)
Fig. S4
Bemisia tabaci behavior in homogenous or heterogeneous habitats. The behavior of newly emerged B. tabaci female individuals was videotaped for five days in selected homogeneous no choice (WT/WT and 35S-MYB51/35S-MYB51) or heterogeneous choice (WT/35S-MYB51) habitats. Each plant genotype is coded with a different color (see legend on figure). Columns with a single color code represent homogenous habitats while dotted columns with two-color codes represent specific heterogeneous habitats (see legend on figure). Genotypes marked in bold in the heterogeneous (Het) habitat indicate which data are presented (WT/35S-MYB51 for WT data and WT/35S-MYB51 for 35S-MYB51 data). (A) Time on the first leaf choice. (B) Mean feeding time (duration on each leaf divided by the number of movements observed during the period) on the first leaf choice. (C) Mean feeding time on the second leaf choice. (D) Mean feeding time during the compete experiment (5 days), excluding the feeding periods on the first and second leaves. Comparisons were made between similar leaf genotypes (WT or 35S-MYB51) in homogeneous and heterogeneous habitats. (#) indicates P-value of 0.09. (PPT 70 kb)
Rights and permissions
About this article
Cite this article
Markovich, O., Kafle, D., Elbaz, M. et al. Arabidopsis thaliana Plants with Different Levels of Aliphatic- and Indolyl-Glucosinolates Affect Host Selection and Performance of Bemisia tabaci . J Chem Ecol 39, 1361–1372 (2013). https://doi.org/10.1007/s10886-013-0358-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-013-0358-0