Abstract
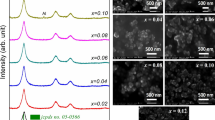
In the present research work, Mg-doped zinc oxide Zn0.1−xMgxO (For x = 0.000, 0.002, 0.006, 0.010) nanoparticles were synthesized by co-precipitation method. The effect of Mg2+ ions substitution on structural, phase transformation, morphological, and optical properties was investigated using X-ray diffraction (XRD), Raman spectroscopy, Field emission scanning electron microscopy (FE-SEM), and UV–Visible spectroscopy (UV–Vis), respectively. The XRD study shows the formation of pure hexagonal wurtzite phase which was also confirmed using Rietveld refinement performed using Fullprof software. The average crystallite size of Mg-doped zinc oxide nanoparticles calculated using Debye–Scherrer relation found to vary from 43.36 to 36.12 nm with increase in Mg concentration from 0 to 10%. The UV–Vis study reveals the blue shift in optical band gap for all the samples as molar concentration of Mg increases in ZnO lattice which was attributed to quantum confinement effect. Further, results of other studies including XRD viz., Raman, UV–Vis, and FE-SEM show that this method of synthesis can be utilized for increasing the optical band gap of the Zn0.1−xMgxO nanocrystals by retaining the pure ZnO phase up to 10% doping of Mg2+ ions concentration.






Similar content being viewed by others
References
M. Bouchenafa, A. Benmakhlouf, M. Sidoumou, A. Bouhemadou, S. Maabed, M. Halit, Y. Al-Douri, Mater Sci Semicond Process 114, 105085 (2020)
S. Touam, R. Belghit, R. Mahdjoubi, Y. Megdoud, H. Meradji, M.S. Khan, Y. Al-Douri, Bull. Mater. Sci. 43(1), 1–11 (2020)
A. Belhachemi, H. Abid, Y. Al-Douri, M. Sehil, A. Bouhemadou, M. Ameri, Chin. J. Phys. 55(3), 1018–1031 (2017)
I. Bziz, E.H. Atmani, N. Fazouan, M. Aazi, Surf. Interfaces 24, 101126 (2021)
S. Benalia, M. Merabet, D. Rached, Y. Al-Douri, B. Abidri, R. Khenata, M. Labair, Mater Sci Semicond Process 31, 493–500 (2015)
M. Ameri, F. Mired, I. Ameri, Y. Al-Douri, Mater. Express 4(6), 521–532 (2014)
R.M. Hewlett, M.A. McLachlan, Adv. Mater. 28, 3893–3921 (2016)
A.K. Zak, W.A. Majid, M.R. Mahmoudian, M. Darroudi, R. Yousefi, Adv Powder Technol. 24, 618–624 (2013)
S. Singh, P. Thiyagarajan, K.M. Kant, D. Anita, S. Thirupathiah, N. Rama, M.R. Rao, J. Phys. D: Appl. Phys. 40, 6312 (2007)
O. Mangla, S. Roy, Solid State Phenom. 287, 75–79 (2019)
B.G. Shohany, L. Motevalizadeh, M.E.J. Abrishami, Theor. Appl. Phys. 12, 219–225 (2018)
R. Zha, R. Nadimicherla, X.J. Guo, Mater. Chem. A 3(12), 6565–6574 (2015)
X.Y. Yan, C.B. Yao, J. Li, J.Y. Hu, Q.H. Li, S.B. Yang, Opt. Mater. 55, 73–77 (2016)
R. Al-Gaashani, S. Radiman, Y. Al-Douri, N. Tabet, A.R. Daud, J. Alloys Compd. 521, 71–76 (2012)
Y. Zhao, M. Zhou, Z. Li, Z. Lv, X. Liang, J. Min, W. Shi, J. Lumin. 131, 1900–1903 (2011)
Y. Ammaih, A. Lfakir, B. Hartiti, A. Ridah, P. Thevenin, M. Siadat, Opt. Quant. Electron. 46(1), 229–234 (2014)
S. Ayaz, P. Mishra, S. Sen, J. Appl. Phys. 126(2), 024302 (2019)
G. Zheng, P. Zhu, L. Sun, J. Jiang, J. Liu, X. Wang, W. Li, AIP Adv. 6(12), 125306 (2016)
N. Fathima, N. Pradeep, J. Balakrishnan, Sol. Energy Mater Sol. Cells 194, 207–214 (2019)
C.F. Klingshirn, Semiconductor Optics (Springer Science & Business Media, New York, 2012)
M. Ghaffari, F. Moztarzadeh, M. Safavi, Ceram. Int. 45(1), 1179–1188 (2019)
A. Saaedi, R. Yousefi, F.A. Sheini, M. Cheraghizade, A.K. Zak, N.M. Huang, Ceram. Int. 40, 4327 (2014)
A.S. Hameed, C. Karthikeyan, S. Sasikumar, V.S. Kumar, S. Kumaresan, R.J. Ganesan, Mater. Chem. B 1, 5950–5962 (2013)
M.S. Rad, A. Kompany, A.K. Zak, M.E. Abrishami, Mod. Phys. Lett. B 29, 1450251 (2015)
R. Yousefi, A.K. Zak, M.R. Mahmoudian, J. Solid State Chem. 184, 2678 (2011)
A.B. Lavand, Y.S. Malghe, Int. J. Photochem. 2015, 1–9 (2015)
N. Badi, Y. Al-Douri, S. Khasim, Opt. Mater. 89, 554–558 (2019)
F. Bigdeli, A. Morsali, Mater. Lett. 64, 4–5 (2010)
S. Vyas, M. Johnson, Technol. Rev. 64, 2 (2020)
W.B. Russel, W.B. Russel, D.A. Saville, W.R. Schowalter, Colloidal Dispersions (Cambridge University Press, Cambridge, 1991)
M. Saeidi, M. Abrari, M. Ahmadi, Appl. Phys. A 125, 1–9 (2019)
C.L. Kuo, C.L. Wang, H.H. Ko, W.S. Hwang, K.M. Chang, W.L. Li, M.C. Wang, Ceram. Int. 36, 693 (2010)
H.R. Ghorbani, F.P. Mehr, H. Pazoki, B.M. Rahmani, Orient. J. Chem. 31(2), 1219–1221 (2015)
S. Sarkar, E. Guibal, F. Quignard, A.K. SenGupta, J. Nanopart. Res. 14(2), 1–24 (2012)
D.B. Bharti, A.V. Bharati, Luminious 32(3), 317–320 (2017)
M. Zare, K. Namratha, K. Byrappa, D.M. Surendra, S. Yallappa, B.J. Hungund, Mater. Sci. Technol. 34(6), 1035–1043 (2018)
R. Razali, A.K. Zak, W.A. Majid, M. Darroudi, Ceram. Int. 37, 3657 (2011)
K. Handore, S. Bhavsar, A. Horne, P. Chhattise, K. Mohite, J. Ambekar, V.J. Chabukswar, Macromol. Sci. A 51(12), 941–947 (2014)
S.D. Lee, S.H. Nam, M.H. Kim, J.H. Boo, Phys. Procedia 32, 320 (2012)
Y. Al-Douri, Colloidal Metal Oxide Nanoparticles (Elsevier, Amsterdam, 2020), pp. 25–38
T.D. Nguyen, Q.D. Nguyen, T.T. Nguyen, Adv. Nat. Sci. 5, 035011 (2014)
R. Zamiri, A. Zakaria, H.A. Ahangar, M. Darroudi, A.K. Zak, G.P.J. Drummen, Alloys Compd. 516, 41 (2012)
T.P. Yadav, R.M. Yadav, D.P. Singh, Nanosci. Nanotechnol. 2, 22 (2012)
C.B. Ong, L.Y. Ng, A.W. Mohammad, Renew. Sustain. Energy Rev. 81, 536–551 (2018)
S. Ahmed, S.A. Chaudhry, S.J. Ikram, Photochem. Photobiol. 166, 272–284 (2017)
V.K. Sharma, R.A. Yngard, Y. Lin, Adv. Colloid Interface Sci. 145, 8396 (2009)
A.K. Zak, M.E. Abrishami, W.A. Majid, R. Yousefi, S.M. Hosseini, Ceram. Int. 37, 393–398 (2011)
A. Samanta, M.N. Goswami, P.K. Mahapatra, Mater. Sci. Eng. B 245, 1–8 (2019)
Y. Xiao, L. Li, Y. Li, M. Fang, L. Zhang, Nanotechnology 16, 671 (2005)
R. Sagheer, M. Khalil, V. Abbas, Z.N. Kayani, U. Tariq, F. Ashraf, Optik 200, 163428 (2020)
S. Fabbiyola, L.J. Kennedy, A.A. Dakhel, M. Bououdina, J.J. Vijaya, T. Ratnaji, J. Mol. Struct. 1109, 89–96 (2016)
H. Jeon, Y.J. Min, S.H. Ahn, S.M. Hong, J.S. Shin, J.H. Kim, K.B. Lee, Colloids Surf. A. Physicochem. Eng. Asp. 414, 75 (2012)
A. Samanta, M.N. Goswami, P.K. Mahapatra, J. Mater. Sci. Mater. Electron. 27, 12271 (2016)
B.D. Cullity, Elements of X-Ray Diffraction (Addison-Wesley Publishing, New York, 1956)
J. Iqbal, T. Jan, M. Ismail, N. Ahmad, A. Arif, M. Khan, A. Arshad, Ceram. Int. 40, 7487–7493 (2014)
M.N.H. Mia, M.F. Pervez, M.K. Hossain, M.R. Rahman, M.J. Uddin, M.A. Al Mashud, M. Hoq, Results Phys. 7, 2683–2691 (2017)
V. Etacheri, R. Roshan, V. Kumar, Appl. Mater. Interfaces 4(5), 2717–2725 (2012)
A.F. Jaramillo, R. Baez-Cruz, L.F. Montoya, C. Medinam, E. Pérez-Tijerina, F. Salazar, M.F. Melendrez, Ceram. Int. 43(15), 11838–11847 (2017)
A. Sahoo, M. Miryala, T. Dixit, A. Klimkowicz, B. Francis, M. Murakami, S. Krishnan, Nanomaterials 10(7), 1326 (2020)
V. Russo, M. Ghidelli, P. Gondoni, C.S. Casari, A. Li Bassi, J. Appl. Phys. 115, 073508 (2014)
Z.Y. Jiang, K.R. Zhu, Z.Q. Lin, S.W. Jin, G. Li, Rare Met. 37, 881–885 (2018)
P. Kumar, J.P. Singh, Y. Kumar, A. Gaur, H.K. Malik, K. Asokan, Curr. App. Phys. 12, 1166–1172 (2012)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Azad, S., Kumar, N. & Chand, S. Effect of Mg2+ ions substitution on phase formation, structural, morphological, and optical properties of Zn0.1−xMgxO structure. J Mater Sci: Mater Electron 33, 861–870 (2022). https://doi.org/10.1007/s10854-021-07356-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-021-07356-6