Abstract
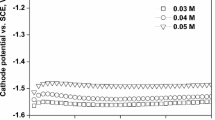
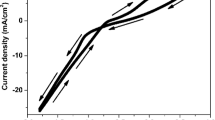
Nanocrystalline CoFe thin films were electrodeposited from baths containing sodium citrate as complexing agent. Cyclic voltammogrames of CoFe baths showed that addition of sodium citrate to the electrolytes shifted reduction potential of metals toward more negative values. X-ray diffraction patterns of CoFe thin films deposited at different current densities illustrated a transition from FCC(Co) phase to FCC(Co) + BCC(Fe) phases with increasing applied current density. Estimation of average grain size (D) of CoFe thin films by Scherrer’s equation showed all coatings had nanocrystalline structures. The accuracy of results obtained by Scherrer’s equation was confirmed by transmutation electron microscope images. Study of magnetic properties by vibrating sample magnetometer indicated that reduction in grain size of CoFe films resulted in noticeable decrease in coercivity, according to “D6” law. Moreover, decreasing grain size in CoFe thin films led to reduction in resistivity which could be attributed to scattering of conduction electrons, according to “Scattering Hypotheses”. However, average grain size of nanocrystalline CoFe films had no effect on the saturation magnetization which is mostly controlled by chemical composition. The results showed that increasing iron content in the deposited CoFe films from 17 to 31 at.% caused enhancement of saturation magnetization.











Similar content being viewed by others
References
B. Crozier, Q. Liu, D.G. Ivey, J. Mater. Sci. Mater. Electron. 22, 614–625 (2011)
M. Salehi, P. Marashi, M. Salehi, R. Ghannad, J. Ultrafine Grained Nanostruct. Mater. 47, 27–35 (2014)
A. Ghasemi, A.M. Davarpanah, M. Ghadiri, Int. J. Nanosci. Nanotechnol. 4, 207–214 (2012)
E.I. Cooper, C. Bonhote, J. Heidmann, Y. Hsu, P. Kern, J.W. Lam, M. Ramasubramanian, N. Robertson, L.T. Romankiw, H. Xu, IBM J. Res. Dev. 49, 103–126 (2005)
X. Zhang, S. Wang, J. Zhou, J. Li, D. Jiao, X. Kou, J. Alloys Compd. 474, 273–278 (2009)
W. Wanga, G.H. Yuea, Y. Chena, W.B. Mib, H.L. Baib, D.L. Peng, J. Alloys Compd. 475, 440–445 (2009)
R.H. Yu, S. Basu, L. Ren, Y. Zhang, A. Parvizi-Majidi, K.M. Unruh, J.Q. Xiao, IEEE Trans. Magn. 36, 3388–3393 (2000)
Z. Jamili-Shirvan, M. Haddad-Sabzevar, J. Ultrafine Grained Nanostruct. Mater. 46, 55–59 (2013)
S. Mehrizi, M. Heydarzadeh Sohi, S.A. Seyyed Ebrahimi, Surf. Coat. Technol. 205, 4757–4763 (2011)
G. Herzer, IEEE Trans. Magn. 26, 1397–1402 (1990)
Y. Zhang, D.G. Ivey, Mater. Sci. Eng. B 140, 15–22 (2007)
M. Raghasudha, D. Ravinder, P. Veerasomaia, J. Nanostruct. Chem. 3, 63–68 (2013)
M. Nazari, N. Ghasemi, H. Maddah, M. Motlagh, Nanostruct. Chem. 4, 99–103 (2014)
B. Mueller, ChemEQL, A Program to Calculate Chemical Speciation, Version 3.0. Limnological Research Center EAWAG/ETH, CH-6047 Kastanienbaum, Switzerland (1996)
A.E Martell, R.M. Smith, Critical Stability Constants, vols. 1, 6 (Plenum Press, London, 1974, 1989)
A.E. Martell, R.M. Smith, R.J. Motekaitis Critical Stability Constants Database Version 6.0. NIST (Texas A&M University, College Station, 2001)
A. Brenner, Electrodeposition of Alloys (Academic Press, New York, 1963)
H. Bakar, ASM Handbook, Alloy Phase Diagrams, vol. 3 (ASM International, Materials Park, 1992)
P. Wissmann, H. Ulrich Finzel, Electrical Resistivity of Thin Metal Films (Springer, Berlin, 2007)
S. Mehrizi, M. Heydarzadeh Sohi, E. Shafahian, A.A. Khangholi, J. Mater. Sci. Mater. Electron. 23, 1174–1396 (2012)
Acknowledgments
The authors would like to thank University of Tehran and Iranian nanotechnology initiative council for financial support of this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mehrizi, S., Heydarzadeh Sohi, M. Electrical resistivity and magnetic properties of electrodeposited nanocrystalline CoFe thin films. J Mater Sci: Mater Electron 26, 7381–7389 (2015). https://doi.org/10.1007/s10854-015-3368-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-015-3368-6