Abstract
The cultivation of peat mosses (‘Sphagnum farming’) is a new wet and climate-friendly agricultural use of degraded bog sites. However, it is largely unknown to what extent these surrogate habitats are used by bog fauna. This study investigated the potential of Sphagnum cultivation sites as surrogate habitats for beetles and evaluated the relationship between the vegetation structure and the occurrence of beetle species. In 2017 and 2018, comparative surveys were carried out at different sub-areas of two Sphagnum cultivation sites and one near-natural bog. Beetles living in and on the upper Sphagnum layer were studied by manually extracting quadrat samples. Vegetation surveys were conducted in the same quadrats to analyze the occurrence of beetle species based on vegetation parameters. We collected 926 individuals of beetles belonging to 89 species out of 17 families. At the different sections of the cultivation sites, 8–16 bog-typical beetle species were found, while at the sub-areas of the near-natural bog, there were 15 and 19 bog-typical species, respectively. The statistical analyses showed that vegetation structure influenced the numbers of beetle species and individuals at both the cultivation and near-natural sites.
Implications for insect conservation
A dense and high Sphagnum carpet is a fundamental habitat structure for the beetle fauna of cultivation sites, while vascular plants initially help to create habitat structures on bare peat. Conversely, if the vascular plants overgrow and shade the Sphagnum, this can have a negative effect on bog-typical beetle species, as shown at the near-natural site.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The fauna of peat bogs consists of highly specialized species that have adapted to the extreme site conditions of these unique habitats (Rydin and Jeglum 2006; Spitzer and Danks 2006). In the case of beetles, as well as for other insects, there are particularly close connections at the microhabitat level with the vegetation structure and the plant species present (Görn et al. 2014; Hollmen et al. 2008; Powell et al. 2013; Sushko 2017; Tobisch et al. 2023). These connections are partly direct when species feed on plant material or their larvae develop in them (e.g., Byrrhidae, Chrysomelidae, Curculionidae) (Ries et al. 2021; Sushko 2017). But they are also partly indirect since the vegetation influences the microclimatic conditions, which in turn affects, for example, ground beetles (Brigić et al. 2017; Saunders et al. 1991; Spitzer et al. 1999). If these site conditions change, the specialized bog species lose not only their habitat along with the corresponding habitat structures but also their competitive advantage over less specialized and, therefore, more competitive species (Elo et al. 2015; Hollmen et al. 2008; van Kleef et al. 2012).
Species specialized in bogs have experienced a drastic qualitative and quantitative habitat loss in Central Europe, especially in the nineteenth and twentieth centuries (Hughes et al. 2008; Joosten et al. 2017b). In Germany, large-scale drainage as well as land-use modification for agricultural and forestry cultivation, nutrient inputs, and industrial peat extraction, among other causes, have resulted in more than 95% of peatlands being severely degraded or destroyed (Succow and Joosten 2001; Trepel et al. 2017). Near-natural growing bogs with intact vegetation structures that meet the unique bog fauna's habitat requirements exist only to a minimal extent (Joosten and Couwenberg 2001). Thus, there is an increasing decline in the distribution and relative abundance of specialized bog species (Habel et al. 2019; Sperle and Bruelheide 2021; van Grunsven et al. 2020; Vries and Boer 1990). It can be assumed that the consequences of climate change will further exacerbate the existing threats to the species and habitat types of peatlands (Thomas et al. 2004).
Since the end of the twentieth century, increasing efforts have been made to counteract these adverse developments and to restore degraded peatlands (Tanneberger et al. 2017). However, large restoration projects primarily focus on areas after peat extraction (Schmatzler 2015), whereas most peat soils in Europe continue to be found in agricultural and forestry use (Joosten et al. 2017a; Schmatzler 2015). To stop the high greenhouse gas emissions from drainage-based use of peatlands, the water table on these lands needs to be raised to the soil surface and thus to peat preserving conditions (Tanneberger et al. 2021; Tiemeyer et al. 2020; Wilson et al. 2016). For this reason, paludiculture is being tested as a wet and climate-friendly form of agricultural use of peatlands (Wichtmann et al. 2016). On bog soils, the cultivation of peat mosses ('Sphagnum farming') has emerged as a promising, sustainable form of land use (Gaudig et al. 2018). Crop production trials show that Sphagnum biomass is suitable as a constituent for horticultural growing media and is a sustainable alternative to peat due to similar physical and chemical properties (Jobin et al. 2014; Müller and Glatzel 2021).
It can be assumed that the large-scale establishment of Sphagnum combined with permanent water control at the cultivation sites creates a potential habitat for bog species. However, only a few pilot projects exist for Sphagnum farming worldwide, mainly in Germany and Canada (Gaudig et al. 2018; Grobe et al. 2021; Pouliot et al. 2015). Due to this, the extent to which these substitute habitats are used by bog fauna is still largely unknown, apart from single studies on the species groups of spiders (Muster et al. 2015, 2020), birds, amphibians, dragonflies, and butterflies (Zoch and Reich 2020, 2022). So far, the only study known on beetles was done by Mainda (2021), who studied Carabidae, Dytiscidae, and Staphylinidae on a cultivation site in northwestern Germany and found a few characteristic bog species out of all three families. In addition, more knowledge about epigeic arthropod assemblages or microhabitat preferences is still needed, even for intact natural sites, because fauna is recorded much less frequently than flora (Kato et al. 2009).
The aim of our study was:
-
to assess the potential of Sphagnum cultivation sites as habitats for beetles
-
and to evaluate the relationship between the vegetation structure and the occurrence of beetle species at Sphagnum cultivation sites and near-natural sites.
We focused on all beetles living in the ground vegetation layer since habitat specialists can be found in many different beetle families, such as Byrrhidae, Carabidae, Chrysomelidae, Hydrophilidae, or Staphylinidae (Koch 1989; Peus 1932; Sprick et al. 2013). Due to their diverse habitat requirements and functional traits, they show different connections to the vegetation and provide information about the prevailing environmental conditions (Bettacchioli et al. 2012; Bohac 1999; Ries et al. 2021).
Material and methods
Study sites
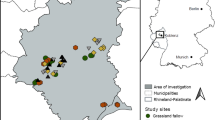
The survey of beetles and vegetation structure was conducted at two Sphagnum cultivation sites (‘Provinzialmoor’, 52°40′ N, 07°06′ E and ‘Drenth’, 52°41′ N, 07°05′ E) and a near-natural peat bog (‘Meerkolk’, 52° 38′ N, 07° 08′ E). All sites are located in northwestern Germany, near the Dutch-German border (Fig. 1). The region was originally covered by extensive peatlands, which are now degraded mainly by agricultural use and peat extraction.
Within the two Sphagnum cultivation sites, we compared two different treatments of rewetting. Both sites were established from 2015 to 2016 on cutover bog after milled peat extraction (Graf et al. 2017; Grobe et al. 2021). The ‘Provinzialmoor’ site was already rewetted in 2008 as a shallow polder. To establish Sphagnum cultivation, the water table of the inundated polder was lowered to make the site accessible. The site was provided with open ditches for irrigation in the summer months and drainage of excess water in the winter months. The water was let in manually from the surrounding polders with pipes. The pH (mean ± SE) of the irrigation water was 4.2 ± 0.03. In contrast, the ‘Drenth’ cultivation site was established directly after the termination of peat extraction in 2015 without any previous re-wetting. During the study, the area was still surrounded by active peat extraction. Seven polders separated by peat bunds on the cultivation site ‘Drenth’ were formed and equipped for irrigation and drainage either with open ditches or subsurface drainage pipes combined with above-ground drip tubes. The water required for irrigation at Drenth was pumped in from two ponds, which could be refilled with groundwater if rainwater ran out in the summer months. This led to higher values of pH 5.5 ± 0.15 of the irrigation water at Drenth.
During the establishment, Sphagnum fragments were spread manually on the bare peat surface following the moss-layer-transfer technique (Quinty and Rochefort 2003). The cultivation sites were mainly inoculated with the peat moss species Sphagnum papillosum and S. palustre. Both are characteristic of natural peatlands in the region and are suitable as raw material for growing media (Gaudig et al. 2018; Hölzer 2010). Biomass of both Sphagnum species was gathered at different near-natural donor sites in adjacent regions in Germany and the Netherlands. With the donor material of the target species, various other mosses and vascular plants typical of bogs were unintentionally introduced to the sites (Grobe 2023; Grobe and Rode 2023). The most frequent vascular plants at both cultivation sites were Eriophorum angustifolium, Erica tetralix, Molinia caerulea, and Rhynchospora alba (Grobe and Rode 2023). During the establishment, the Sphagnum fragments were covered with straw mulch or geotextile to improve the microclimate for the Sphagnum in the initial phase. The cultivation sites were mown 1–2 times per year to reduce competition from vascular plants. Until the end of this study, no Sphagnum had been harvested on the cultivation sites. We established four study sites at the cultivation sites that differ in age (inoculation time) and donor sites (Sphagnum biomass) (Table 1).
We compared the cultivation sites with a near-natural bog, the 'Meerkolk'. It is a nature reserve of 31 ha at a distance of 4 and 7 km from the cultivation sites, respectively. A quaking bog with floating Sphagnum mats and typical bog vegetation characterizes the center of the area. In October 2016, Sphagnum biomass was collected from a part of the site to inoculate the ‘Provinzialmoor’ cultivation site. The collection of Sphagnum fragments (5–10 cm length) was done manually, cutting only half of the individual hummocks to limit the damage to the vegetation. Samples were taken at the donor site ‘Meerkolk’ (nat-MD) as well as at a reference site (nat-MR), which was not affected by the collection of Sphagnum biomass (Table 1).
Sampling, determination, and classification
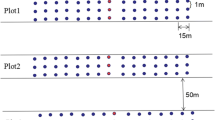
In 2017 and 2018, beetles living in and on the upper Sphagnum layer were studied by manually extracting a total of 100 quadrat samples per study site. Quadrat samples allow conclusions about the relationship between vegetation structure and the occurrence of beetles, as they show absolute densities of individuals (Andersen 1995; Coulson and Butterfield 1985). In addition, less inactive species primarily residing among mosses can be captured better than with the commonly used pitfall traps (Coulson and Butterfield 1985).
Sampling was conducted on five dates each year from April to October. Ten samples were taken on each date and randomly distributed at each study site. Each sample included a vegetation survey and the manual collection of beetles in the same sample frame (30 cm × 30 cm). First, during the vegetation survey, the following parameters were recorded: moss height, vascular plant height, moss cover, vascular plant cover, litter, bare peat, and open water. When present, the height of Sphagnum mosses was measured with a yardstick at five points (four corners and middle) in each frame and then averaged. For the vascular plants, the maximum height was measured. In addition, the cover of Sphagnum mosses, vascular plants, litter, bare peat, and water area was estimated according to a modified scale of Braun-Blanquet (1964) (scale: < 1, 1–5, 5–25, 25–50, 50–75, 75–95, 95–100%). The mean values of the scale classes were used for analysis. For the hand collection of beetles, the top layer of living Sphagnum (maximum 5 cm) and other vegetation or litter was cut off in the sample frame and examined in a white tray. All beetles from all families were collected from the material for identification in the laboratory. The cut vegetation was placed back so the Sphagnum could continue growing.
Beetles were identified according to Lompe (2002). Nomenclature follows Gruttke et al. (2016) and Ries et al. (2021). The beetles were determined to the species level. Critical species were confirmed by various experts (see Acknowledgements). However, 120 individuals were determined only to genera (mainly Euaesthetus and Stenus) or subfamily (Aleocharinae). These individuals were indicated with spec. or agg. and the genera or subfamily count as one additional species in the total number of species. For all determined species, a binary classification as “threatened” (categories 1, 2, 3, G, V on the German Red List, see Appendix for abbreviations) or “not threatened” (categories *, D, n.b.) (Gruttke et al. 2016; Ries et al. 2021) and as “bog-typical” or “bog-tolerant” species was made. Those species were evaluated as bog-typical, which are considered tyrphobiont or tyrphophilous according to Koch (1989). Additionally, Carabid species were included as bog-typical, which have a regional distribution focus in bogs in northwestern Germany according to Bräunicke and Trautner (2009). All other species were classified as bog-tolerant. For species not appearing in Koch (1989), classification was based on habitat information in Lompe (2002).
Data analysis
All statistical analyses were performed using R software (R version 4.1.1, R Core Team 2022). First, the vegetation parameters were tested for collinearity using the variance inflation factor (VIF). The function vifcor from the package "usdm" (Naimi et al. 2014) was used and, as recommended by Dormann et al. (2013), pairs of variables with a correlation coefficient greater than 0.7 were first searched for and, if found, the variable from the pair with the higher VIF was removed. The VIF was then calculated for all remaining variables. All pairs of the seven parameters of interest (moss height, vascular plant height, moss cover, vascular plant cover, litter, bare peat, open water) had a correlation coefficient lower than 0.7, and none of the variables showed a severe collinearity problem (VIFs < 3.8). Therefore, all variables were included in further analyses.
Generalized linear mixed models (GLMM, with negative binomial error distribution) were used to investigate the influence of vegetation parameters on beetle occurrence. The experimental design encompasses three primary grouping variables: ‘study site’, ‘year’, and ‘date’. Interactions between these grouping variables resulted in additional hierarchical grouping structures. Firstly, the interaction between ‘study site’ and ‘year’ resulted in a grouping variable with twelve levels, allowing us to consider variance between study sites in the same year. Secondly, the interaction between ‘study sites’, ‘year’, and ‘date’ resulted in a grouping variable with 60 levels, allowing us to account for the variance between the study sites in the same year and on the same date. This led to five grouping variables that were included in the model. As the grouping variables ‘study site’ and ‘year’ have only a few levels, they were specified as fixed effects, as recommended by Crawley (2002) and Piepho et al. (2003). The remaining three grouping variables were specified as random effects. All seven vegetation parameters were included as fixed effects.
The near-natural and the cultivation sites were analyzed separately since different primary site conditions prevailed. This is mainly due to the naturally dense moss carpet at the near-natural compared to the only 1–3 years old initial stages at the cultivation sites. Models were run for the total number of species and individuals, and in addition, models were run for the individual number of bog-typical and threatened species. No meaningful estimates could be obtained for the species number of bog-typical and threatened species as the models did not converge.
The package ‘glmmTMB’ (Brooks et al. 2017) was used to model the GLMMs. Based on the fitted models, an analysis of deviance was performed to test the significance of the parameters.
To interpret and summarize major patterns of variation in vegetation structure between sites, principal component analysis (PCA) was performed after standardization using spectral decomposition with the packages ‘ggrepel’ (Slowikowski 2021) and ‘factoextra’ (Kassambara and Mundt 2020).
Results
Numbers of species and individuals
In total, we collected 926 individuals of beetles belonging to 89 species out of 17 families (Appendix). Of these, 30 beetle species were classified as bog-typical and 15 as threatened. Whereby 13 of the threatened species were also classified as bog-typical.
At the cultivation sites, most species and individuals were found at cult-PA in terms of the total numbers and the mean values per sample (Table 2). The same applies to bog-typical and threatened species, with 16 and seven species, respectively, at cult-PA. Fewer individuals and species were found at the other three sites (cult-D, cult-PB, cult-PC), with little differences. There were 8–11 bog-typical and 2–3 threatened species. As the only difference, more individuals overall and of bog-typical species were found at cult-PB than at cult-PC and cult-D. A similar number of species and even more individuals were found at the near-natural bog compared to cult-PA. Here, 15 and 19 bog-typical species and six and eight threatened species, respectively, were recorded. The two sub-areas of the near-natural bog show no apparent differences. While species numbers were slightly higher at nat-MD, marginally more individuals were found at nat-MR.
Species composition
At the cultivation sites, Staphylinidae, Carabidae, and Hydrophilidae were most abundant, while at the near-natural bog, the families of Staphylinidae, Dytiscidae, Hydrophilidae, and Scirtidae were dominant (Table 5 in Appendix). The most abundant species, in general, were Myllaena intermedia (204 individuals), Euaesthetus spec. (73 individuals) and Cyphon variabilis (61 individuals). At the near-natural bog, the bog-typical species with the largest number of individuals were Cyphon variabilis, C. hilaris, Hydroporus scalesianus and Acylophorus wagenschieberi. At the same time, these species occurred only with single individuals at the cultivation sites, distinguishing the near-natural bog from the cultivation sites. In contrast to the other families, more species and individuals of Carabidae were present at the cultivation sites than at the near-natural bog. Nevertheless, the bog-typical species most abundant at the cultivation sites (e.g., Pterostichus diligens, Erichsonius cinerascens) were also found with some individuals at the near-natural bog. Cult-D and cult-PA are distinguished from the other sites by the ground beetle species Acupalpus dubius, Bradycellus harpalinus and Stenolophus mixtus. Cult-PA stands out for the only records of Trichocellus cognatus and the rove beetles Tachyporus transversalis and Myllaena kraatzi. As the only pill beetle species, Cytilus sericeus was found only at cult-PB and cult-PC.
Vegetation structure
At the near-natural bog, vegetation was primarily characterized by a thick and dense Sphagnum carpet. However, at nat-MD this was thinner due to Sphagnum collection, with a mean of 6.7 cm, than at nat-MR, which had a height of 9.4 cm (Table 3). Mean Sphagnum carpet height was lower at all cultivation sites than at the near-natural bog, averaging 2.8 cm at cult-PA and 1.6–1.9 cm at the other sites. Mean Sphagnum cover was also higher at the near-natural bog, averaging around 97%, than at the cultivation sites, ranging from 56% (cult-D) to 83% (cult-PA). No clear differences were found among samples for vascular plants. Height averaged 11–19 cm and covered 13–33% at all sites. Litter was most abundant at the two younger cultivation sites, cult-PB and cult-PC, with 21–23%, and least much at the near-natural bog, with 1–2%. There were no bare peat areas at the near-natural bog. At the cultivation sites, the largest proportion of bare peat was found at cult-D, with an average of 25%, and the lowest at cult-PA, with an average of 5%. Smaller open water areas occurred occasionally at all sites, although the variation in extent was greater at the cultivation sites.
The two dimensions of the PCA could explain 64% of the vegetation structure data (Fig. 2). Dimension 1 (horizontal axis) captured 47.3% of the variance, showing a gradient from a structure characterized by bare peat or litter to a structure characterized by tall and dense vegetation (Sphagnum mosses and vascular plants). The slight angles of the vectors reveal a close correlation between Sphagnum height and cover and vascular plant height and cover. For dimension 2 (vertical axis), describing a supplemental 16.7% variance, the proportion of open water area had greater weight, and the differences between vegetation were more characterized by vascular plants or Sphagnum.
Principal Component Analysis (PCA) Biplot of the vegetation structure in n = 100 samples at each study site. Each dot represents one quadrat sample at which the vegetation structure was assessed. See Table 1 for abbreviations of study sites
The PCA shows that the cultivation sites are relatively similar in the measured parameters. The two sub-areas of the near-natural bog overlap with the other sites. Still, it becomes apparent here that the bare peat and litter cover at the near-natural bog is lower than at the cultivation sites, and the height and the cover of Sphagnum mosses are somewhat higher on average, while the vascular plants show lower cover and height. In addition, the relative concentration of plots of the near-natural bog in the PCA shows an overall homogeneity of vegetation. In contrast, the plots of the cultivation sites are much more scattered.
Impact of vegetation structure on beetle occurrence
The statistical analyses showed that vegetation structure influenced the numbers of beetle species and individuals at both the cultivation and near-natural sites. At the cultivation sites, moss height in particular was found to be an important parameter. Therefore, there was a significant positive influence on the total number of species and individuals as well as on the number of individuals of bog-typical species (Table 4). Thus, the number of beetle species per unit moss height showed an average increase of 1.16, 95% CI [1.06, 1.27], the number of all individuals increased by 1.24, 95% CI [1.10, 1.42], and the number of bog-typical individuals increased by 1.26, 95% CI [1.01, 1.57]. The latter was also positively influenced by the height of vascular plants (on average, 1.03 times, 95% CI [1.00, 1.07], as many bog-typical individuals per unit increase). In contrast, a negative effect was found with an increased proportion of bare peat on the number of species and individuals of all species. For each percent increase in bare peat cover, the number of species decreased by an average factor of 0.99, 95% CI [0.98, 1.00], and the number of individuals by 0.98, 95% CI [0.97, 1.00].
The height of vascular plants was determined as a significant factor at the near-natural bog, which was analyzed separately due to the widely differing site conditions. Thereby, in the samples, there were on average 0.94 times, 95% CI [0.90, 0.98], fewer individuals of bog-typical species and 0.87 times, 95% CI [0.81, 0.94], fewer individuals of threatened species with each unit that the height of vascular plants increased. A similar trend, although not significant, was seen in the total number of species. This decreased by a factor of 0.98, 95% CI [0.96, 1.00], on average, when vascular plant height increased by one unit (p = 0.0774). In addition, there was a tendency for the year of the collection also to affect the total species number at the near-natural bog. Thus, 2018 tended to have, on average, 0.593 times, 95% CI [0.33, 1.05] fewer species count than 2017 (p = 0.0725).
Discussion
Influence of vegetation structure
In this study, we showed that the influence of vegetation structure on the occurrence of beetles differed between the Sphagnum cultivation sites and the near-natural bog. The studied cultivation sites were still in the initial stage, so no dense moss layer and large bare peat areas existed. At the cultivation sites, it was determined that the abundance of beetles, particularly of bog-typical species, was promoted with the increasing height of Sphagnum and vascular plants. This is consistent with other studies that found vegetation height a key factor for beetle occurrences (Dennis et al. 2004; Görn et al. 2014; Hoffmann et al. 2016). The close relationship between height and cover, both of vascular plants and Sphagnum mosses (Fig. 2), suggests that increasing cover also positively affects beetle occurrence. This was confirmed by studies of beetle assemblages on large intact peat bogs (Sushko 2019a). Accordingly, the influence of bare peat was negative on the total number of beetle species and individuals. Muster et al. (2020) also found a positive effect of high Sphagnum cover on epigeic arthropods at Sphagnum cultivation sites. We found no relationship between moss height and cover with the beetle occurrence at the near-natural bog. This could be related to the fact that the dense Sphagnum carpet meant that the values of the mosses varied little. Different from the cultivation sites, increasing heights of vascular plants even had a negative effect on the numbers of individuals of bog-typical and threatened beetle species at the near-natural bog. Therefore, the influence of vegetation can be expected to change as succession increases at the cultivation sites. Initially, vegetation structures dominated by vascular plants provide better conditions for bog-typical species than bare peat, but with a dense moss layer, vascular plants can sometimes have adverse effects. Sushko (2019b) also found that beetle diversity was highest in the mid-successional stages of bog vegetation. One explanation of the disadvantages of older stages is that high vascular plants lead to the shading of the moss layer, which changes the microclimatic conditions (Matlack 1993).
The vegetation structure at the sites partly explains the differences in the composition of the beetle fauna (number of species and individuals per sample). The statistical analysis showed that the greatest influencing factor was moss height. At cult-PA, Sphagnum mosses were on average 1 cm higher than at the other cultivation sites. Thus, the most bog-typical and threatened species were also found here. At cult-D, on the other hand, open peat was dominant, which was reflected in lower species and individual numbers and thus also corresponds to the results of the GLMM. There were more mosses and correspondingly more beetles at the near-natural sites.
Collecting Sphagnum at the donor site resulted in a clear difference in moss height between nat-MR and nat-MD, and there was a trend toward fewer individuals at the donor site. It can be assumed that a reduction in moss height can have a negative effect. However, this effect is limited by the rapid regeneration of mosses and will hardly be of any significance in the medium to long term (Sanders and Winterbourn 1993). The influence of maintenance activities, such as the mowing of vascular plants and Sphagnum harvesting at the cultivation sites, was not studied. However, based on the results, it can be assumed that removing Sphagnum mosses has a temporary negative impact on habitat structures. In contrast, the mowing of vascular plants can have a different effect depending on the succession stage, as mentioned before. In addition, it is important to note that mechanical harvesting and biomass removal can have lethal effects on different developmental stages and weaken populations (Humbert et al. 2009). To better evaluate this aspect, further research is needed on the impact of Sphagnum harvesting at cultivation sites and Sphagnum collection at donor sites.
Even though the cultivation sites were more heterogeneous in the initial phase than the near-natural sites, this will change over time due to constant maintenance. Sphagnum farming aims to achieve high productivity by cultivating strong-growing Sphagnum species that form an even moss layer with only a small proportion of other plant species that would reduce its suitability as a raw material (Gaudig et al. 2018). However, it is the natural hummock-hollow structure with different moss and vascular plant species and small-scale differences in vegetation at different heights above the water level that meets the habitat requirements of the bog-typical species (Spitzer and Danks 2006). This natural diversity of microhabitats is prevented and homogenized by site management and harvest, so fewer habitat structures for bog-typical beetle species will be provided.
Another critical factor influencing the occurrence of beetles in peatland areas is moisture (Blake et al. 2003; Görn and Fischer 2015; Hoffmann et al. 2016). This was indirectly demonstrated in this study through the influence of the extreme drought and heat in the second year of recording in 2018. While in 2017, annual precipitation of 841 mm was measured at the next meteorological station (Lingen), in 2018, it was 561 mm, which was only 70% of the long-term average value of 799 mm from 1960 to 1990 (Deutscher Wetterdienst 2022). These extreme conditions possibly had a negative impact on the number of species and individuals found in 2018 compared to 2017, which is consistent with other entomological studies conducted during this period (Lange-Kabitz et al. 2021; Muster et al. 2020), although this is not certain due to lack of data from other years, so possible natural fluctuations may also be responsible. At the near-natural bog, a negative effect was also shown in the model results, with a tendency towards fewer species and lower numbers of individuals in 2018. These effects were less pronounced at the cultivation sites and were not evident in the statistical analyses. Here, several opposing developments coincided that partially overlaid the weather effects. These include active irrigation as well as rapid succession at the new sites.
Species composition
The cultivation sites provide a substitute habitat for beetle species specialized in bogs. Of the 22 bog-typical species found on at least one of the cultivation sites, the three rove beetles Myllaena kraatzi, Philonthus nigrita, and Tachyporis transversalis are tyrphobiont according to Koch (1989). The occurrence of Trichocellus cognatus with its Red List status as "Threatened with Extinction" in Germany at cult-PA is also particularly noteworthy. These highly specialized or threatened species were found at the cultivation sites only in areas with high moss cover (> 85%) and high moss carpet (1.6 –1.6 cm), again highlighting the importance of these structures.
It should be noted that the cultivation sites studied were still in the initial stages. In particular, cult-D was characterized by strongly patchy vegetation in which Sphagnum mosses or other plants could not yet form a closed vegetation layer (Grobe et al. 2021). This was also evidenced by species typically inhabiting open sandy soils at cult-D, such as the ground beetles Acupalpus brunnipes or Harpalus tardus (Bräunicke and Trautner 2009). Out of 18 ground beetle species found at the cultivation sites in this study, there are eight matches to the observations of Mainda (2021) at the German Sphagnum farm ‘Rastede’. Among them is Agonum gracile as a tyrphophil species according to Koch (1989). As expected, large mobile species, especially of ground beetles (e.g., Carabus spec.), were missed with the quadrat samples, while Carabus granulatus was one of the dominant species at ‘Rastede’ (Mainda 2021). Pterostichus rhaeticus, the most abundant species at Mainda (2021), was found with only one individual at cult-PA. Although it can be assumed that running-active beetles were under-captured compared to pitfall trapping (Andersen and Arneberg 2016; Spence and Niemelä 1994), it is noticeable that chiefly small-bodied species were present in large numbers in the quadrat samples (e.g., Myllaena intermedia, Euaesthetus spec.). It is known that small species are rather underrepresented in pitfall traps (Arneberg and Andersen 2003; Spence and Niemelä 1994). Out of 19 determined species in Staphylinidae, six were found from Mainda (2021), including the tyrphophil species Philonthus nigrita. In contrast, of the seven species of Dytiscidae, no matches are found with Mainda. It can be expected that with the succession of the vegetation, the species composition of the fauna will change again (cf. Muster et al. 2020). Even though a typical beetle composition of Sphagnum cultivation sites cannot be deduced, it can be assumed that bog species, as the two tyrphophil species Agonum gracile and Philonthus nigrita, benefit from this land use and could find a substitute habitat here. Mainly species with similar habitat requirements, i.e., wet Sphagnum carpet (Koch 1989), can be expected to benefit.
The studied near-natural bog was characterized by floating Sphagnum mats with a stable water table (Oestmann et al. 2021). The higher numbers of Dytiscidae and Hydrophilidae found here reflect this. In this context, the tyrphobiont species Hydroporus scalesianus showed increased individual numbers. This fits with many individuals of the rove beetle Acylopherus wagenschieberi, which is also tyrphobiont and lives in wet Sphagnum carpet, and of the Scirtidae species Cyphon hilaris and C. variabilis, which are associated with water bodies.
Chaetocnema sahlbergii (cult-PA, cult-PC) and Plateumaris discolor (nat-MR, nat-MD) were found as herbivore bog beetles. For both species, Eriophorum angustifolium was probably the host plant (Böhme 2001; Sprick et al. 2013) since other Cyperaceae were present only very sporadically.
Conclusions
The study has shown that at Sphagnum cultivation sites correlations between vegetation structure and beetle occurrence exist, especially for the specialized bog species. A dense and high moss carpet is a fundamental habitat structure for the beetle fauna at bog sites, not only at natural sites but also at Sphagnum cultivation sites. Furthermore, it can be assumed that vascular plants initially help create habitat structures on bare peat. Optimally, however, the vascular plants are then overgrown by the Sphagnum with succession. Conversely, if the vascular plants overgrow and shade the Sphagnum, this can have a negative effect on bog-typical beetle species. These principles should be considered not only in planning Sphagnum cultivation sites but also in restoration or conservation.
The suitability of Sphagnum cultivation sites as substitute habitats for individual bog-typical species was demonstrated. However, cultivation sites as artificial systems with continuous management influence can never develop the same vegetation structure as near-natural sites, so their suitability as beetle habitats will remain limited. It remains to be seen if a typical species composition for cultivation sites will result from the widespread establishment of this land use.
References
Andersen J (1995) A comparison of pitfall trapping and quadrat sampling of Carabidae (Coleoptera) on river banks. Entomol Fennica 6:65–77. https://doi.org/10.33338/ef.83842
Andersen J, Arneberg P (2016) Hand collection as a method for assessing the community structure of carabid beetles. Pedobiologia 59:73–81. https://doi.org/10.1016/j.pedobi.2016.02.003
Arneberg P, Andersen J (2003) The energetic equivalence rule rejected because of a potentially common sampling error: evidence from carabid beetles. Oikos 101:367–375. https://doi.org/10.1034/j.1600-0706.2003.11527.x
Bettacchioli G, Taormina M, Bernini F, Migliorini M (2012) Disturbance regimes in a wetland remnant: implications for trait-displacements and shifts in the assemblage structure of carabid beetles (Coleoptera: Carabidae). J Insect Conserv 16:249–261. https://doi.org/10.1007/s10841-011-9412-9
Blake S, McCracken DI, Eyre MD, Garside A, Foster GN (2003) The relationship between the classification of Scottish ground beetle assemblages (Coleoptera, Carabidae) and the National Vegetation Classification of British plant communities. Ecography 26:602–616. https://doi.org/10.1034/j.1600-0587.2003.03491.x
Bohac J (1999) Staphylinid beetles as bioindicators. Agric Ecosyst Environ 74:357–372. https://doi.org/10.1016/B978-0-444-50019-9.50020-0
Böhme J (2001) Phytophage Käfer und ihre Wirtspflanzen in Mitteleuropa: Ein Kompendium. bioform
Braun-Blanquet J (1964) Pflanzensoziologie. Grundzüge der Vegetationskunde, 3rd edn. Springer, Berlin
Bräunicke M, Trautner J (2009) Lebensraumpräferenzen der Laufkäfer Deutschlands: Wissensbasierter Katalog. Angewandte Carabidologie Supplement V
Brigić A, Bujan J, Alegro A, Šegota V, Ternjej I (2017) Spatial distribution of insect indicator taxa as a basis for peat bog conservation planning. Ecol Indic 80:344–353. https://doi.org/10.1016/j.ecolind.2017.05.007
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Maechler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:378–400. https://doi.org/10.32614/RJ-2017-066
Coulson JC, Butterfield JEL (1985) The Invertebrate communities of peat and upland Grasslands in the North of England and some conservation implications. Biol Cons 34:197–225. https://doi.org/10.1016/0006-3207(85)90093-X
Crawley MJ (2002) Statistical computing: an introduction to data analysis using S-plus. Wiley, Chichester
de Vries HH, den Boer PJ (1990) Survival of populations of Agonum ericeti Panz. (Col., Carabidae) in relation to fragmentation of habitats. Neth J Zool 40:484–498. https://doi.org/10.1163/156854290X00046
Dennis P, Doering J, Stockan JA, Jones JR, Rees ME, Vale JE, Sibbald AR (2004) Consequences for biodiversity of reducing inputs to upland temperate pastures: effects on beetles (Coleoptera) of cessation of nitrogen fertilizer application and reductions in stocking rates of sheep. Grass Forage Sci 59:121–135. https://doi.org/10.1111/j.1365-2494.2004.00412.x
Dormann CF, Elith J, Bacher S, Buchmann C, Carl G, Carré G, Marquéz JRG, Gruber B, Lafourcade B, Leitão PJ, Münkemüller T, McClean C, Osborne PE, Reineking B, Schröder B, Skidmore AK, Zurell D, Lautenbach S (2013) Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36:27–46. https://doi.org/10.1111/j.1600-0587.2012.07348.x
Elo M, Penttinen J, Kotiaho JS (2015) The effect of peatland drainage and restoration on Odonata species richness and abundance. BMC Ecol. https://doi.org/10.1186/s12898-015-0042-z
Gaudig G, Krebs M, Prager A, Wichmann S, Barney M, Caporn SJ, Emmel M, Fritz C, Graf M, Grobe A, Gutierrez Pacheco S, Hogue-Hugron S, Holzträger S, Irrgang S, Kämäräinen A, Karofeld E, Koch G, Köbbing JF, Kumar S, Matchutadze I, Oberpaur C, Oestmann J, Raabe P, Rammes D, Rochefort L, Schmilewski G, Sendžikaitė J, Smolders A, St-Hilaire B, van de Riet B, Wright B, Wright N, Zoch L, Joosten H (2018) Sphagnum farming from species selection to the production of growing media: a review. Mires Peat 20:1–30. https://doi.org/10.19189/MaP.2018.OMB.340
Görn S, Fischer K (2015) Measuring the efficiency of fen restoration on carabid beetles and vascular plants: A case study from north-eastern Germany. Restor Ecol 23:413–420. https://doi.org/10.1111/rec.12203
Görn S, Dobner B, Suchanek A, Fischer K (2014) Assessing human impact on fen biodiversity: Effects of different management regimes on butterfly, grasshopper, and carabid beetle assemblages. Biodivers Conserv 23:309–326. https://doi.org/10.1007/s10531-013-0602-5
Graf M, Bredemeier B, Grobe A, Köbbing JF, Lemmer M, Oestmann J, Rammes D, Reich M, Schmilewski G, Tiemeyer B, Zoch L (2017) Torfmooskultivierung auf Schwarztorf: ein neues Forschungsprojekt in Niedersachsen. Telma 47:109–128. https://doi.org/10.23689/fidgeo-2936
Grobe A (2023) Plant species composition and vegetation structure of Sphagnum cultivation sites. Appl Veg Sci. https://doi.org/10.1111/avsc.12744
Grobe A, Rode M (2023) Factors influencing the establishment of vascular plants at Sphagnum cultivation sites. Wetl Ecol Manag. https://doi.org/10.1007/s11273-023-09927-2
Grobe A, Tiemeyer B, Graf M (2021) Recommendations for successful establishment of Sphagnum farming on shallow highly decomposed peat. Mires Peat 27:1–18. https://doi.org/10.19189/MaP.2020.APG.StA.2022
Gruttke H, Balzer S, Binot-Hafke M, Haupt H, Hofbauer N, Ludwig G, Matzke-Hajek G, Ries M (eds) (2016) Rote Liste gefährdeter Tiere, Pflanzen und Pilze Deutschlands, Band 4: Wirbellose Tiere (Teil 2). Naturschutz und Biologische Vielfalt, 70 (4). Landwirtschaftsverlag, Münster
Habel JC, Trusch R, Schmitt T, Ochse M, Ulrich W (2019) Long-term large-scale decline in relative abundances of butterfly and burnet moth species across south-western Germany. Sci Rep 9:14921. https://doi.org/10.1038/s41598-019-51424-1
Hoffmann H, Michalik P, Görn S, Fischer K (2016) Effects of fen management and habitat parameters on staphylinid beetle (Coleoptera: Staphylinidae) assemblages in north-eastern Germany. J Insect Conserv 20:129–139. https://doi.org/10.1007/s10841-016-9847-0
Hollmen A, Välimäki P, Itämies J, Oksanen J (2008) The value of open power line habitat in conservation of ground beetles (Coleoptera: Carabidae) associated with mires. J Insect Conserv 12:163–177. https://doi.org/10.1007/s10841-007-9076-7
Hölzer A (2010) Die Torfmoose Südwestdeutschlands und der Nachbargebiete. Weissdorn-Verlag, Jena
Hughes P, Lomas-Clarke SH, Schulz J, Barber KE (2008) Decline and localized extinction of a major raised bog species across the British Isles: evidence for associated land-use intensification. The Holocene 18:1033–1043. https://doi.org/10.1177/0959683608095574
Humbert J-Y, Ghazoul J, Walter T (2009) Meadow harvesting techniques and their impacts on field fauna. Agric Ecosyst Environ 130:1–8
Jobin P, Caron J, Rochefort L (2014) Developing new potting mixes with Sphagnum fibers. Can J Soil Sci 94:585–593. https://doi.org/10.4141/cjss2013-103
Joosten H, Couwenberg J (2001) Zur anthropogenen Veränderung der Moore: Bilanzen zum Moorverlust: Das Beispiel Europa. In: Succow M, Joosten H (eds) Landschaftsökologische Moorkunde, 2nd edn. E. Schweizerbart’sche Verlagsbuchhandlung, Stuttgart, pp 406–408
Joosten H, Tanneberger F, Moen A (eds) (2017a) Mires and peatlands of Europe: Status, distribution and conservation. Schweizerbart Science Publishers, Stuttgart
Joosten H, Tanneberger F, Moen A (2017b) Peatland use in Europe. In: Joosten H, Tanneberger F, Moen A (eds) Mires and peatlands of Europe: status, distribution and conservation. Schweizerbart Science Publishers, Stuttgart, pp 151–172
Kassambara A, Mundt F (2020) factoextra: Extract and Visualize the Results of Multivariate Data Analyses. R package version 1.0.7. https://CRAN.R-project.org/package=factoextra. Accessed 31 May 2022
Kato Y, Takemon Y, Hori M (2009) Invertebrate assemblages in relation to habitat types on a floating mat in Mizorogaike Pond, Kyoto, Japan. Limnology 10:167–176. https://doi.org/10.1007/s10201-009-0274-8
Koch K (1989) Die Käfer Mitteleuropas - Ökologie, vol 1. Goecke & Evers, Krefeld
Lange-Kabitz C, Reich M, Zoch A (2021) Extensively managed or abandoned urban green spaces and their habitat potential for butterflies. Basic Appl Ecol 54:85–97. https://doi.org/10.1016/j.baae.2021.04.012
Lompe A (2002) Die Käfer Europas: Ein Bestimmungswerk im Internet. http://www.coleo-net.de. Accessed 4 May 2022
Mainda T (2021) Ten years of beetle community succession in a Sphagnum farm with a focus on Staphylinidae, Carabidae and Dytiscidae. Bachelorarbeit, Universität Greifswald
Matlack GR (1993) Microenvironment variation within and among forest edge sites in the eastern United States. Biol Cons 66:185–194. https://doi.org/10.1016/0006-3207(93)90004-K
Müller R, Glatzel S (2021) Sphagnum farming substrate is a competitive alternative to traditional horticultural substrates for achieving desired hydro-physical properties. Mires Peat 27:1–12. https://doi.org/10.19189/MaP.2021.OMB.StA.2157
Muster C, Gaudig G, Krebs M, Joosten H (2015) Sphagnum farming: the promised land for peat bog species? Biodivers Conserv 24:1989–2009. https://doi.org/10.1007/s10531-015-0922-8
Muster C, Krebs M, Joosten H (2020) Seven years of spider community succession in a Sphagnum farm. J Arachnol. https://doi.org/10.1636/0161-8202-48.2.119
Naimi B, Hamm NA, Groen TA, Skidmore AK, Toxopeus AG (2014) Where is positional uncertainty a problem for species distribution modelling? Ecography 37:191–203. https://doi.org/10.1111/j.1600-0587.2013.00205.x
Oestmann J, Tiemeyer B, Düvel D, Grobe A, Dettmann U (2021) Greenhouse gas balance of sphagnum farming on highly decomposed peat at former peat extraction sites. Ecosystems. https://doi.org/10.1007/s10021-021-00659-z
Peus F (1932) Die Tierwelt der Moore. Handbuch der Moorkunde. III, Berlin
Piepho HP, Büchse A, Emrich K (2003) A Hitchhiker’s guide to mixed models for randomized experiments. J Agron Crop Sci 189:310–322. https://doi.org/10.1046/j.1439-037X.2003.00049.x
Pouliot R, Hugron S, Rochefort L (2015) Sphagnum farming: a long-term study on producing peat moss biomass sustainably. Ecol Eng 74:135–147. https://doi.org/10.1016/j.ecoleng.2014.10.007
Powell CJ, Ades P, Collett N, Phillips C (2013) Epigaeic beetle communities of the Green Triangle plantation landscape, southern Australia: effect of remnant size, vegetation quality and structural complexity. Aust for 76:128–139. https://doi.org/10.1080/00049158.2013.844090
Quinty F, Rochefort L (2003) Peatland restoration guide, 2nd edn. Canadian Sphagnum Peat Moss Association and New Brunswick Department of Natural Resources and Energy, Québec
R Core Team (2022) R: The R Project for Statistical Computing. https://www.R-project.org/. Accessed 12 May 2022
Ries M, Balzer S, Gruttke H, Haupt H, Hofbauer N, Ludwig G, Matzke-Hajek G (eds) (2021) Rote Liste gefährdeter Tiere, Pflanzen und Pilze Deutschlands, Band 5: Wirbellose Tiere (Teil 3). Naturschutz und Biologische Vielfalt, 70 (5). Landwirtschaftsverlag, Münster
Rydin H, Jeglum JK (2006) The biology of peatlands. Oxford University Press, New York
Sanders MD, Winterbourn MJ (1993) Effect of sphagnum harvesting on invertebrate species diversity and community size. Sciene & Research Series, vol 57. Head Office, Department of Conservation, Wellington
Saunders DA, Hobbs RJ, Margules CR (1991) Biological consequences of ecosystem fragmentation: a review. Conserv Biol 5:18–32. https://doi.org/10.1111/J.1523-1739.1991.TB00384.X
Schmatzler E (2015) Moornutzung und Moorschutz in Niedersachsen – Geschichtlicher Rückblick und zukünftige Entwicklung. Telma, Beiheft 5:19–38
Slowikowski K (2021) ggrepel: Automatically Position Non-Overlapping Text Labels with ‘ggplot2’. R package version 0.9.1. https://CRAN.R-project.org/package=ggrepel. Accessed 31 May 2022
Spence JR, Niemelä JK (1994) Sampling carabid assemblages with pitfall traps: the madness and the method. Can Entomol 126:881–894
Sperle T, Bruelheide H (2021) Climate change aggravates bog species extinctions in the Black Forest (Germany). Divers Distrib 27:282–295. https://doi.org/10.1111/ddi.13184
Spitzer K, Danks HV (2006) Insect biodiversity of boreal peat bogs. Annu Rev Entomol 51:137–161. https://doi.org/10.1146/annurev.ento.51.110104.151036
Spitzer K, Bezděk A, Jaroš J (1999) Ecological succession of a relict Central European peat bog and variability of its insect biodiversity. J Insect Conserv. https://doi.org/10.1023/A:1009634611130
Sprick P, Schmidt L, Gärtner E (2013) Bemerkenswerte Kurzflügler (Staphylinidae), phytophage (Chrysomelidae, Curculionoidea) und diverse Käfer aus der Hannoverschen Moorgeest - 1. Beitrag zur Käferfauna (Coleoptera). Telma 43:123–162
Succow M, Joosten H (eds) (2001) Landschaftsökologische Moorkunde, 2nd edn. E. Schweizerbart’sche Verlagsbuchhandlung, Stuttgart
Sushko GG (2017) Diversity and species composition of beetles in the herb-shrub layer of a large isolated raised bog in Belarus. Mires and Peat 19:1–14
Sushko GG (2019a) Key factors affecting the diversity of Sphagnum cover inhabitants with the focus on ground beetle assemblages in Central-Eastern European peat bogs. Commun Ecol 20:45–52. https://doi.org/10.1556/168.2019.20.1.5
Sushko GG (2019b) Autogenic ecological succession of a pristine peat bog: focus on factors affecting beetle diversity. Ecosci 26:165–175. https://doi.org/10.1080/11956860.2018.1564185
Tanneberger F, Joosten H, Moen A, Whinam J (2017) Mire and peatland conservation in Europe. In: Joosten H, Tanneberger F, Moen A (eds) Mires and peatlands of Europe: status, distribution and conservation. Schweizerbart Science Publishers, Stuttgart, pp 173–196
Tanneberger F, Abel S, Couwenberg J, Dahms T, Gaudig G, Günther A, Kreyling J, Peters J, Pongratz J, Joosten H (2021) Towards net zero CO2 in 2050: an emission reduction pathway for organic soils in Germany. Mires and Peat 27:1–17. https://doi.org/10.19189/MaP.2020.SNPG.StA.1951
Thomas CD, Cameron A, Green RE, Bakkenes M, Beaumont LJ, Collingham YC, Erasmus BFN, de Siqueira MF, Grainger A, Hannah L, Hughes L, Huntley B, van Jaarsveld AS, Midgley GF, Miles L, Ortega-Huerta MA, Peterson AT, Phillips OL, Williams SE (2004) Extinction risk from climate change. Nature 427:145–148. https://doi.org/10.1038/nature02121
Tiemeyer B, Freibauer A, Borraz EA, Augustin J, Bechtold M, Beetz S, Beyer C, Ebli M, Eickenscheidt T, Fiedler S, Förster C, Gensior A, Giebels M, Glatzel S, Heinichen J, Hoffmann M, Höper H, Jurasinski G, Laggner A, Leiber-Sauheitl K, Peichl-Brak M, Drösler M (2020) A new methodology for organic soils in national greenhouse gas inventories: Data synthesis, derivation and application. Ecol Indic 109:105838. https://doi.org/10.1016/j.ecolind.2019.105838
Tobisch C, Rojas-Botero S, Uhler J, Müller J, Kollmann J, Moning C, Brändle M, Gossner MM, Redlich S, Zhang J, Steffan-Dewenter I, Benjamin C, Englmeier J, Fricke U, Ganuza C, Haensel M, Riebl R, Uphus L, Ewald J (2023) Plant species composition and local habitat conditions as primary determinants of terrestrial arthropod assemblages. Oecologia 201:813–825. https://doi.org/10.1007/s00442-023-05345-6
Trepel M, Pfadenhauer J, Zeitz J, Jeschke L (2017) Germany. In: Joosten H, Tanneberger F, Moen A (eds) Mires and peatlands of Europe: Status, distribution and conservation. Schweizerbart Science Publishers, Stuttgart, pp 413–424
van Grunsven R, Bos G, Poot M (2020) Strong changes in Dutch dragonfly fauna. Agrion 24:134–138
van Kleef HH, van Duinen G-JA, Verberk WC, Leuven RS, van der Velde G, Esselink H (2012) Moorland pools as refugia for endangered species characteristic of raised bog gradients. J Nat Conserv 20:255–263. https://doi.org/10.1016/j.jnc.2012.05.001
Deutscher Wetterdienst (2022) Klimadaten Deutschland. https://www.dwd.de/DE/leistungen/klimadatendeutschland/klimadatendeutschland.html?nn=495662. Accessed 14 June 2022
Wichtmann W, Schröder C, Joosten H (eds) (2016) Paludiculture - productive use of wet peatlands: Climate protection - biodiversity - regional economic benefits. Schweizerbart Science Publishers, Stuttgart
Wilson D, Blain D, Couwenberg J, Evans CD, Murdiyarso D, Page SE, Renou-Wilson F, Rieley JO, Strack M, Tuittila ES (2016) Greenhouse gas emission factors associated with rewetting of organic soils. Mires Peat 17:1–28. https://doi.org/10.19189/MaP.2016.OMB.222
Zoch L, Reich M (2020) Torfmooskultivierungsflächen als neuer Lebensraum für Moorlibellen. Libellula 39:27–48
Zoch L, Reich M (2022) Torfmooskultivierungsflächen als Lebensraum für Vögel, Amphibien, Libellen und Tagfalter. Naturschutz Und Landschaftsplanung 54:22–31. https://doi.org/10.1399/NuL.2022.11.02
Acknowledgements
We are incredibly grateful to Peter Sprick, Benedikt Feldmann, Ludger Schmidt, and Dieter Siede for confirming the determination of critical species. We thank Marie-Lena Hass and all the other assistants for collecting the ‘‘beetle samples’’. We thank our project partners at Klasmann-Deilmann GmbH for their productive cooperation. The permissions granted by the Weser-Ems Office for Regional State Development (State Bog Administration) and the County Emsland have facilitated the project.
Funding
Open Access funding enabled and organized by Projekt DEAL. This work was supported by the Lower Saxony Ministry of Food, Agriculture and Consumer Protection (ML, AZ 105.1-3234/1-13-3) and the German Federal Environmental Foundation (DBU, AZ 33305/01–33/0).
Author information
Authors and Affiliations
Contributions
LZ and MR created the study conception and design. Material preparation and data collection were performed by LZ. Statistical analysis was done by SB. The first draft of the manuscript was written by LZ, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there were no conflicts of interest for any of the authors regarding this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
See Table 5.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zoch, L., Budig, S. & Reich, M. Sphagnum cultivation sites as habitat for beetles (Coleoptera) and the effect of vegetation structure on species occurrence and abundance. J Insect Conserv 28, 75–88 (2024). https://doi.org/10.1007/s10841-023-00524-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-023-00524-5