Abstract
Objective
The primary objective of this study was to test the hypotheses that compared to IVF cycles undergoing preimplantation genetic testing for aneuploidy (PGT-A) with or without testing for monogenic disorders (PGT-M), IVF cycles undergoing PGT for structural rearrangements (PGT-SR) will have (1) a poorer blastocyst conversion rate and (2) fewer usable blastocysts available for transfer. Secondarily, the study aimed to compare pregnancy outcomes among PGT groups.
Patients
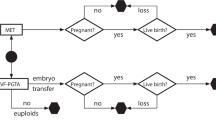
Retrospective cohort study including cycles started from January 1, 2012, to March 30, 2020, with the intent of pursuing PGT-A, PGT-A with PGT-M, and PGT-SR, with trophectoderm biopsy on days 5 or 6.
Results
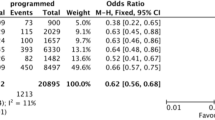
A total of 658 women underwent 902 cycles, including 607 PGT-A, 216 PGT-A&M, and 79 PGT-SR cycles. When compared with the blastocyst conversion rate for the PGT-A group (59.4%), and after adjustment for patient age, total number of mature oocytes, BMI, and ICSI, there were no significant differences for either the PGT-A&M (69.7%, aRR 1.03, 95% CI 0.96–1.10) or PGT-SR (63.2%, aRR1.04, 95% CI 0.96–1.13) groups. Compared to the PGT-A group, the proportion of usable blastocysts was statistically significantly lower in the PGT-SR group: 35.1% versus 24.4% (aRR 0.57, 95% CI 0.46–0.71) and the PGT-A&M group: 35.1% versus 31.5% (aRR 0.68, 95% CI 0.58–0.81). Implantation, pregnancy, and miscarriage rates were equivalent for all groups.
Conclusion
Patients with structural rearrangements have similar blastocyst development but significantly fewer usable blastocysts available for transfer compared to PGT-A testers. Nevertheless, with the transfer of a usable embryo, PGT-SR testers perform as well as those testing for PGT-A.
Similar content being viewed by others
References
Blue NR, Page JM, Silver RM. Genetic abnormalities and pregnancy loss. Semin Perinatol. 2019;43(2):66–73. https://doi.org/10.1053/j.semperi.2018.12.002.
Kaser D. The status of genetic screening in recurrent pregnancy loss. Obstet Gynecol Clin N Am. 2018;45(1):143–54. https://doi.org/10.1016/j.ogc.2017.10.007.
Morin SJ, Eccles J, Iturriaga A, Zimmerman RS. Translocations, inversions and other chromosome rearrangements. Fertil Steril. 2017;107(1):19–26. https://doi.org/10.1016/j.fertnstert.2016.10.013.
Hamerton JL, Canning N, Ray M, Smith S. A cytogenetic survey of 14,069 newborn infants: I. Incidence of chromosome abnormalities. Clin Genet. 1975;8(4):223–43. https://doi.org/10.1111/j.1399-0004.1975.tb01498.x.
Vasilevska M, Ivanovska E, Sabit KK, Angelovska SE, Dimeska G. The incidence and type of chromosomal translocations from prenatal diagnosis of 3800 patients in the Republic of Macedonia. Balkan J Med Genet. 2013;16(2):23–8. https://doi.org/10.2478/bjmg-2013-0027.
Ogilvie CM, Braude P, Scriven PN. Successful pregnancy outcomes after preimplantation genetic diagnosis (PGD) for carriers of chromosome translocations. Hum Fertil (Camb). 2001;4(3):168–71 http://www.ncbi.nlm.nih.gov/pubmed/11591275. Accessed July 13, 2018.
Braekeleer MD, Dao T-N. Cytogenetic studies in couples experiencing repeated pregnancy losses. Hum Reprod. 1990;5(5):519–28. https://doi.org/10.1093/oxfordjournals.humrep.a137135.
Practice Committee of the American Society for Reproductive Medicine T. Evaluation and treatment of recurrent pregnancy loss: a committee opinion. 2012. doi:10.1016/j.fertnstert.2012.06.048
Iews M, Tan J, Taskin O, Alfaraj S, AbdelHafez FF, Abdellah AH, et al. Does preimplantation genetic diagnosis improve reproductive outcome in couples with recurrent pregnancy loss owing to structural chromosomal rearrangement? A systematic review. Reprod BioMed Online. 2018;36(6):677–85. https://doi.org/10.1016/j.rbmo.2018.03.005.
Ikuma S, Sato T, Sugiura-Ogasawara M, Nagayoshi M, Tanaka A, Takeda S. Preimplantation genetic diagnosis and natural conception: a comparison of live birth rates in patients with recurrent pregnancy loss associated with translocation. PLoS One. 2015;10(6). https://doi.org/10.1371/journal.pone.0129958.
Koji S-O, Aoki AE, Tomoyuki Fujii AE, Tomio Fujita AE, Rie Kawaguchi AE, Tetsuo Maruyama AE, et al. Maruyama T, Ozawa N, et al. Subsequent pregnancy outcomes in recurrent miscarriage patients with a paternal or maternal carrier of a structural chromosome rearrangement. J Hum Genet. 2008;53:622–8. https://doi.org/10.1007/s10038-008-0290-2.
Chen C-K, Wu D, Yu H-T, et al. Preimplantation genetic diagnosis by fluorescence in situ hybridization of reciprocal and Robertsonian translocations. doi:https://doi.org/10.1016/j.tjog.2012.04.043, 2014
Idowu D, Merrion K, Wemmer N, Mash JG, Pettersen B, Kijacic D, et al. Pregnancy outcomes following 24-chromosome preimplantation genetic diagnosis in couples with balanced reciprocal or Robertsonian translocations. Fertil Steril. 2015;103(4):1037–42. https://doi.org/10.1016/J.FERTNSTERT.2014.12.118.
Munné S, Escudero T, Sandalinas M, Sable D, Cohen J. Gamete segregation in female carriers of Robertsonian translocations. Cytogenet Cell Genet. 2000;90(3-4):303–8. https://doi.org/10.1159/000056793.
Mateu-Brull E, Rodrigo L, Peinado V, Mercader A, Campos-Galindo I, Bronet F, et al. Interchromosomal effect in carriers of translocations and inversions assessed by preimplantation genetic testing for structural rearrangements (PGT-SR). J Assist Reprod Genet. 2019;36(12):2547–55. https://doi.org/10.1007/s10815-019-01593-9.
Gardner DK, Schoolcraft WB. Jansen R MD. In vitro culture of human blastocysts, toward reproductive certainty: fertility and genetics beyond 1999. London: UK Parthenon Publishing; 1999.
Bakkensen JB, Brady P, Carusi D, Romanski P, Thomas AM, Racowsky C. Association between blastocyst morphology and pregnancy and perinatal outcomes following fresh and cryopreserved embryo transfer. J Assist Reprod Genet. 2019;36(11):2315–24. https://doi.org/10.1007/s10815-019-01580-0.
Gianaroli L, Magli MC, Ferraretti AP, et al. Possible interchromosomal effect in embryos generated by gametes from translocation carriers. Vol 17.; 2002. https://academic.oup.com/humrep/article-abstract/17/12/3201/569611. Accessed August 4, 2020.
Violeta F, Alesja D, Baiba A, et al. The application of PGT-A for carriers of balanced structural chromosomal rearrangements. Gynecol Endocrinol. 2019;35(sup1):18–23. https://doi.org/10.1080/09513590.2019.1632091.
Huang C, Jiang W, Zhu Y, Li H, Lu J, Yan J, et al. Pregnancy outcomes of reciprocal translocation carriers with two or more unfavorable pregnancy histories: before and after preimplantation genetic testing. J Assist Reprod Genet. 2019;36(11):2325–31. https://doi.org/10.1007/s10815-019-01585-9.
Roque M, Haahr T, Geber S, Esteves SC, Humaidan P. OUP accepted manuscript. Hum Reprod Update. 2018;25:2–14. https://doi.org/10.1093/humupd/dmy033.
Munné S, Sandalinas M, Escudero T, Fung J, Gianaroli L, Cohen J. Outcome of preimplantation genetic diagnosis of translocations. Fertil Steril. 2000;73(6):1209–18. https://doi.org/10.1016/S0015-0282(00)00495-7.
Nynas J, Narang P, Kolikonda MK, Lippmann S. Depression and anxiety following early pregnancy loss. Prim Care Companion CNS Disord. 2015;17(1). https://doi.org/10.4088/pcc.14r01721.
Kaneko M, Ohashi H, Takamura T, Kawame H. Psychosocial responses to being identified as a balanced chromosomal translocation carrier: a qualitative investigation of parents in Japan. J Genet Couns. 2015;24(6):922–30. https://doi.org/10.1007/s10897-015-9828-6.
Brier N. Understanding and managing the emotional reactions to a miscarriage. Obstet Gynecol. 1999;93(1):151–5. https://doi.org/10.1016/S0029-7844(98)00294-4.
Hirshfeld-Cytron J, Sugiura-Ogasawara M, Stephenson M. Management of recurrent pregnancy loss associated with a parental carrier of a reciprocal translocation: a systematic review. Semin Reprod Med. 2011;29(06):470–81. https://doi.org/10.1055/s-0031-1293201.
Franssen MTM, Musters AM, van der Veen F, Repping S, Leschot NJ, Bossuyt PMM, et al. Reproductive outcome after PGD in couples with recurrent miscarriage carrying a structural chromosome abnormality: a systematic review. Hum Reprod Update. 2011;17(4):467–75. https://doi.org/10.1093/humupd/dmr011.
Maithripala S, Durland U, Havelock J, Kashyap S, Hitkari J, Tan J, et al. Prevalence and treatment choices for couples with recurrent pregnancy loss due to structural chromosomal anomalies. J Obstet Gynaecol Can. 2018;40(6):655–62. https://doi.org/10.1016/j.jogc.2017.09.024.
Evsikov S, Cieslak J, Verlinsky Y. Effect of chromosomal translocations on the development of preimplantation human embryos in vitro. Fertil Steril. 2000;74(4):672–7. https://doi.org/10.1016/S0015-0282(00)01513-2.
De Krom G, Arens YHJM, Coonen E, et al. Recurrent miscarriage in translocation carriers: no differences in clinical characteristics between couples who accept and couples who decline PGD. Hum Reprod. 2015;30(2):484–9. https://doi.org/10.1093/humrep/deu314.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Insogna, I.G., Lanes, A., Dobson, L. et al. Blastocyst conversion rate and ploidy in patients with structural rearrangements. J Assist Reprod Genet 38, 1143–1151 (2021). https://doi.org/10.1007/s10815-021-02131-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-021-02131-2