Abstract
Purpose
The purpose of the study was to investigate changes in adiponectin system expression in granulosa cells (GCs) and high molecular weight adiponectin levels in serum and follicular fluid (FF) of 40 women with polycystic ovary syndrome (PCOS) compared to those in 40 women with normal ovary function.
Methods
Adiponectin (Adipo), adiponectin receptor 1 (AdipoR1), and adiponectin receptor 2 (AdipoR2) messenger RNA (mRNA) expression levels were measured using quantitative real-time polymerase chain reaction (qRT-PCR). High molecular weight (HMW) adiponectin protein concentration was evaluated by ELISA method. Data were analyzed using Student’s t test and one-way ANOVA in SPSS 21 software. At oocyte retrieval, FF was aspirated and GCs were obtained from a pooled collection of FF per each patient.
Results
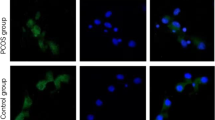
PCR results showed expression of adiponectin, AdipoR1, AdipoR2, follicle-stimulating hormone receptor (FSHR), and luteinizing hormone receptor (LHR) in GCs. After controlling body mass index (BMI) values, qRT-PCR demonstrated a decreased expression of adiponectin system in GCs of PCOS patients compared to those in controls (p = 0.001). There was a strong positive correlation among AdipoR1 and AdipoR2 expression and also among FSH and LH receptor expression. (Both r = 0.8, p = 0.001). There were low levels of high molecular weight adiponectin in the serum of PCOS patients with controlled ovarian hyperstimulation (30.19 ± 4.3 ng/ml) compared to the controls (48.47 ± 5.9 ng/ml) and in the FF of PCOS patients with controlled ovarian hyperstimulation (7.86 ± 1.44 ng/ml) compared to the controls (14.22 ± 2.01 ng/ml; p = 0.02).
Conclusions
Lower expression of adiponectin and its receptors in GCs might be an important manifestation in gonadotropin-stimulated PCOS patients which could influence the physiologic adiponectin roles such as interaction with insulin and LH in induction of GC gene expression.



Similar content being viewed by others
References
Webber LJ, Stubbs SA, Stark J, Margara RA, Trew GH, Lavery SA, et al. Prolonged survival in culture of preantral follicles from polycystic ovaries. J Clin Endocrinol Metab. 2007;92(5):1975–8. doi:10.1210/jc.2006-1422.
Sartor BM, Dickey RP. Polycystic ovarian syndrome and the metabolic syndrome. Am J Med Sci. 2005;330(6):336–42.
Yamauchi T, Nio Y, Maki T, Kobayashi M, Takazawa T, Iwabu M, et al. Targeted disruption of AdipoR1 and AdipoR2 causes abrogation of adiponectin binding and metabolic actions. Nat Med. 2007;13(3):332–9. doi:10.1038/nm1557.
Takemura Y, Osuga Y, Yamauchi T, Kobayashi M, Harada M, Hirata T, et al. Expression of adiponectin receptors and its possible implication in the human endometrium. Endocrinology. 2006;147(7):3203–10.
Guerre-Millo M. Adiponectin: an update. Diabetes Metab. 2008;34(1):12–8. doi:10.1016/j.diabet.2007.08.002.
Maddineni S, Metzger S, Ocon O, Hendricks 3rd G, Ramachandran R. Adiponectin gene is expressed in multiple tissues in the chicken: food deprivation influences adiponectin messenger ribonucleic acid expression. Endocrinology. 2005;146(10):4250–6. doi:10.1210/en.2005-0254.
Chabrolle C, Tosca L, Crochet S, Tesseraud S, Dupont J. Expression of adiponectin and its receptors (AdipoR1 and AdipoR2) in chicken ovary: potential role in ovarian steroidogenesis. Domest Anim Endocrinol. 2007;33(4):480–7. doi:10.1016/j.domaniend.2006.08.002.
Chappaz E, Albornoz MS, Campos D, Che L, Palin MF, Murphy BD, et al. Adiponectin enhances in vitro development of swine embryos. Domest Anim Endocrinol. 2008;35(2):198–207. doi:10.1016/j.domaniend.2008.05.007.
Kadowaki T, Yamauchi T. Adiponectin and adiponectin receptors. Endocr Rev. 2005;26(3):439–51. doi:10.1210/er.2005-0005.
Michalakis KG, Segars JH. The role of adiponectin in reproduction: from polycystic ovary syndrome to assisted reproduction. Fertility and Sterility. 2010;94(6).
Richards JS, Liu Z, Kawai T, Tabata K, Watanabe H, Suresh D, et al. Adiponectin and its receptors modulate granulosa cell and cumulus cell functions, fertility, and early embryo development in the mouse and human. Fertil Steril. 2012;98(2):471–9. e1.
Pierre P, Froment P, Negre D, Rame C, Barateau V, Chabrolle C et al. Role of adiponectin receptors, AdipoR1 and AdipoR2, in the steroidogenesis of the human granulosa tumor cell line, KGN. Human reproduction. 2009:dep292.
Ledoux S, Campos DB, Lopes FL, Dobias-Goff M, Palin MF, Murphy BD. Adiponectin induces periovulatory changes in ovarian follicular cells. Endocrinology. 2006;147(11):5178–86. doi:10.1210/en.2006-0679.
Mao X, Kikani CK, Riojas RA, Langlais P, Wang L, Ramos FJ, et al. APPL1 binds to adiponectin receptors and mediates adiponectin signalling and function. Nat Cell Biol. 2006;8(5):516–23.
Nechamen CA, Thomas RM, Cohen BD, Acevedo G, Poulikakos PI, Testa JR, et al. Human follicle-stimulating hormone (FSH) receptor interacts with the adaptor protein APPL1 in HEK 293 cells: potential involvement of the PI3K pathway in FSH signaling. Biol Reprod. 2004;71(2):629–36.
Mihm M, Evans A. Mechanisms for dominant follicle selection in monovulatory species: a comparison of morphological, endocrine and intraovarian events in cows, mares and women. Reprod Domest Anim. 2008;43(s2):48–56.
Jeppesen JV, Kristensen SG, Nielsen ME, Humaidan P, Dal Canto M, Fadini R, et al. LH-receptor gene expression in human granulosa and cumulus cells from antral and preovulatory follicles. J Clin Endocrinol Metab. 2012;97(8):E1524–31.
Yding Andersen C, Bungum L, Nyboe Andersen A, Humaidan P. Preovulatory progesterone concentration associates significantly to follicle number and LH concentration but not to pregnancy rate. Reprod biomed online. 2011;23(2):187–95.
Goswami D, Conway GS. Premature ovarian failure. Hum Reprod Update. 2005;11(4):391–410.
Voronina E, Lovasco LA, Gyuris A, Baumgartner RA, Parlow AF, Freiman RN. Ovarian granulosa cell survival and proliferation requires the gonad-selective TFIID subunit TAF4b. Dev Biol. 2007;303(2):715–26.
Jonard S, Dewailly D. The follicular excess in polycystic ovaries, due to intra‐ovarian hyperandrogenism, may be the main culprit for the follicular arrest. Hum Reprod Update. 2004;10(2):107–17.
Das M, Djahanbakhch O, Hacihanefioglu B, Saridogan E, Ikram M, Ghali L, et al. Granulosa cell survival and proliferation are altered in polycystic ovary syndrome. J Clin Endocrinol Metab. 2008;93(3):881–7.
Ferrero H, Delgado-Rosas F, Garcia-Pascual CM, Monterde M, Zimmermann RC, Simón C, et al. Efficiency and purity provided by the existing methods for the isolation of luteinized granulosa cells: a comparative study. Hum Reprod. 2012;27(6):1781–9.
Acosta E, Peña Ó, Naftolin F, Ávila J, Palumbo A. Angiotensin II induces apoptosis in human mural granulosa-lutein cells, but not in cumulus cells. Fertil Steril. 2009;91(5):1984–9.
Quinn M, McGregor S, Stanton J, Hessian P, Gillett W, Green D. Purification of granulosa cells from human ovarian follicular fluid using granulosa cell aggregates. Reprod Fertil Dev. 2006;18(5):501–8.
Chilvers R, Bodenburg Y, Denner L, Urban R. Development of a novel protocol for isolation and purification of human granulosa cells. J Assist Reprod Genet. 2012;29(6):547–56.
Centurione L, Giampietro F, Sancilio S, Piccirilli M, Artese L, Tiboni GM, et al. Morphometric and ultrastructural analysis of human granulosa cells after gonadotrophin-releasing hormone agonist or antagonist. Reprod biomed online. 2010;20(5):625–33.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2< sup>− ΔΔCT</sup> method. Methods. 2001;25(4):402–8.
Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative CT method. Nat Protoc. 2008;3(6):1101–8.
Blüher M, Williams CJ, Klöting N, Hsi A, Ruschke K, Oberbach A, et al. Gene expression of adiponectin receptors in human visceral and subcutaneous adipose tissue is related to insulin resistance and metabolic parameters and is altered in response to physical training. Diabetes Care. 2007;30(12):3110–5.
Ahima RS, Lazar MA. Adipokines and the peripheral and neural control of energy balance. Mol Endocrinol. 2008;22(5):1023–31.
Trujillo ME, Scherer PE. Adipose tissue-derived factors: impact on health and disease. Endocr Rev. 2006;27(7):762–78.
Carmina E, Bucchieri S, Mansueto P, Rini G, Ferin M, Lobo RA. Circulating levels of adipose products and differences in fat distribution in the ovulatory and anovulatory phenotypes of polycystic ovary syndrome. Fertil Steril. 2009;91(4):1332–5.
Aroda V, Ciaraldi TP, Chang S-A, Dahan MH, Chang RJ, Henry RR. Circulating and cellular adiponectin in polycystic ovary syndrome: relationship to glucose tolerance and insulin action. Fertil Steril. 2008;89(5):1200–8.
Zhang N, Shi Y-H, Hao C-F, Gu HF, Li Y, Zhao Y-R, et al. Association of +45G15G (T/G) and +276 (G/T) polymorphisms in the ADIPOQ gene with polycystic ovary syndrome among Han Chinese women. Eur J Endocrinol. 2008;158(2):255–60.
Orio Jr F, Palomba S, Cascella T, Milan G, Mioni R, Pagano C, et al. Adiponectin levels in women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2003;88(6):2619–23.
Panidis D, Kourtis A, Farmakiotis D, Mouslech T, Rousso D, Koliakos G. Serum adiponectin levels in women with polycystic ovary syndrome. Hum Reprod. 2003;18(9):1790–6.
Shroff R, Kerchner A, Maifeld M, Van Beek EJ, Jagasia D, Dokras A. Young obese women with polycystic ovary syndrome have evidence of early coronary atherosclerosis. J Clin Endocrinol Metab. 2007;92(12):4609–14.
Barber TM, Hazell M, Christodoulides C, Golding SJ, Alvey C, Burling K, et al. Serum levels of retinol-binding protein 4 and adiponectin in women with polycystic ovary syndrome: associations with visceral fat but no evidence for fat mass-independent effects on pathogenesis in this condition. J Clin Endocrinol Metab. 2008;93(7):2859–65.
Chabrolle C, Tosca L, Rame C, Lecomte P, Royere D, Dupont J. Adiponectin increases insulin-like growth factor I-induced progesterone and estradiol secretion in human granulosa cells. Fertil Steril. 2009;92(6):1988–96. doi:10.1016/j.fertnstert.2008.09.008.
Chabrolle C, Tosca L, Dupont J. Regulation of adiponectin and its receptors in rat ovary by human chorionic gonadotrophin treatment and potential involvement of adiponectin in granulosa cell steroidogenesis. Reproduction. 2007;133(4):719–31. doi:10.1530/REP-06-0244.
Maillard V, Uzbekova S, Guignot F, Perreau C, Ramé C, Coyral-Castel S, et al. Effect of adiponectin on bovine granulosa cell steroidogenesis, oocyte maturation and embryo development. Reprod Biol Endocrinol. 2010;8(23):10.1186.
Campos DB, Palin MF, Bordignon V, Murphy BD. The ‘beneficial’ adipokines in reproduction and fertility. Int J Obes (Lond). 2008;32(2):223–31. doi:10.1038/sj.ijo.0803719.
Kadowaki T, Yamauchi T. Adiponectin and adiponectin receptors. Endocr Rev. 2005;26(3):439–51.
Combs TP, Pajvani UB, Berg AH, Lin Y, Jelicks LA, Laplante M, et al. A transgenic mouse with a deletion in the collagenous domain of adiponectin displays elevated circulating adiponectin and improved insulin sensitivity. Endocrinology. 2004;145(1):367–83.
Madsen EL, Rissanen A, Bruun JM, Skogstrand K, Tonstad S, Hougaard DM, et al. Weight loss larger than 10% is needed for general improvement of levels of circulating adiponectin and markers of inflammation in obese subjects: a 3-year weight loss study. Eur J Endocrinol. 2008;158(2):179–87.
Spranger J, Möhlig M, Wegewitz U, Ristow M, Pfeiffer AF, Schill T, et al. Adiponectin is independently associated with insulin sensitivity in women with polycystic ovary syndrome. Clin Endocrinol. 2004;61(6):738–46.
Orio F, Palomba S, Zullo F, Colao A, Lombard G. Are serum adiponectin levels really reduced in obese women with polycystic ovary syndrome? Hum Reprod. 2004;19:215–6.
Ardawi MS, Rouzi AA. Plasma adiponectin and insulin resistance in women with polycystic ovary syndrome. Fertil Steril. 2005;83(6):1708–16. doi:10.1016/j.fertnstert.2004.11.077.
Garcia V, Orostica L, Poblete C, Rosas C, Astorga I, Romero C, et al. Endometria from obese PCOS women with hyperinsulinemia exhibit altered adiponectin signaling. Horm Metab Res. 2015. doi:10.1055/s-0035-1555806.
Schwarz PE, Towers GW, Fischer S, Govindarajalu S, Schulze J, Bornstein SR, et al. Hypoadiponectinemia is associated with progression toward type 2 diabetes and genetic variation in the ADIPOQ gene promoter. Diabetes Care. 2006;29(7):1645–50.
Jalovaara K, Santaniemi M, Timonen M, Jokelainen J, Kesäniemi YA, Ukkola O, et al. Low serum adiponectin level as a predictor of impaired glucose regulation and type 2 diabetes mellitus in a middle-aged Finnish population. Metabolism. 2008;57(8):1130–4.
Zyriax B-C, Algenstaedt P, Hess UF, Schöffauer M, Bamberger C, Boeing H, et al. Factors contributing to the risk of cardiovascular disease reflected by plasma adiponectin: data from the coronary risk factors for atherosclerosis in women (CORA) study. Atherosclerosis. 2008;200(2):403–9.
Rotterdam E, ASRM-Sponsored P. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Human reprod (Oxford, England). 2004;19(1):41.
Jakimiuk AJ, Weitsman SR, Navab A, Magoffin DA. Luteinizing hormone receptor, steroidogenesis acute regulatory protein, and steroidogenic enzyme messenger ribonucleic acids are overexpressed in thecal and granulosa cells from polycystic ovaries 1. J Clin Endocrinol Metab. 2001;86(3):1318–23.
Liu N, Ma Y, Wang S, Zhang X, Zhang Q, Zhang X, et al. Association of the genetic variants of luteinizing hormone, luteinizing hormone receptor and polycystic ovary syndrome. Reprod Biol Endocrinol. 2012;10(1):36.
Catteau-Jonard S, Jamin SP, Leclerc A, Gonzalès J, Dewailly D, di Clemente N. Anti-Mullerian hormone, its receptor, FSH receptor, and androgen receptor genes are overexpressed by granulosa cells from stimulated follicles in women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2008;93(11):4456–61.
Wickham 3rd EP, Tao T, Nestler JE, McGee EA. Activation of the LH receptor up regulates the type 2 adiponectin receptor in human granulosa cells. J Assist Reprod Genet. 2013;30(7):963–8. doi:10.1007/s10815-013-0012-3.
Deepa SS, Dong LQ. APPL1: role in adiponectin signaling and beyond. Amn J Physiology-Endocrinol Metab. 2009;296(1):E22–36.
Acknowledgments
We express our appreciation to the Research Center for Endometrium and Endometriosis and Research Center for Molecular Medicine, Hamadan University of Medical Sciences, for their technical assistance.
This work was supported by the Research and Technology Deputy of Iran University of Medical Sciences as a Ph.D. thesis (grant no. 16169).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This cross-sectional study was reviewed and approved by the Iran University of Medical Sciences Ethics Committee. An informed consent was obtained from each participant.
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Capsule The low expression of adiponectin and its receptors in PCOS patients could influence the oocyte quality and may lead to intrinsic oocyte abnormalities.
Rights and permissions
About this article
Cite this article
Artimani, T., Saidijam, M., Aflatoonian, R. et al. Downregulation of adiponectin system in granulosa cells and low levels of HMW adiponectin in PCOS. J Assist Reprod Genet 33, 101–110 (2016). https://doi.org/10.1007/s10815-015-0620-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-015-0620-1