Abstract
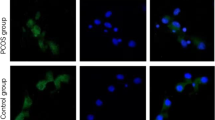
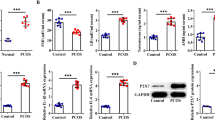
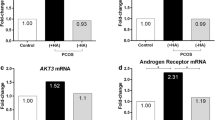
Hyperandrogenemia is the leading defect in patients with polycystic ovary syndrome (PCOS) and considered to be involved in the ovulation dysfunction of PCOS. During the process of ovulation, granulosa cells (GCs) undergo epithelial–mesenchymal transition (EMT), and integrin-interacting protein kindlin 2 is a well-known regulator in EMT. Therefore, our objective here was to compare the expression levels of kindlin 2 in luteinized GCs between patients with PCOS and control women and the relationship between kindlin 2 and PCOS pathogenesis. In this study, kindlin 2 expression was significantly increased in luteinized GCs from patients with PCOS, and kindlin 2 could be induced by testosterone both in vitro and in vivo. Meanwhile, kindlin 2 was positively correlated with androgen receptor (AR) in PCOS GCs. Taken together, kindlin 2 may play a role in luteinized GCs, especially in the case of excess androgen. Further studies are required to assess the specific role of kindlin 2 in follicular development and PCOS pathogenesis.
Similar content being viewed by others
References
Azziz R, Carmina E, Dewailly D, et al. The androgen excess and PCOS society criteria for the polycystic ovary syndrome: the complete task force report. Fertil Steril. 2009;91(2):456–488.
Norman RJ, Dewailly D, Legro RS, Hickey TE. Polycystic ovary syndrome. Lancet. 2007;370(9588):685–697.
Homburg R. Androgen circle of polycystic ovary syndrome. Hum Reprod. 2009;24(7):1548–1555.
Jonard S, Dewailly D. The follicular excess in polycystic ovaries, due to intra-ovarian hyperandrogenism, may be the main culprit for the follicular arrest. Hum Reprod Update. 2004; 10(2):107–117.
Wickenheisser JK, Nelson-DeGrave VL, Hendricks KL, Legro RS, Strauss JF 3rd, McAllister JM. Retinoids and retinol differentially regulate steroid biosynthesis in ovarian theca cells isolated from normal cycling women and women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2005;90(8):4858–4865.
Tziomalos K, Katsikis I, Papadakis E, Kandaraki EA, Macut D, Panidis D. Comparison of markers of insulin resistance and circulating androgens between women with polycystic ovary syndrome and women with metabolic syndrome. Hum Reprod. 2013;28(3): 785–793.
Qiao J, Feng HL. Extra- and intra-ovarian factors in polycystic ovary syndrome: impact on oocyte maturation and embryo developmental competence. Hum Reprod Update. 2011;17(1):17–33.
Duggavathi R, Murphy BD. Development. ovulation signals. Science. 2009;324(5929):890–891.
Catteau-Jonard S, Jamin SP, Leclerc A, Gonzalès J, Dewailly D, di Clemente N. Anti-Mullerian hormore, its receptor, FSH receptor, and androgen receptor genes are overexpressed by granulosa cells from stimulated follicles in women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2008;93(11): 4456–4461.
Tu Y, Wu S, Shi X, Chen K, Wu C. Migfilin and Mig-2 link focal adhesions to filamin and the actin cytoskeleton and function in cell shape modulation. Cell. 2003;113(1):37–47.
An Z, Dobra K, Lock JG, Strömblad S, Hjerpe A, Zhang H. Kindlin-2 is expressed in malignant mesothelioma and is required for tumor cell adhesion and migration. Int J Cancer. 2010;127(9): 1999–2008.
Gong X, An Z, Wang Y, et al. Kindlin-2 controls sensitivity of prostate cancer cells to cisplatin-induced cell death. Cancer Lett. 2010;299(1):54–62.
Yu Y, Wu J, Wang Y, et al. Kindlin 2 forms a transcriptional complex with β-catenin and TCF4 to enhance Wnt signalling. EMBO Rep. 2012;13(8):750–758.
Zhan J, Zhu X, Guo Y, et al. Opposite role of kindlin-1 and kindlin-2 in lung cancers. PLoS One. 2012;7(11):e50313.
Zhao T, Guan L, Yu Y, et al. Kindlin-2 promotes genome instability in breast cancer cells. Cancer Lett. 2013;330(2):208–216.
Kalluri R, Weinberg RA. The basics of epithelial-mesenchymal transition. J Clin Invest. 2009;119(6):1420–1428.
Rodgers RJ, Irving-Rodgers HF, van Wezel IL, Krupa M, Lavranos TC. Dynamics of the membrana granulosa during expansion of the ovarian follicular antrum. Mol Cell Endocrinol. 2001; 171(1–2):41–48.
Rodgers RJ, Irving-Rodgers HF. Extracellular matrix of the bovine ovarian membrana granulosa. Mol Cell Endocrinol. 2002;191(1):57–64.
Franks S, Stark J, Hardy K. Follicle dynamics and anovulation in polycystic ovary syndrome. Hum Reprod Update. 2008;14(4): 367–378.
Das M, Djahanbakhch O, Hacihanefioglu B, et al. Granulosa cell survival and proliferation are altered in polycystic ovary syndrome. J Clin Endocrinol Metab. 2008;93(3):881–887.
The Rotterdam ESHRE/ASRM-Sponsored PCOS consensus workshop group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Hum Reprod. 2004;19(1):41–47.
Escobar-Morreale HF, Carmina E, Dewailly D, et al. Epidemiology, diagnosis and management of hirsutism: a consensus statement by the androgen excess and polycystic ovary syndrome society. Hum Reprod Update. 2012;18(2):146–170.
Gu Y, Liang X, Wu W, et al. Multicenter contraceptive efficacy trial of injectable testosterone undecanoate in Chinese men. J Clin Endocrinol Metab. 2009;94(6):1910–1915.
Wood JR, Dumesic DA, Abbott DH, Strauss JF 3rd. Molecular abnormalities in oocytes from women with polycystic ovary syndrome revealed by microarray analysis. J Clin Endocrinol Metab. 2007;92(2):705–713.
Dumesic DA, Abbott DH. Implication of polycystic ovary syndrome on oocyte development. Semin Reprod Med. 2008;26(1): 53–61.
Franks S. Do animal models of polycystic ovary syndrome help to understand its pathogenesis and management? yes, but their limitations should be recognized. Endocrinology. 2009;150(9): 3983–3985.
Jansen E, Laven JS, Dommerholt HB, et al. Abnormal gene expression profiles in human ovaries from polycystic ovary syndrome patients. Mol Endocrinol. 2004;18(12):3050–3063.
Ehrmann DA. Polycystic ovary syndrome. N Engl J Med. 2005; 352(12):1223–1236.
Bennett NC, Gardiner RA, Hooper JD, Johnson DW, Gobe GC. Molecular cell biology of androgen receptor signalling. Int J Biochem Cell Biol. 2010;42(6):813–827.
Sen A, Hammes SR. Granulosa cell-specific androgen receptors are critical regulators of ovarian development and function. Mol Endocrinol. 2010;24(7):1393–1403.
Weil SJ, Vendola K, Zhou J, et al. Androgen receptor gene expression in the primate ovary: cellular localization, regulation, and functional correlations. J Clin Endocrinol Metab. 1998;83(7): 2479–2485.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yang, M., Du, J., Lu, D. et al. Increased Expression of Kindlin 2 in Luteinized Granulosa Cells Correlates With Androgen Receptor Level in Patients With Polycystic Ovary Syndrome Having Hyperandrogenemia. Reprod. Sci. 21, 696–703 (2014). https://doi.org/10.1177/1933719113512536
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719113512536