Abstract
Purpose
The aim of this study was to detect the effects of different cooling rates and different ice-seeding temperatures on the cryopreservation of whole ovaries.
Methods
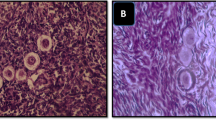
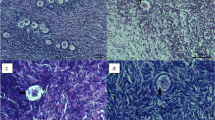
Cow whole ovaries were slowly frozen using different protocols with different cooling rates and different ice-seeding temperatures. Follicular viability was assessed using the trypan blue test; the percentage of morphologically normal primordial follicles and the follicular densities of grafts were measured.
Results
Protocol IIb was most effective protocol. Protocol Ib was more effective than protocol Ia and protocol Ic, and protocol IIIb was more effective than protocol IIIa and protocol IIIc.
Conclusions
Protocol IIb (the cooling rate was 0.2°C/min, and the ice-seeding temperature was −5°C) was appropriate for slow freezing of cow whole ovaries.








Similar content being viewed by others
References
Apperley JF, Reddy N. Mechanism and management of treatment-related gonadal failure in recipients of high dose chemoradiotherapy. Blood Rev. 1995;9:93–116.
Baker TG. Radiosensitivity of manmmalian oocytes with partiular reference to the human female. Am J Obstet Gynecol. 1971;110:746–61.
Oktay K, Karlikay G. Ovarian function after transplantation of frozen, banked autologous ovarian tissue. N Engl J Med. 2000;342:1919.
Oktay K, Buyuk E. Fertility preservation in women undergoing cancer treatment. Lancet. 2004;363:1830.
Radford JA, Lieberman BA, Brison DR, Smith AR, Critchlow JD, Russell SA, et al. Orthotopic reimplantation of cryopreserved ovarian cortical strips after high-dose chemotherapy for Hodgkin’s lymphoma. Lancet. 2001;357:1172–5.
Tryde Schmidt KL, Yding Andersen C, Starup J, Loft A, Byskov AG, Nyboe Andersen A. Orthotopic autotranplantation of cryopreserved ovarian tissue to a woman cured of cancer-follicular growth, steroid production and oocyte retrieval. Reprod Biomed Online. 2004;8:448–53.
Schmidt KL, Andersen CY, Loft A, Byskov AG, Ernst E, Andersen AN. Follow-up of ovarian function post-chemotherapy following ovarian cryopreservation and transplantation. Hum Reprod. 2005;20:3539–46.
Nugent D, Newton H, Gosden RG, Rutherford AJ. Investigation of follicle survival after human heterotopic grafting. Hum Reprod. 1998;13:23–4.
Liu J, Van der Elst J, Van den Broecke R, Dhont M. Early massive follicle loss and apoptosis in heterotopically grafted newborn mouse ovaries. Hum Reprod. 2002;17:605–11.
Aubard Y, Piver P, Cogni Y, Vermeaux V, Poulin N, Driancourt MA. Orthotopic and heterotopic autografts of frozen-thawed ovarian cortex in sheep. Hum Reprod. 1999;14:2149–54.
Imhof M, Bergmeiter H, Lipovac M, Rudas M, Hofstetter G, Huber J. Orthotopic microvascular reanastomosis of whole cryopreserved ovine ovaries resulting in pregnancy and live birth. Fertil Steril. 2006;85(1 Suppl):1208–15.
Courbiere B, Caquant L, Mazoyer C, Franck M, Lornage J, Salle B. Difficulties improving ovarian functional recovery by microvascular transplantation and whole ovary vitrification. Fertil Steril. 2009;91:2697–706.
Martinez-Madrid B, Dolmans MM, Van Langendonckt A, Defrere S, Donnez J. Freeze-thawing intact human ovary with its vascular pedicle with a passive cooling device. Fertil Steril. 2004;82:1390–4.
Trad FS, Toner M, Biggers JD. Effects of cryoprotectants and ice-seeding temperature on intracellular freezing and survival of human oocytes. Hum Reprod. 1998;14:1569–77.
Gook DA, Edgar DH, Stern C. Effect of cooling and dehydration regimen on the histological appearance of human ovarian cortex following cryopreservation in 1,2-propanediol. Hum Reprod. 1999;14:2061–8.
Miyamoto H, Ishibashi T. Effects of the temperature of ice-seeding on survival of frozen-and-thawed mouse morulae. Experientia. 1981;37:187–8.
Gerritse R, Beerendonk CCM, Tijink MSL, Heetkamp A, Kremer JAM, Braat DDM, et al. Optimal perfusion of an intact ovary as a prerequisite for successful ovarian cryopreservation. Hum Reprod. 2008;23:329–35.
Nisolle M, Casanas-Roux FQuJ, Motta P, Donnez J. Histologic and ultrastructural evaluation of fresh and frozen-thawed human ovarian xenografts in nude mice. Fertil Steril. 2000;74:122–9.
Chen CH, Chen SG, Wu GJ, Wang J, Yu CP, Liu JY. Autologous heterotopic transplantation of intact rabbit ovary after frozen banking at −196°C. Fertil Steril. 2006;86(4 Suppl):1059–66.
Yin H, Wang X, Kim SS, Chen H, Tan SL, Gosden RG. Transplantation of intact rat gonads using vascular anastomosis: effects of cryopreservation, ischemia and genotype. Hum Reprod. 2003;18:1165–72.
Wang X, Chen H, Yin H, Kim SS, Lin Tan S, Gosden RG. Fertility after intact ovary transplantation. Nature. 2002;415:385.
Fabbri R, Venturoli SD, Errico A, Iannascoli C, Gabusi E, Valeri B, et al. Ovarian tissue banking and fertility preservation in cancer patients: histological and immunohistochemical evaluation. Gynecol Oncol. 2002;89:259–66.
Neto V, Buff S, Lornage J, Bottolier B, Guérin P, Joly T. Effects of different freezing parameters on the morphology and viability of preantral follicles after cryopreservation of doe rabbit ovarian tissue. Fertil Steril. 2008;89:1348–56.
Sadeu JC, Cortvindt R, Ron-EI R, Kasterstein E, Smitz J. Morphological and ultrastructural evaluation of cultured frozen-thawed human fetal ovarian tissue. Fertil Steril. 2006;85:1130–5.
Perdrix A, Macé B, Milazzo JP, Liard-Zmuda A, Baron M, Rives N. Ovarian tissue thawing: a comparison of two conditions. Fertil Steril. 2010;93:307–10.
Wang Y. Reply: human ovarian tissue: vitrification versus conventional freezing. Hum Reprod. 2009;24:1768–9.
Fauque P, Amor AB, Joanne C, Agnani G, Bresson JL, Roux C. Use of trypan blue staining to assess the quality of ovarian cryopreservation. Fertil Steril. 2007;87:1200–7.
Courbiere B, Massardier J, Salle B, Mazoyer C, Guerin JF, Lornage J. Follicular viability and histological assessment after cryopreservation of whole sheep ovaries with vascular pedicle by vitrification. Fertil Steril. 2005;84(2 Suppl):1065–71.
Courbiere B, Odagescu V, Baudot A, Massardier J, Mazoyer C, Salle B, et al. Cryopreservation of the ovary by vitrification a an alternative to slow-cooling protocols. Fertil Steril. 2006;86(4 Suppl):1243–51.
Nakamura T, Takagi H, Shima J. Effects of ice-seeding temperature and intracellular trehalose contents on survival of frozen saccharomyces cerevisiae cells. Cryobiology. 2009;58:170–4.
Acknowledgements
Appreciation is extended to Dr Bin Li, Ph.D., for assistance in statistics analysis and Dr Yu Wang Ph.D. from department of pathology for supporting this work.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by: 1. Postdoctoral Foundation for Innovation Project of Shandong Province, China (Grant No: 200903074). 2. National Basic Research Program of China (973 Program) (2007CB947403).
Capsule
Protocol IIb (the cooling rate was 0.2°C/min, and the ice-seeding temperature was −5°C) was appropriate for slow freezing of cow whole ovaries.
Rights and permissions
About this article
Cite this article
Zhang, JM., Sheng, Y., Cao, YZ. et al. Effects of cooling rates and ice-seeding temperatures on the cryopreservation of whole ovaries. J Assist Reprod Genet 28, 627–633 (2011). https://doi.org/10.1007/s10815-011-9557-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-011-9557-1