Abstract
We examined the impact of adding the seaweed Cystoseira baccata (Ochrophyta, Sargassaceae) in various forms to two different growing substrates: pine bark and gorse compost. Specifically, we examined the influence of the seaweed on the physical and chemical properties of the substrates, and on their agronomic performance on a lettuce crop. The seaweed was used in a 20% (v/v) proportion and three different forms, namely: fresh (FS), washed fresh (WFS), and washed and dried (WDS). The mixed substrates exhibited no signs of instability. FS and DWS increased the total water retention capacity of pine bark by 20% and 27%, respectively. Adding the seaweed in any of its three forms to this type of substrate, which is poor in nutrients and has a low electrical conductivity (EC), significantly increased its P, K, Mg and Na contents, as well as its EC (from 0.08 dS m–1 in the control substrate to 0.69, 0.12 and 0.27 dS m–1 in those containing FS, WFS and WDS, respectively). On the other hand, only in fresh form (FS) altered the salinity and total K content of a substrate rich in nutrients and salts such gorse compost (from 0.89 to 1.42 dS m−1 in terms of EC and 0.59% to 0.98% in K). All mixtures performed well as substrates for a lettuce crop. Those containing DWS increased aerial mass in gorse compost, while any of the tested formats increased aerial mass in pine bark.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The expansion of intensive agriculture has raised an increasing to need to grow plants off-ground (particularly in pots and other containers). This in turn has made new inputs such as plant growing substrates necessary and highly useful for protected intensive horticulture, nursery cropping, improved sowing, gardening and soil reclamation, among other purposes. Ultimately, the unstoppable growth of soil-less cropping has prompted a search for suitable materials (Caron and Rochefort 2013).
Peat continues to be the most widely used substrate for soil-less cultivation, mainly on the grounds of its good properties and easy handling (Schmilewski 2008). However, peat cannot be renewed and its withdrawal from nature can lead to the destruction of highly fragile ecosystems (Holmes 2007) which act as major carbon sinks (Bullock et al. 2012; Ceglie et al. 2015). Also, peat is largely imported from other countries. The previous reasons have prompted a search for alternative materials to be used as growing substrates (Morel et al. 2000; Caron and Rochefort 2013; Raviv 2013). Such materials, however, can contain toxins, pathogens, weed seeds and immobilized nitrogen, and be unstable and oxygen-deficient (Naasz et al. 2009). Coir, pine bark and composts have emerged as effective, solid alternatives to peat in recent times (Schmilewski 2008).
The search for non-peat substrates has increasingly focused on valorised organic wastes or by-products to avoid their dumping (Raviv 2008) and to obtain them from the vicinity of farms to additionally reduce their carbon footprint. Alluvial seaweed possesses a very high ecological significance but can pose serious disposal problems if it accumulates in large amounts (Harb and Chow 2022). This is particularly so when eutrophication associated to farming, urban and industrial wastewater, or aquaculture led to green tides (Bárbara and Cremades 2010) or during the summer, when seaweed tends to accumulate and decay on beaches, thus raising off-odours or even public health problems.
Seaweed is a practical, economical source of useful compounds for the nutraceutical, pharmaceutical, chemical, food and cosmetic industries (Biris-Dorhoi et al. 2020). For example, beach cast seaweed, especially of the brown kind, has traditionally been applied to crops grown on coastal areas (Metting et al. 1990). Although the main agricultural use of seaweed has been in amendments or fertilizers directly applied to soil, the increasing development of “blue technology”, which is based on marine organisms (Berthon et al. 2021), has promoted the use of seaweed for a variety of agricultural purposes. In fact, seaweed is currently used in various forms in fertilizers. In some cases, it is previously charged with specific ions by bioabsorption from wastewater containing heavy metals (Bădescu et al. 2017). Seaweed is also being increasingly used in biostimulants and as a means of improving soil properties (Illera-Vives et al. 2020). However, it has been little explored as a growing substrate and no commercial forms for this use exist as yet.
Agricultural seaweeds could be an optimal component of growing substrates by virtue of its being natural, biodegradable, free of weed seeds and acceptable for organic production (EC Council 889/2008). In addition, they supply crops with substantial amounts mineral nutrients (Verkleij 1992; Cabrita et al. 2016), vitamins (Bourgougnon et al. 2011) and complex organic compounds, and primary and secondary metabolites (Biris-Dorhoi et al. 2020). Furthermore, the enzymes and biocidal compounds they contain (Illera-Vives et al. 2020) act as biostimulants facilitating crop development (Crouch et al. 1992; Berthon et al. 2021), and their good rheological properties can help avoid moisture losses. In any case, the agronomically significant properties of seaweeds differ among taxonomic groups, species, year, season or even location (Villares et al. 2007; Adams et al. 2011; Sharma et al. 2012; Schiener et al. 2015). Therefore, developing effective seaweed-based products for addition to agricultural substrates requires careful assessment of their properties and performance.
The primary aims of this work was to examine the use of the brown alga Cystoseira baccata as a component of growing substrates to be applied to seedbox-grown horticultural crops. Cystoseira baccata in fresh, unwashed (FS), washed (WFS), and dried and washed form (WFS), was examined for its effect on the physical and chemical properties of two widely used commercial substrates for organic agriculture, namely: pine bark and gorse compost.
Material and methods
Preparation of substrates
Seaweed
Cystoseira baccata (S. G. Gmelin) P. C. Silva is a brown seaweed growing in Atlantic waters and widely distributed in the northwest of the Iberian Peninsula (García-Fernández 2020). Cystoseira baccata was collected during low tide on the As Margaridas beach (Santa Cruz-Oleiros, A Coruña, NW Spain; 43°21′47.5″ N, 8°20′46.5″ W). Its composition is summarized in Table 1.
The material was used in three different forms, namely:
-
Fresh seaweed (FS), which was obtained simply by hand clearing all other species and removing spurious material such as seashells or plants reaching the shore, followed by cutting into largely uniform fragments not longer than 2 cm.
-
Washed fresh seaweed (WFS), in which the high salinity of the previously collected material was reduced prior to use by immersion in a 1:4 (v/v) proportion in water without stirring, followed by cutting as with FS.
-
Dried washed seaweed (DWS), which was prepared by drying a fraction of WFS in a Selecta Dry-big forced air oven at 40 °C for 48 h to reduce moisture from 78 to 13% prior to cutting as with the previous two forms.
Substrates
The three forms of seaweed used were mixed with two different commercial growing substrates, namely: (a) pine bark from Dermont, which was placed under a hot air stream at 300 °C to degrade phytotoxins, pathogens and adventitious seeds; and (b) gorse (Ulex europaeus L.) compost from Abonos Lourido (http://abonoslourido.com) (see Table 1).
The fresh seaweed mixture was prepared by adding a 1:5 (v/v) proportion of FS to the substrate (pine bark or compost). The resulting dry weight ratio was 1:0.06 for pine bark and 1:0.13 for the compost. The substrates containing WFS or DWS were prepared on the basis of dry weights to avoid the impact of drying on the results. Table 2 shows the amount of seaweed used in each sample.
All mixtures were placed in 60 L plastic containers in the laboratory and checked for stability by using an aliquot to measure temperature, pH and moisture on a weekly basis for one month. Appropriate aeration and substrate uniformity were ensured by hand turnover (specifically, with two successive transfers where the material was gently tapped for increased aeration and sponginess).
Physical and chemical characterization of substrates
As stated above, the substrates were turned over on a weekly basis for 1 month to ensure adequate aeration and uniformity. Once all samples were confirmed to be stable, they were characterized chemically by analysis for pH, electrical conductivity (EC), organic matter, water-soluble elements and CaCl2 + DTPA extractives (viz., N-NH4+, K+, Ca2+, Mg2+, Na+, Cl– and PO43–) according to European standards EN 13037 (2011), EN 13038 (2011), EN 13039 (2011) and EN 13652 (2001), respectively. Total C and N were determined on a Leco 2000 autoanalyser; total Ca, Mg, Na and K by inductively coupled plasma–optical emission spectroscopy (ICP–OES) following attack with H2SO4 (Thomas et al. 1967); P colorimetrically (Chapman and Pratt 1997); and heavy metals (Cd, Cu, Cr, Hg, Mn, Ni and Zn) by ICP–OES upon attack with HNO3 in an ETHOS 900 Microwave Labstation oven.
Bulk density, particle density and total porosity were determined according to EN 13041 (2012), with measurements at 1, 3, 5, 7.5 and 10 kPa. Moisture contents were expressed as volume fractions (v:v). Aeration capacity (AC) was calculated as the difference between total porosity and the moisture content by volume at 1 kPa, easily available water (EAW) as the volume fraction of moisture retained by each substrate at 1 − 5 kPa, buffering capacity water (BCW) as the amount of moisture retained from 5 to 10 kPa, and unavailable water (UW) as that retained above 10 kPa (Felipó et al. 1979).
Agronomic evaluation of substrates
The agronomic potential of the substrates, and their interactions with plants, were assessed in terms of seed germination and plant growth.
Seed germination
Seaweed phytotoxicity was assessed according to Zucconi et al. (1981a) using watercress (Lepidium sativum L.) seeds. For this purpose, 60 mL of sample was added to 300 mL of distilled water and mixed for 2 h. Then, 10 seeds were placed on pieces of filter paper and supplied with 5 mL of aqueous extract in Petri dishes. The seeds were incubated at 28 °C in the dark and then used to determine the average germination (AG), average root length per plant (ARLP) and Munoo–Liisa vitality index (MLV). The last parameter was calculated as follows:
where GRsi is the average germination of each replicate i in a treatment (in percentage); GRc that of the control sample (peat); RLsi the average total length of roots for each replicate i in a treatment; and RLc that for the control treatment (peat).
Lettuce growth in a seedbox
Agronomic tests were conducted in a glass greenhouse with automatic air ventilation and sprinkling. A total of 10 seeds of lettuce (Lactuca sativa L.) were sown in each of 75 mL tray cells. Forty-one days later the fresh root and aerial weight were measured to calculate the growth promotion index (GrP) as
where APW is the average plant weight for each treatment.
Statistical analysis
Data were processed with the software package IBM SPSS Statistics v. 23.0. Treatment means were compared via ANOVA and significant differences (p < 0.05) between treatments identified with Duncan’s test following confirmation of data normality with the Kolmogorov–Smirnov test and variance homoscedasticity with Levene’s test. When the latter failed, data were subjected to the Games–Howell test.
Results
Mixture stability
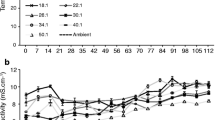
None of the mixtures used exhibited a large enough increase in temperature to suggest microbial activity (Fig. 1). The greatest increase was that in the pine bark–FS mixture, whose temperature was up to 4.8 °C higher than that of the control substrate. The difference, however, soon vanished and the temperature was eventually similar to those resulting from the other treatments. This pine bark–FS mixture also exhibited the greatest pH decrease (Fig. 1), which however was very small (only 0.25 units, from 5.22 in the control substrate to 4.97 in the mixture). No appreciable acidification was observed in any other mixture.
Physical properties
All mixtures were highly porous —porosity was invariably above 85% (Table 3). This was an intrinsic feature of both substrates that was slightly increased by fresh and washed seaweed; the increase, however, was less 1% in all cases. Also, the initial substrates featured too high aeration capacity and poor retention of easily available water (EAW) (50% in gorse compost and 47% in pine bark) as a result of their particle size distribution. Adding seaweed to pine bark reduced its aeration capacity, especially with the dried washed form (DWS), which decreased it to 26% —a level close to the optimum value (20–30%).
The addition of fresh (FS) or dried seaweed (DWS) increased the total water retention capacity (EAW + WBC + UW) of pine bark (by 20% with FS and 27% with DWS). The increase was largely the result of one in easily available water by up to 70% with DWS. The increased water retention capacity of the pine bark substrate led to an R value falling in the recommended range (1–3). No similar effect, however, was observed in gorse compost. In fact, adding seaweed to the compost increased the proportion of EAW to a statistically significant, but very small, extent for practical purposes.
Chemical properties
Adding FS to pine bark decreased its pH from 5.54 to 5.13 (Fig. 2). The pH of the pine bark mixtures ranged from 5.73 to 5.13 and was thus optimal for their use as growing substrates. On the other hand, the pH of the gorse compost mixtures was invariably below 5 (4.44–4.56) and hence bound to compromise nutrient availability.
Chemical and physico–chemical changes in pine bark by effect of the addition of seaweed in three different forms (mean ± standard deviation, n = 3). Different letters in each column denote significant differences between treatments at p < 0.05 as per Duncan’s test. FS fresh seaweed; WFS washed fresh seaweed; DWS dried washed seaweed
The seaweed forms used altered EC to a different extent with each substrate. Thus, addition of any form to a low conductivity substrate such as pine bark (EC = 0.08 dS m–1) increased its conductivity (by 0.69, 0.12 and 0.27 dS m–1 with FS, WFS and WDS, respectively). However, the increase with WFS was significantly less than it was with the other two forms. However, EC in the gorse compost substrate, which had a higher initial value (0.89 dS m–1), was only increased significantly by FS.
As expected, adding seaweed to the starting materials failed to appreciably increase their contents in organic matter (OM), which were already high. Whereas OM was invariably below the 80% recommended minimum value in the gorse compost–seaweed mixtures, it exceeded that level in both pine bark and its mixtures.
The addition of C. baccata increased the total N content of pine bark, from very low levels (0.15%) to 0.31% (Fig. 2). However, adding the seaweed to gorse compost, which initially contained a much higher proportion of N (2.16%), failed to increase the content in this nutrient (Fig. 3).
Chemical and physico–chemical changes in the gorse compost by effect of the addition of seaweed in three different forms (mean ± standard deviation, n = 3). Different letters in each column denote significant differences between treatments at p < 0.05 as per Duncan’s test. FS fresh seaweed; WFS washed fresh seaweed; DWS dried washed seaweed
Adding the seaweed to the substrates also increased their total content in mineral elements. The greatest increase was obtained by adding FS to pine bark, which raised Mg, Na and N contents by 114%, 550% and 408%, respectively, relative to the control substrate. Adding the seaweed in any of the three forms to gorse compost only increased Na significantly; also, only FS increased K significantly (by 59%). The seaweed also increased the contents in Ca and P of pine bark (from 0.26% to 0.25 − 0.42% and 0.01% to 0.28 − 0.31%, respectively), but not those of gorse compost —which initially contained much higher levels of both elements.
A similar effect was observed on all soluble elements, the contents in which were increased in pine bark but, again, not in gorse compost owing to its initially high levels. In fact, the seaweed only increased K in the latter substrate, the effect being especially marked with FS. However, adding FS raised the levels of unwanted ions such as Na+ and Cl– above the tolerated limits for award of a European ecolabel (viz., 150 mg Na+ L–1 and 500 mg Cl– L–1).
As can be seen from Figs. 4 and 5, the seaweed did not significantly alter the contents in heavy metals of the substrates except for a significant reduction in Cu by effect of adding any of the three seaweed forms to gorse compost.
Differences in heavy metal contents in gorse compost according to the seaweed form used (mean ± standard deviation, n = 3). All values in dry weight. Different letters in each column denote significant differences between treatments (rows) at p < 0.05 as per Duncan’s test. FS fresh seaweed; WFS washed fresh seaweed; DWS dried washed seaweed
Differences in heavy metal contents in pine bark according to the seaweed form used (mean ± standard deviation, n = 3). All values in dry weight. Different letters in each column denote significant differences between treatments (rows) at p < 0.05 as per Duncan’s test. FS fresh seaweed; WFS washed fresh seaweed; DWS dried washed seaweed
Agronomic evaluation
Germination
Adding seaweed to the substrates had no phytotoxic effects (Table 4). FS increased germination with gorse compost from 73 to 90%. By contrast, adding the seaweed to pine bark had no effect on germination but increased root length; again, the increase was significant only with FS, which raised the level by 38%. Together with a slight increase in germination, this effect led to a Munoo–Liisa vitality index of 149% with the pine bark − FS mixtures.
Agronomic performance
Adding seaweed in any of the three forms to the pine bark substrate increased aerial growth in lettuce (Table 5) —by up to 728% with WFS. In addition, the pine bark − WFS substrate led to a significant increase in root mass (583%) and also in growth promotion index as a result. With gorse compost, however, only DWS led to increased aerial and root mass; also, the increase was less marked than with pine bark (240% in aerial mass and 232% in root mass).
Discussion
Ideal growing substrates usually consist of two or more substances that are mixed to ensure an optimum balance of physical, chemical and biological properties for the receiving crop. With fresh seaweed, the primary targets are biological stability to avoid cropping problems, and to improve some physical and chemical properties of the substrate. In this work, fresh (FS) and washed seaweed (WFS) were used untreated. On the other hand, dry seaweed (DWS) was obtained by removing most moisture, which made it somewhat unstable. According to Kaplan et al. (1980) microbial growth is optimal with a moisture content of 40–65%. This condition, however, is reversed when a crop is established and moisture increased through irrigation. In order to ensure that all mixtures would be stable and perform well as growing substrates, they were monitored for temperature and pH for 1 month prior to the experiment (Fig. 1). Temperature is the primary indicator of microbial activity (Liang et al. 2003; Miyatake and Iwabuchi 2006), an increase above ambient level suggesting the presence of microbes and hence instability. pH in immature materials tends to decrease through decomposition of labile organic matter and the resulting release of organic acids. Temperature and pH were measured on a weekly basis following turnover by hand to ensure aeration and sponginess, and the presence of oxygen, in the material. No significant increase in temperature or decrease in pH was detected for one month, so all mixtures were deemed stable and strong biological activity ruled out.
Unlike its chemical properties, the physical properties of a growing substrate are not easily altered. Therefore, in mixing components one should ensure that the resulting substrate will have a solid structure to achieve a sound balance in air and water for optimal plant development while avoiding anoxia and drought. These ideal conditions reflect in properties such as bulk density, porosity and water retention capacity (Barrett et al. 2016).
Phycocolloids are natural polymers of cell walls in seaweed and widely used industrially on account of their good rheological properties. Thus, they produce specific matrix polysaccharides forming grids capable of retaining large amounts of water (Verkleij 1992; García and Martel 2000; Lattner et al. 2003). The main phycocolloids in brown seaweed such as Cystoseira are alginates, which influence moisture distribution in soil (Nabti et al. 2017). Abad et al. (2001) established the optimum air- and water-related values for substrates as follows: aeration capacity 20–30%, easily available water 55–70% and total water retention capacity 60–100%. Neither of our substrates had properties falling in these ranges. In fact, both substrates exhibited excessive aeration capacity and poor retention of easily available water. However, adding seaweed improved water retention by pine bark, where it substantially increased total water retention capacity (EAW + WBC + UW) —the content in easily available water was raised to 18%, which is close to the minimum acceptable level proposed by De Boodt and Verdonck (1972): 20%. Adding the seaweed to pine bark also increased R, a measure of water availability at low pressures, with values above 3 kPa suggesting root anoxia by effect of excessive moisture and values below 1 kPa a deficiency in available water (Ansorena 1994). On the other hand, adding the seaweed had little effect on gorse compost, which is consistent with previous results of Illera-Vives et al. (2022a) with coir. This outcome may have resulted from increased adhesion of phycocolloids in pine bark by effect of its pore distribution (USDA Forest Service 1971), but no experimental confirmation for this hypothesis has to date been provided.
One of the benefits of adding seaweed to a growing substrate is that it increases the contents in nutrients such as N, K, Ca, and Mg, thereby allowing the requirements of some crops to be better fulfilled. Although seaweed has a modest P content, it can still be useful given the scarcity of natural phosphorus sources. In fact, some authors (e.g., Cordell et al. 2009) recommend using seaweed as a sustainable P fertilizer. This is especially so with P-poor substrates such as pine bark, addition of seaweed to which substantially increases its nutrient contents (particularly those in N, P, K, Ca, and Mg). On the other hand, adding the seaweed to a nutrient-rich substrate such as gorse compost only increased the content in K —and exclusively with the unwashed form and the result of introducing unwanted ions such as Na+ and Cl–.
One other very important chemical variable associated to the agricultural use of seaweed is its content in heavy metals. Thus, the components of seaweed cell walls and, particularly, alginates, allow them to accumulate large amounts of metals. One case in point is that of brown seaweed species such as Cystoseira, which are highly rich in alginates and can thus act as bioaccumulators (Davis et al. 2003). For this reason, seaweed to be added to growing substrates should never be collected from contaminated areas. The European Union has set maximum tolerated levels of heavy metals in substrates for award of its ecolabel (EU 2019/1009) and Spanish law (RD 506/2013) has issued the levels above which substrates should not be applied to edible horticultural crops. The heavy metal contents of all pine bark mixtures were well below the tolerated levels for edible horticultural crops (class A substrates) and remained so upon addition of the seaweed. On the other hand, the contents of gorse compost were initially high and not substantially altered by the seaweed.
The tests used here to establish the essential properties of the substrates were followed by others aimed at assessing the response of crops. One test evaluated the impact on germination as a measure of phytotoxicity (viz., the ability to have adverse effects on plant growth). Phytotoxic substances range from secondary metabolites formed as a result of microflora degrading fresh organic matter (Zucconi et al. 1981a, b) or the substrate containing substances that hinder plant growth (e.g., salts, heavy metals, or high concentrations of plant hormones). Compost maturity is widely assessed in terms of the index of Zucconi et al. (1981a), which is a measure of germination and root length relative to a control substrate. The index allows both high levels of phytotoxicity, which affect germination, and low levels, which affect plant growth, to be assessed (Zucconi et al. 1981b).
None of the studied mixtures exhibited any phytotoxic effects. Unlike the other seaweed forms, however, fresh seaweed (FS) substantially promoted root growth in pine bark and germination in gorse compost. Thus, FS increased the most critical factor in both types of substrates, namely: the relatively low germination capacity of gorse compost (73% vs 83% in pine bark) as the likely result of excessive salinity, and the less marked root growth in pine bark. One plausible explanation for this outcome is that the benefits of growth hormones present in seaweed become especially apparent under stressing conditions. In fact, some studies have shown their beneficial effects to be especially strong in suboptimal conditions for crop growth (Crouch and van Staden 1994; Craigie 2011; Kumar et al. 2020).
No phytotoxic effects of a potentially unstable additive such as seaweed were observed here. In fact, there were no appreciable differences in plant failure between substrates. Also, adding the seaweed in any form invariably increased crop mass. In fact, the seaweed considerably boosted plant growth in such a nutritionally poor substrate as pine bark; thus, WFS raised GeR to 763% in this substrate but only to 113% in gorse compost. In any case, the latter substrate was initially very rich in nutrients, so the improvement cannot be ascribed to their supply but rather to a biostimulating effect of the seaweed. Plant biostimulants are defined as “substances and materials other than nutrients and pesticides capable of altering physiological processes in plant in a way that may increase growth and/or stress resistance upon addition to plants, seeds or growing substrates” (du Jardin 2012). As a rule, plant responses to seaweed extracts are ascribed to their containing hormones (García and Martel 2000; du Jardin 2015) such as cytokinins, auxins and abscisic acid, and other, hormone-like substances (Stirk et al. 2003; Khan et al. 2009; Craigie 2011; Stirk et al. 2014; Battacharyya et al. 2015; du Jardin 2015). However, the hormone-like activity of seaweed extracts cannot be solely ascribed to their containing hormones (Battacharyya et al. 2015). According to Craigie (2011), other organic molecules including oligomers and polysaccharide elicitors can contribute to the effect. Also, according to Wally et al. (2013), the activity may be due to seaweed altering the biosynthesis of endogenous plant hormones rather than to the supply of exogenous hormones present in seaweed extracts. In addition, seaweed contains a wide range of agriculturally useful biological antioxidants such as polyphenols in the brown varieties, which protect plants from damage by free radicals and other oxidants; also, it possesses anti-microbial activity and is thus effective against some plant diseases (Cotas et al. 2020).
Conclusions
Our results confirm the starting hypothesis that Cystoseira baccata is an effective additive for growing substrates. In fact, this brown seaweed is natural, biodegradable, and pathogen- and weed-free; also, it showed no signs of instability or phytotoxicity during use even in the absence of a stabilization treatment. In fresh (FS) and dried form (DWS), the seaweed increased the total water retention capacity of pine bark by 20% and 27%, respectively. Application to pine bark, which is poor in nutrients, significantly increased its contents in P, K and Mg; however, it also increased the contents in other, undesirable elements such as Na, as well as the electrical conductivity (from 0.08 dS m–1 in the control substrate to 0.69, 0.12 and 0.27 dS m–1 with FS, WFS and WDS, respectively). By contrast, only FS increased salinity and the K content in gorse compost, which is rich in nutrients and salts.
The fresh seaweed form (FS) had no phytotoxic effects on a lettuce crop; rather, it acted as a biostimulant by boosting germination in mixture with gorse compost and led to longer roots in mixture with pine bark. All mixed substrates performed well on the lettuce crop. This was especially so with WFS, addition of which to pine bark increased lettuce aerial mass by 728% relative to the control substrate.
Data availability
The main data are shown in the manuscript. The raw data are available from the corresponding author on reasonable request.
References
Abad M, Nogueira P, Burés S (2001) National inventory of organic wastes for use as growing media for ornamental potted plant production: case study in Spain. Bioresour Technol 7:197–200
Adams JMM, Ross AB, Anastasakis K, Hodgson EM, Gallagher JA, Jones JM, Donnison IS (2011) Seasonal variation in the chemical composition of the bioenergy feedstock Laminaria digitata for thermochemical conversion. Bioresour Technol 102:226–234
Ansorena J (1994) Sustratos. Propiedades y caracterización. Ediciones Mundi-Prensa
Bădescu IS, Bulgariu D, Bulgariu L (2017) Alternative utilization of algal biomass (Ulva sp.) loaded with Zn(II) ions for improving of soil quality. J Appl Phycol 29:1069–1079
Bárbara I, Cremades J (2010) Mareas verdes, de su biología y problemática a su solución y aplicaciones. In: Rey-Méndez M, Lodeiros C, Fernández Casal J, Guerra A (eds) XII Foro dos recursos mariños e da acuicultura das rías galegas. Asociación Foro dos Recursos Mariños e da Acuicultura das Rías Galegas, España, pp 465–476
Barrett GE, Alexander PD, Robinson JS, Bragg NC (2016) Achieving environmentally sustainable growing media for soilless plant cultivation systems – A review. Sci Hortic 212:220–234
Battacharyya D, Babgohari MZ, Rathor P, Prithiviraj B (2015) Seaweed extracts as biostimulants in horticulture. Sci Hortic 196:39–48
Berthon J, Michel T, Wauquier A, Joly P, Gerbore J, Filaire E (2021) Seaweed and microalgae as major actors of blue biotechnology to achieve plant stimulation and pest and pathogen biocontrol – a review of the latest advances and future prospects. J Agric Sci 159:523–534
Biris-Dorhoi E-S, Michiu D, Pop CR, Rotar AM, Tofana M, Pop OL, Socaci SA, Farcas AC (2020) Macroalgae − A sustainable source of chemical compounds with biological activities. Nutrients 12:3085
Bourgougnon N, Bedoux G, Sangiardi A, Stiger-Pouvreau V (2011) Las algas: potencial nutritivo y aplicaciones cosméticas. In: Las algas como recurso. Valorización. Aplicaciones industriales y tendencias. Centro Tecnológico del Mar-Fundación CETMAR
Bullock C, Collier M, Convery F (2012) Peatlands their economic value and priorities for their future management – The example of Ireland. Land Use Pol 29:291–928
Cabrita ARJ, Maia MRG, Oliveira HM, Sousa-Pinto I, Almeida AA, Pinto E, Fonseca AJM (2016) Tracing seaweeds as mineral sources for farm-animals. J Appl Phycol 28:3135–3150
Caron J, Rochefort L (2013) Use of peat in growing media: state of the art on industrial and scientific efforts envisioning sustainability. Acta Hortic 982:15–22
Ceglie F, Bustamante M, Amara M, Tittarelli F (2015) The challenge of peat substitution in organic seedling production: optimization of growing media formulation through mixture design and response surface analysis. PLoS ONE 10:e0128600
Chapman H, Pratt PF (1997) Métodos de análisis para suelos plantas y aguas. Trillas, Mexico City
Cordell D, Drangert JO, White S (2009) The story of phosphorus: global food security and food for thought. Glob Environ Chang 19:292–305
Cotas J, Leandro A, Monteiro P, Pacheco D, Figueirinha A, Gonçalves AMM, da Silva GJ, Pereira L (2020) Seaweed phenolics: From extraction to applications. Mar Drugs 18:384
Craigie JS (2011) Seaweed extract stimuli in plant science and agriculture. J Appl Phycol 23:371–393
Crouch IJ, Smith MT, Van Staden J, Lewis MJ, Hoad GV (1992) Identification of auxins in a commercial seaweed concentrate. J Plant Physiol 139:590–594
Crouch IJ, Van Staden J (1994) Commercial seaweed products as biostimulants in horticulture. J Home Consum Hortic 1:19–73
Davis TA, Volesky B, Mucci A (2003) A review of the biochemistry of heavy metal biosorption by brown algae. Water Res 37:4311–4330
De Boodt M, Verdonck O (1972) The physical properties of the substrates in horticulture. Acta Hortic 26:37–44
du Jardin P (2012) The science of plant biostimulants – A bibliographic analysis ad hoc study report. European Commission Brussels. Available online at http://hdl.handle.net/2268/169257. Accessed June 2022
du Jardin P (2015) Plant biostimulants: definition concept main categories and regulation. Sci Hortic 196:3–14
EC Council 889/2008. Commission Regulation (EC) No 889/2008 of 5 September 2008 laying down detailed rules for the implementation of Council Regulation (EC) No 834/2007 on organic production and labelling of organic products with regard to organic production labelling and control
EN 13037 (2011) Soil improvers and growing media. Determination of pH. European Committee for Standarization. Brussels
EN 13038 (2011) Soil improvers and growing media. Determination of electrical conductivity. European Committee for Standarization. Brussels
EN 13039 (2011) Soil improvers and growing media. Determination of organic matter content and ash. European Committee for Standarization. Brussels
EN 13652 (2001) Soil improvers and growing media. Extraction of water soluble nutrients and elements. European Committee for Standardization. Brussels
EN 13041 (2012) Soil improvers and growing media – Determination of physical properties – Dry bulk density air volume water volume shrinkage value and total pore space
EU 2019/1009 Regulation (EU) 2019/1009 of the European Parliament and of the Council of 5 June 2019 laying down rules on the making available on the market of EU fertilising products and amending Regulations (EC) No 1069/2009 and (EC) No 1107/2009 and repealing Regulation (EC) No 2003/2003
Felipó M, Verdonck O, Cappaert I, De Boodt M (1979) Estudio de las propiedades físicas de los sustratos hortícolas. An Edafol Agrobiol 38:603–611
García G, Martel A (2000) Usos y aplicaciones de macroalgas microalgas y cianobacterias en agricultura ecológica. Actas IV Congreso SEAE. https://fci.uib.es/Servicios/libros/conferencias/seae/Usos-y-aplicaciones-de-macroalgas-microalgas-y.cid221515 (Accessed Jan 2022)
García-Fernández A (2020) La comunidad de "Cystoseira baccata" (Fucales, Ochrophyta) en el noroeste ibérico. PhD Thesis. Universidade da Coruña
Harb TB, Chow F (2022) An overview of beach-cast seaweeds: Potential and opportunities for the valorization of underused waste biomass. Algal Res 62:102643
Holmes S (2007) Growing media developments in the UK. Acta Hortic 819:23–26
Illera-Vives M, Labandeira SS, Fernández-Labrada M, López-Mosquera ME (2020) Agricultural uses of seaweed. In: Torres MD, Kraan E, Dominguez H (eds) Sustainable Seaweed Technologies. Elsevier, Amsterdam pp 591–612
Illera-Vives M, López-Fabal A, Fonseca F, López-Mosquera E (2022a) Production and evaluation of seaweed-containing plant growth adjuvant formulation. In: Ravishankar GA, Rao AR (eds) Sustainable Global Resources of Seaweeds. Springer, Cham, pp 451–488
Illera-Vives M, López-Fabal A, López-Mosquera M.E (2022b) Citoquininas en distintas algas marinas de Galicia para su uso como bioestimulante. Jornadas de fertilización, sustratos y fresón y otros frutos rojos. May 18 -20, 2022. Lugo (in press)
Kaplan D, Hartenstein R, Neuhauser E, Malecki M (1980) Physicochemical requirements in the environment of the earthworm Eisenia fetida. Soil Biol Biochem 12:347–352
Khan W, Rayirath UP, Subramanian S, Jithesh MN, Rayorath P, Hodges DM, Critchley AT, Craigie JS, Norrie J, Prithiviraj B (2009) Seaweed extracts as biostimulants of plant growth and development. J Plant Growth Regul 28:386–399
Kumar R, Trivedi K, Anand KGV, Ghosh A (2020) Science behind biostimulant action of seaweed extract on growth and crop yield: insights into transcriptional changes in roots of maize treated with Kappaphycus alvarezii seaweed extract under soil moisture stressed conditions. J Appl Phycol 32:599–613
Lattner D, Flemming H, Mayer C (2003) C-NMR study of the interaction of bacterial alginate with bivalent cations. Int J Biol Macromol 33:81–88
Liang C, Das K, McClendon R (2003) The influence of temperature and moisture contents regimes on the aerobic microbial activity of a biosolids compositing blend. Bioresour Technol 86:31–137
Metting B, Zimmerman WJ, Crouch I, van Staden J (1990) Agronomic uses of seaweed and microalgae. In: Akatsuka I (ed) Introduction to applied phycology. SPB, The Hague, pp 589–628
Miyatake F, Iwabuchi K (2006) Effect of compost temperature on oxygen uptake rate specific growth rate and enzymatic activity of microorganisms in dairy cattle manure. Bioresour Technol 97:961–965
Morel P, Poncet L, Rivière LM (2000) Les supports de culture. INRA éditions, Paris
Naasz R, Caron J, Legault J, Pichette A (2009) Efficiency factors for bark substrates: biostability aeration or phytotoxicity. Soil Sci Soc Am J 73:780–791
Nabti E, Jha B, Hartmann A (2017) Impact of seaweeds on agricultural crop production as biofertilizer. Int J Environ Sci Tech 14:1119–1134
Raviv M (2008) The use of compost in growing media as suppressive agent against soil-borne diseases. Acta Hortic 779:39–50
Raviv M (2013) Composts in growing media: what’s new and what’s next? Acta Hortic 982:39–52
RD 506/2013 Real Decreto 506/2013 de 28 de junio sobre productos fertilizantes. Boletín Oficial del Estado no. 164. pp. 51119–51207
Schiener P, Black KD, Stanley MS, Green DH (2015) The seasonal variation in the chemical composition of the kelp species Laminaria digitata, Laminaria hyperborea, Saccharina latissima and Alaria esculenta. J Appl Phycol 27:363–373
Schmilewski G (2008) The role of peat in assuring the quality of growing media. Mires Peat 3:1–8
Sharma SHS, Lyons G, McRoberts C, McCall D, Carmichael E, Andrews F, McCormack R (2012) Brown seaweed species from Strangford Lough: compositional analyses of seaweed species and biostimulant formulations by rapid instrumental methods. J Appl Phycol 24:1141–1157
Stirk WA, Novák O, Strnad M, Van Staden J (2003) Cytokinins in macroalgae. Plant Growth Regul 41:13–24
Stirk WA, Tarkowská D, Turecová V, Strnad M, Van Staden J (2014) Abscisic acid gibberellins and brassinosteroids in Kelpak® a commercial seaweed extract made from Ecklonia maxima. J Appl Phycol 26:561–567
Thomas R, Sheard R, Moyer J (1967) Comparison of conventional and automated procedures for nitrogen phosphorus and potassium analysis of plant material using a single digestion. Agron J 59:240–243
USDA Forest Service (1971) Bark and its possible uses. US Departement of Agriculture Forest Service. Forest Products Laboratory, Madison Wisconsin
Verkleij FN (1992) Seaweed extracts in agriculture and horticulture: a review. Biol Agric Hortic 8:309–324
Villares R, Carral E, Lorenzana F, Mosquera ME (2007) Drift-seaweed evaluation for fertilizer use in Galiza (Northwest Spain): tissue elemental characterization and site-sampling differences. J Sustain Agric 31:45–60
Wally OS, Critchley AT, Hiltz D, Craigie JS, Han X, Zaharia LI, Abrams SR, Prithiviraj B (2013) Regulation of phytohormone biosynthesis and accumulation in Arabidopsis following treatment with commercial extract from the marine macroalga Ascophyllum nodosum. J Plant Growth Regul 32:324–339
Zucconi F, Pera A, Forte M, De Bertoldi M (1981a) Evaluating toxicity of immature compost. Biocycle 22:54–57
Zucconi F, Pera A, Forte M, De Bertoldi M (1981b) Biological evaluation of compost maturity. Biocycle 22:27–29
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rodríguez, M.A., Illera-Vives, M., Labrada, M.F. et al. Improving growing substrates by adding the seaweed Cystoseira baccata. J Appl Phycol 34, 3177–3188 (2022). https://doi.org/10.1007/s10811-022-02846-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-022-02846-4