Abstract
Micro-algae play important roles in primary production, nutrient cycling, and applied biotechnology. Mico-algae are phylogenetically, morphologically and physiologically diverse and often easy to grow in cultures. Growth is the key parameter of viability and crucial for survival in situ and efficiency in vitro. Therefore, growth is often used in eco-physiological experiments to describe the fundamental niche and optimum conditions. In vivo chlorophyll-a fluorescence (IVCF) is a well-established proxy for the growth of micro-algae and is based on constant cell properties during exponential growth. High-throughput IVCF measurements have been applied for about 20 years for few, mostly green algal strains. Here, we tested the IVCF using microtiter plates on strains of four different algal phyla (Chlorophyceae, Cyanobacteria, Bacillariophyceae, Rhodophyceae) and compared the data with cell counts and chlorophyll contents. The microtiter plate reader based IVCF (MPR-IVCF) approach worked best for the diatom Cylindrotheca closterium and the cyanobacterium Synechococcus elongatus. Differences between IVCF and cell count-based growth rates were found for Chlorella vulgaris and Rhodella sp. The IVCF growth rates were lower than the growth rates based on cell counts in both cases, thus underestimating absolute growth rates. The MPR-IVCF method is robust in terms of light scattering and non-photosynthetic contamination but prone to changes in light regimes and also to aggregating algal cells. Thus, we recommend using the MPR-IVCF approach (1) for diatoms and cyanobacteria as an absolute growth rate measure, (2) for relative measures of growth rates, intra and interspecific during the same experiment, also for all algal phyla except Rhodophyta.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Phototrophic microorganisms such as prokaryotic (cyanobacteria) and eukaryotic micro-algae, are important primary producers as they form the phytoplankton, which accounts for half of the global net primary production (Longhurst et al. 1995; Zhao et al. 2005). Micro-algae driven communities play crucial roles not only for the carbon cycle (Raven and Falkowski 1999) but also for other nutrients, for example, nitrogen (Zehr 2011; Elbert et al. 2012). Furthermore, benthic biofilms and terrestrial biocrusts formed by microalgae stabilize the sediments and soils, thereby preventing erosion by interweaving and immobilizing particles (Belnap and Büdel 2016; Roche et al. 2017).
Micro-algae have developed many strategies to cope with harsh environmental conditions such as high UV (ultraviolet) and PAR radiation, drought, frost, high salinities or nutrient depletion. Therefore, micro-algae dominate the phototrophic community in nearly all aquatic (e.g. as phytoplankton) and non-higher plant vegetated terrestrial habitats (e.g. in biocrusts). The establishment and persistence of these organisms in a habitat are thereby based on the ability to grow at various and often even unfavorable conditions. The growth rate as a measure of biomass increase over time has to be higher than the rate of decay to form a stable population in situ. Therefore, the net growth rate is an integrative parameter which describes the overall ability of organisms to cope with all environmental (positive and negative) conditions present. Consequently, the growth rate is a reliable proxy for viability and is, hence, widely utilized in eco-physiological experiments to quantify the effects of single or multiple factors on the organism’s vitality (Sukenik 1991; Blair et al. 2014; Pálmai et al. 2020; Prelle et al. 2021). Abiotic factors can be manipulated to describe the fundamental niche, the importance of single genes, or the stress response for induction of metabolic pathways using cultivated algal strains or even whole communities (Giovagnetti et al. 2013). Such experiments aim to address a variety of questions about the basic functioning of micro-algal photosynthesis, secondary metabolite production, and ecophysiology.
Micro-algae produce a huge variety of primary and secondary metabolites, such as pigments and fatty acids. Therefore, they are utilized biotechnologically. Both, the biomass and metabolites, can be used as biofuels and food supplements, as well as in medicine or for wastewater treatment (Khan et al. 2018; Esteves et al. 2021). Such algae are usually grown in huge bioreactors to produce the required high biomasses. Optimal growth conditions have to be determined to gain maximum growth and the highest efficiency. The beginning and end of different culturing phases (lag, (exponential) growth, and stationary phase) must be detected, especially if certain metabolites have to be harvested. Thus, there is the necessity to measure growth over a longer period and at different culturing conditions in parallel to test for the growth parameters. High throughput miniaturized methods are needed for these applications to enable fast and reliable multifactorial experiments with sufficient parallels and replicates.
Growth rates of micro-algae can be determined by several invasive methods such as cell counts (microscopy or flow-cytometry), or as biomass parameters (chlorophyll-a, cell carbon, or dry mass) (Butterwick et al. 1982). The optimization of running bio-reactors offers the possibility of larger volumes for subsamples, which enable daily and replicated sampling for invasive methods. These invasive methods are not applicable with miniaturization to conduct many parallel single or multi-factorial tests before bioreactor usage. The most exact and robust method of quantification is cell counts with a microscope. However, this method is very time-consuming and allows for less parallelization in multi-factorial analyses.
The non-invasive methods for growth rate determination are based on the optical density or the in vivo chlorophyll-a auto-fluorescence (IVCF). IVCF has been used for a long time (Hannan and Patouillet 1972; Brand et al. 1981) and outperforms optical density with its selectivity against contamination by heterotrophic bacteria and fungi. These occur not only in environmental samples but also in uni-algal but non-axenic cultures (Gustavs et al. 2009). IVCF has been used as a proxy for micro-algal biomass in bioassays (Hannan and Patouillet 1972; Geis et al. 2000; Bi Fai et al. 2007; Fritzsche and Mandenius 2010; Suresh Kumar et al. 2014; Nagai et al. 2016) and growth experiments (Karsten et al. 1996; Gustavs et al. 2009; Skjelbred et al. 2012; Kitzing et al. 2014; Van Wagenen et al. 2014; Podevin et al. 2015; Berthold and Schumann 2020; Prelle et al. 2021). The basis of these measurements is the linearity of chlorophyll-a fluorescence in a growing culture under constant conditions (Wood et al. 2005). The advantages compared to cell counts or dry mass and other methods are, besides its non-invasive measurement, the easy and fast quantification. Culturing in microtiter plates allows very small volumes < 1 mL (Eisentraeger et al. 2003; Gregor and Maršálek 2005; Van Wagenen et al. 2014).
Micro-algae comprise photoautotrophic microorganisms of different phylogenetic origins and with strongly varying morphologies and physiologies. They occur as single cells, filaments, or in colonies, each with or without colored or colorless mucous envelopes. The surrounding of the cells can be cell walls of different thickness and composition ranging from peptidoglycans (cyanobacteria) to silica frustules (diatoms) (Domozych 2016). These aspects strongly influence the optical characteristics in terms of absorption, shading and scattering of incident light. Furthermore, micro-algae possess specific light-harvesting complexes which make them distinguishable by their specific absorbance and fluorescence spectra as well as ratios (Millie et al. 2002). The main antenna pigments define the spectrum of radiation absorbed and, therefore, the range of photosynthetic active radiation (PAR) that is utilized for photosynthesis. Contents of the pigments per cell can change during growth and life cycle as well as upon abiotic factors (light and nitrogen). Consequently, optical characteristics of the cells change (Griffiths et al. 2011). However, the common feature of all light-harvesting complexes is their central chlorophyll-a molecule to which the electrons are transported (Papageorgiou 2004).
Intracellular compounds, starch particles, polyphosphates, pyrenoids, or gas vesicles further display a strong variability in size and morphology additionally to the variety of light-harvesting complexes. Cell sizes also show conspicuous plasticity and range from very small pico-cyanobacteria (< 1 µm) up to several hundred µm in diatoms. Cell sizes can also vary massively within one species depending on the culturing conditions (Jezberová and Komárková 2007). The size of the cells and the inner structures play crucial roles in the scattering of light as smaller cells and structures scatter much more light than larger cells (Bryant et al. 1969).
Microtiter plates already have been applied in IVCF measurements with a variety of phototrophic organisms such as several green algae (Eisentraeger et al. 2003; Aruoja et al. 2009; Van Wagenen et al. 2014; Podevin et al. 2015; Haire et al. 2018), Pseudochattonella (Ochrophyta flagellate) (Skjelbred et al. 2012) and phytoplankton communities (Gregor and Maršálek 2004). Until now however, some important algal phyla such as Bacillariophyceae and Rhodophyta have not been investigated for miniaturization regarding their optical traits of cells. Most of the beforementioned studies lack information on scattering by the cell walls, frustules, and mucous envelopes. Thus, determination and quantification limits for the IVCF method are missing. Larger cells maintain the linearity of chl-a and cell growth even at very high IVCF values whereas smaller cells experience self-shading at lower IVCF values due to much higher scattering (Bryant et al. 1969). The isolated effects of the cell size and cell wall composition on the scattering signals have not been addressed so far. Furthermore, cross-checks with cell counts and pigment concentration have been rarely addressed. Van Wagenen et al. (2014) tested growth over time in different microtiter plate types with IVCF and optical density, and further checked respective cell numbers for some Chlorophytes. They found IVCF to be 10 times more sensitive than optical density, which was in congruence with data in much larger vessels (Gustavs et al. 2009). Limits of determination for other algae are scarce for microtiter plate measurements although important for the suitability of this method.
This study aims to give determination limits and practical advice for miniaturized IVCF growth measurements in microtiter plates with different algal phyla. Therefore, representatives of four different micro-algal groups (Chlorophyta, Bacillariophyta, Rhodophyta, and Cyanobacteria) were investigated. These algal groups vary not only in their pigmentation but also in cell size and surface properties (e.g. scattering by silica frustules). The main focus of this methodological investigation was on device settings, material choice, scattering effects, and growth rate calculations with both, in vivo chlorophyll-a fluorescence and cell counts. The pigment-signal per cell ratios and self-shading thresholds under continuous ambient conditions were addressed by cross-checks with cell counts.
Materials and methods
Organisms and culture conditions
Four micro-algal strains with different pigmentation and morphological characteristics were investigated in this study. The green alga Chlorella vulgaris SAG 211-11b (diameter: 4 µm) was grown in Bold’s basal medium with triple nitrate and vitamins (3 N-BBM + V) (Starr and Zeikus 1993). The cyanobacterial strain Synechococcus elongatus PCC 6301 (width: 0.8–1.5 µm; length 3–4 µm) was cultivated in BG 11 (Sigma-Aldrich, Germany) and the red alga Rhodella sp. KA 4072 (12 µm diameter) was grown in sterile Baltic Sea water salted with artificial sea salt (hw Marinemix professional, Wiegandt GmbH, Krefeld, Germany) to an absolute salinity of 33 SA and enriched with full-strength Provasoli’s nutrients (Provasoli 1968). The diatom Cylindrotheca closterium KABB1 (width: 2.5–3 µm; length: 35–45 µm) was cultured in sterile Baltic Sea water salted to a salinity of 33 SA and enriched with f/2 medium (Guillard 1975). All cultures were grown at 20 °C and 38 μmol photons m–2 s–1 on a 16 h light: 8 h dark cycle (Lumilux Deluxe Daylight fluorescent bulbs, Osram, Germany). Photon flux density was measured with a Li-Cor LI-250 light meter (sensor model: us-SQS/L, Germany) always at the surface of the plates and the temperature was monitored by HOBO UA-002–64 (oneset, HOBO pendant temperature/light, 64 K, Onset, USA; with 6 h interval measurements).
Pre-cultures were set up in disposable growth cuvettes (Licefa, Germany) to ensure the log phase growth of the different algae. Therefore, the inoculated cuvettes were measured in a growth fluorometer (Hansa-Tech, England, (Karsten et al. 1996), as described in (Gustavs et al. 2009). The inoculum volume was adjusted to the fluorescence signal. The initial IVCF signal was considered as high enough with double the blank value of the respective medium to guarantee sufficient biomass for initiating growth and also to reduce self-shading as much as possible. The IVCF was measured every 24 h and the starting point of the growth experiment was chosen in the early log phase.
Scattering and self-shading
A quality control experiment was performed with bleached algal cells (bleaching process in 35% H2O2 (v/v) for 10 min in 100 °C water bath and subsequent centrifugation for 15 min at 1,520 × g in a tabletop centrifuge (Heraeus BioFuge Pico, Germany) to evaluate scattering of dead cells. Different dilutions of bleached cells were used for all strains.
Additionally, a dilution series with living C. vulgaris was used to test for the potential IVCF signal range. A series of 48 points was used, containing an equidistant 24 points low concentrated (0.24–3.48 × 106 cells mL−1) and an equidistant 24 point high concentrated (5.06–57.13 × 106 cells mL−1) dilution series. The wells were emptied after each measurement, the content preserved in 5% final concentration Lugol’s solution (Carl Roth, Germany) and counted (see cell counting procedure). Media and de-ionized water were used as controls. Bleached and intact cells were transferred to disposable 24 well cell culture plates for additional fluorescence measurements.
In vivo fluorescence measurements
All IVCF growth measurements were performed with a multi-detection microplate reader (SpectraMax iD3, Molecular Devices, USA). The setup of this reader was adjusted to well-scan program, 5 reading points as well as top and bottom reading mode. Different mixing modes were tested of which auto-mix for 10 s with orbital shaking performed best and was used in all subsequent assays. Excitation and emission wavelengths of 444 and 680 nm, respectively, were used to determine IVCF as relative fluorescence units (RFU). A cut-off filter at 630 nm was used and auto mode for the gain performed best with low and high IVCF values alike.
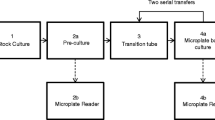
A defined amount of the pre-cultured cell suspension adding up the medium to 2 mL volume per well, which resulted in IVCF signals at approx. twice the medium blanks was inoculated in the 24 well cell culture plates (24 well Costar 3526, USA). The algae were then grown in the closed (lid) micro-titer plates and growth recorded for 12 days. The fluorescence signals were measured every 24 h. All conditions remained unchanged during the experiment. Twelve replicates were continuously measured throughout the twelve days. The other triplicates were sequentially sampled for cell counts (replicates 1 to 3; each one per day) (Fig. 1).
Sampling scheme for growth rate determination. Two plates were used for each algal strain and all 48 wells inoculated at once. The first three replicates were removed by pipetting each respective day for cell counts and the wells were kept empty henceforth. The fourth replicate of each day stayed untouched for continuous measurements over the 12 days
Cell counting procedure
The experiment was set up in two 24 well plates for each strain tested. Each day, the algal suspension was removed by pipetting from three out of four wells of one row (1st day, 1st row; 2nd day, 2nd row; etc., Fig. 1). Each harvested well was separately fixed in Lugol’s solution (final concentration: 5% (v/v)). The Lugol-preserved samples were counted either in sedimentation chambers (big cells; Rhodella sp. and C. closterium), in a Bürker blood cell counting chamber of 0.1 mm height (Fein-optik, Germany) for medium-sized cells; C. vulgaris or in a Neubauer Improved chamber of 0.01 mm height (Paul Marienfeld GmbH & Co. KG (very small cells; S. elongatus) at a 400 × magnification (Olympus CH20, Japan). At least 400 cells per subsample were counted. The samples were diluted at high cell densities when the cells formed several layers in the counting chamber.
Chlorophyll a extraction
The chlorophyll-a growth experiment with C. vulgaris was conducted using three 550 mL disposable cell culture flasks (Greiner BioOne GmbH, Germany) filled to 350 mL with 3 N-BBM + V medium. The log-phase culture was transferred in low density to this medium and grown for 12 days. Every day, 10–40 mL of culture were taken and filtrated onto glass-fiber filters (GF92, Whatman, Germany), then ground and extracted in hot 96% (v/v) ethanol for one hour at 78 °C and measured according to Ritchie (2008).
Growth rate calculation
For the growth rate calculations of IVCF, diagrams were plotted with relative fluorescence units (RFU) data versus days to show the signal changes over time. The RFU values were checked on a logarithmic scale to identify the exponential growth phase visually as a linear slope. Only the days and values which form the log-linear graph were taken for the calculation of the maximum growth rate (µ). Thus, lag- and stationary phase data points were excluded. The maximum growth rate (µ) was calculated with equation (A) (Guillard 1973). A longer lag time was excluded, so that day 0 was not always the day of inoculation, but the beginning of exponential growth.
where Fday 0 = initial fluorescence unit, Ft = fluorescence units after t days, µ = growth rate per day and t = days.
Equation (A) calculates the RFU (= F) over the days of maximum growth. The µ values were optimized using the Solver module of Microsoft Excel. First, a µ has to be given as a rough estimate, which serves as the variable to be solved by stepwise small changes. The target is the sum of the mean normalized deviation squares of measured and calculated values, which has to be minimized by the Solver. Thus, µ is finally best fitting the measured values. All the calculations were performed for each replicate separately and the growth rates from these replicates were averaged. The same procedure was done for the cell numbers.
Results
Device settings and material choice
Log-phase cultures of C. vulgaris were used for testing device settings and material. In a pre-experiment, sealing foils suitable for fluorescence measurements were tested to protect cultures against evaporation and contamination during measurement. The experiment was conducted with three kinds of seals (VIEWseal 676,070, AMPLIseal 676,040 and EASYseal 676,001, Greiner Bio-one, Germany) and without seals. The EASYseal 676,001 performed best in terms of the lowest signal reduction. However, the percentage of reduction in signal increased with rising cell density and signal from -1.7% (signal slightly increased) at 0.2 × 106 cells mL−1 up to 26% of signal reduction at 1 × 106 cells mL−1 (approx. 20% reduction in the median, suppl. Fig S1).
Scattering
The potential scattering effects of the medium and bleached cells were neglectable throughout the experiment compared to the IVCF signals. The mean blank values for each medium were 6,785 and 2,445 RFU (3 N-BBM + V), 8,765 and 3,112 (BG11), 8,625 and 3,341 (F/2), 9,275 and 3,706 (PE) for top and bottom reading mode, respectively. On average, the noise derived from 12 medium blanks was 2.7 times higher in the top reading mode than in the bottom reading mode, which resulted in lower signal-to-noise ratios at low cell numbers for the top reading mode (Table 1). The scattering was very low for all tested algae, even for the diatom with its silica frustules (Table 2).
The signal per cell ratios were 0.11 and 0.19 (C. vulgaris growth experiment), 0.01 and 0.01 (S. elongatus), 0.03 and 0.03 (Rhodella sp.), 0.16 and 0.23 (C. closterium) for top and bottom reading mode, respectively. The IVCF signal-to-noise ratios were calculated from the first day of measured exponential growth (Table 1). Chlorella vulgaris and C. closterium exhibited the highest signal-to-noise ratios as well as the highest IVCF values per cell. The limits of determination (LOD) and quantification (LOQ) were lowest for C. vulgaris, S. elongatus, and C. closterium with bottom reading mode, whereas Rhodella sp. exhibited lower LOD and LOQ with the top reading mode due to the higher signal per cell ratio. The LOD and LOQ were calculated from the medium blank-derived detection and determination limits of RFU and then divided by the median IVCF-signal per cell. All strains exhibited IVCF signals above the LOQ at the first day of exponential growth.
The linearity of RFU per cell number was checked using a 48 steps dilution row of C. vulgaris. The top-reading mode achieved signal linearity up to 6.1 × 106 RFU and 13.9 × 106 cells mL−1 while the bottom reading mode only gave linear results up to 0.84 × 106 RFU and 3.48 × 106 cells mL−1 (Fig. S2). Therefore, the top read mode data was chosen over the bottom read data in the growth experiments.
Growth experiment
The growth experiments showed conspicuous differences in the suitability of the IVCF for absolute algal growth rate measurements for different species. The general trend of exponential growth was well reflected by the IVCF signal values over time (Fig. 2) for C. vulgaris. The linearity of chlorophyll-a fluorescence and cell numbers during the exponential growth (R2 higher than 0.97 for all 4 tested algae) is shown in Fig. 3. However, the growth rates and doubling times differed between IVCF and cell number growth rates, both for C. vulgaris and Rhodella sp. (Table 3). The growth rates of S. elongatus and C. closterium showed more or less the same growth and doubling rates for cell counts and IVCF, respectively (Table 3).
We additionally tested the growth rate for C. vulgaris in a follow-up experiment with higher culture volumina to photometrically test the chlorophyll-a content as a proxy for growth. The growth rate was 1.9 times higher compared to IVCF and equal to cell numbers growth rates. The maximum growth as reflected in chlorophyll-a was also found during days 4 and 7, as with cell numbers and IVCF as these were the only time points with a linear increase on logarithmic scales.
Discussion
Material choice
The tested multi-detection microplate reader is suitable for high-throughput growth measurements of microalgae under different controlled and manipulated environmental conditions. The advantages of this technique are reduced resources such as space, chemicals, and inoculum biomass, i.e. miniaturaziation. Some cautions and limitations, however, originated from the experimental data. The absolute growth rates may deviate from aeriated cultures as no aeriation is possible in micro-titer plates, which influcences the availability of CO2, nutrients and, with higher cell densities, the light reaching the algae due to self-shading (Borowitzka 1998). We do not recommend sealing foil on top of the microtiter plate remaining for a whole growth experiment. Droplets formed due to condensation at the inner surface of the sealing foil during growth over 12 days, which increased the scattering and lowered the signal (data not shown). Additionally, the relative signal reduction by the sealing foil appeared to be shifting steadiliy so that the growth rate may be hampered systematically. Therefore, the main experiment presented here was performed without seals. Lids were taken off for measurements as they also reduced the signals and may damage the sample carrion in some devices. If a sealing foil is necessary to prevent evaporation or contamination during long-term experiments, the foil should be taken off for measurements and be replaced by a new one afterwards.
In our study, the red alga Rhodella sp. exhibited major changes in the IVCF signal per cell after transferring from pre-culture vessels to the 24-well-plates. These rather drastic IVCF changes may result from an altered light regime in the plates due to enhanced scattering by the transparent micro-titer plates. Such optical changes for the algae probably occurred even though the same PFD was applied and measured at the culturing place for both, pre-culture and micro-titer plates. Small changes in the light regime can induce subsequent alteration in the pigmentation, even within one day (Gustavs et al. 2009). Van Wagenen et al. (2014) used dark or white plates for measurements to avoid scattering by the plate itself during incubation and recordings. Dark microtiter plates have one disadvantage against transparent plates as the former require light sources placed directly above or below the plate homogeneously irradiating each well to avoid shading by this type of plate. Bright plates have a rather equal light distribution in the wells as light can pass through the walls and the plate itself does not shade the wells (Van Wagenen et al. 2014).
Device settings
The device settings were optimized for chlorophyll-a IVCF signals and the dilution row proved the linearity of the signals over a broad IVCF range. We assume that the selected wavelengths for excitation, emission, and cut-off filters are suitable to detect reliable IVCF values. However, other studies used slightly different excitation and emission wavelengths (e.g. Gregor and Maršálek 2004; Skjelbred et al. 2012; Van Wagenen et al. 2014), which also proved suitable. Nevertheless, algae with light-harvesting complexes containing mainly pigments, which absorb green light best, are not optimally excited by these published settings. The IVCF signal of such cells is therefore much lower compared to chlorophyll-a-rich organisms. This is one reason for S. elongatus to have very low IVCF values per cell (Schubert et al. 1989), but also the smaller cell size matters. Consequently, higher cell numbers are needed to get above the detection and determination limits, which in turn affects the duration of experiments as self-shading may occur much earlier. A way to overcome the blue excitation gap for cyanobacteria and the relatively low blue excitation of red algae (French and Young 1951) would be the application of different excitation and cut-off filter wavelengths to get the maximum signal strength. The growth fluorimeter setup introduced by (Karsten et al. 1996) applied blue LEDs without a specific excitation narrow-band cut-off filter as the LEDs already emit a range of approx. 100 nm for excitation. A red filter has been used with a peak transmission at approx. 630 nm. The linearity of IVCF-signal to cell numbers at the same physiological status was proven for different algal strains (Gustavs et al. 2009).
The fluorescence measurement of 5 micro-sites with each 1 mm in diameter of the evenly distributed cell population within one 2 mL well proved suitable for the unicellular algae. The areas do not overlap and cover parts of the whole well. Micro-algae that form small aggregates (few cell layers) would need a larger integrated area to give more reliable results, which often is not possible in microplate-readers due to limitations in the optical construction. Therefore, the method is not suitable for colonies and aggregate-forming algae (Gregor and Maršálek 2004) when these form few but large patches instead of being evenly distributed. Information on the well scan settings is not given in other studies, although several publications are available for the IVCF-microtiter plate method (Chen et al. 2012; Skjelbred et al. 2012; Van Wagenen et al. 2014; Podevin et al. 2015). The plates have to be shaken before each measurement for algal suspensions, as they tend to aggregate or sediment unevenly in the wells when transported. In contrast to the pelagic algae tested here, benthic algae should not be shaken since they form more uniform, thin, and stable biofilms at the bottom of the wells.
The signal of the top read mode was proportional to the cell number for four times higher cell numbers than the bottom read mode in the dilution series. The bottom read mode showed overall much less signal strength and a slope less than half as steep as the top read, which indicates sensitivity towards scattering and self-shading effects. Therefore, the bottom read mode should only be used with low cell densities. In addition, the settings might be adjusted for algae settling at the vessel bottom, which must be tested in pre-experiments. However, in the present study none of the tested algal species showed such a behavior. Hence, in theory, the cell density decides on the reading mode. Both reading modes gave similar results in the growth experiments, which aim to start with cell numbers just above the limit of quantification and often do not reach potential signal saturation even at the last days of the experiment.
Other potential factors such as nutrient limitation or self-shading begin to lower the signal increase at much lower cell numbers (1.2 × 106 cells mL−1) than what was tested in the dilution row in this study (57 × 106 cells mL−1). The upper cell density to which exponential growth occurs depends on the organism used and can vary between 0.15 × 106 cells mL−1 (Skjelbred et al. 2012) and 10 × 106 cells mL−1 (Chlamydomonas, (Haire et al. 2018).
Lightpath aspects
The sensitivity of IVCF was at least twice as high as optical density measurements for green algae (Van Wagenen et al. 2014) and diatoms (Butterwick et al. 1982). That allows for low cell numbers as a starting point of experiments. Further, IVCF is very robust against non-auto-fluorescent contamination, for example, by heterotrophic bacteria, which on the other hand would interfere with optical density measurements. The absorption of pigments may also interfere with optical density measurements and does not necessarily show linearity with biomass (Griffiths et al. 2011), which is excluded in IVCF as well. However, some limitations of IVCF measurements result from technical features such as the geometry of the light path, which in our case went from top to bottom or vice versa. Such a construction is prone to the uneven water level towards the edge of the well. This effect gets more relevant with decreased vessel size and hinders the usage of a large part of the well for measurements, which in turn, enhances the effects of the uneven distribution of algal cells.
Best results would be achieved by applying the bottom excitation and bottom measurement as it is depending only on the lowest layers of algal biomass at the bottom of the culturing vessel. The excitation and emission do not have to pass through the whole vessel, thus they are less scattered or absorbed by particles or pigments and lead to more reproducible signals. This is especially important as cells specifically reduce certain wavelengths by their pigmentation (reviewed in Lehmuskero et al. 2018). The more suitable light paths require a different optical construction and geometry, which excites and measures at a 90 degrees angle with excitation from the side of the vessel and detection from below (Karsten et al. 1996), which cannot be achieved in microtiter plates. Another option would be the excitation from a 45 degrees angle below and detection from another 45 degrees angle to achieve 90 degrees optical difference between excitation and emission. Nevertheless, a larger area for emission detection is needed to be less dependent on the microscale distribution of the algal cells within the vessel. Therefore, the growth fluorimeter of (Karsten et al. 1996) shows a better technical concept. However, this device can only be used for individual samples but not for high-throughput parallel measurements.
The IVCF methods are reproducible and best suited for unicellular solitary living algae (Gregor and Maršálek 2005) or for filamentous forms (Karsten et al. 1996), which either distribute evenly over the whole vessel or lie in one or few layers at the bottom. Thus, self-shading is reduced to a minimum under these types of algal morphology. Aggregate- or colony-forming algae are challenging due to self-shading, which affects both, the pigment biochemistry and fluorescence signal path. Under few cell layers conditions, the self-shading reduces most of the incident light, especially those wavelengths collected by the pigments (Jørgensen and Des Marais 1988). Colonial algae furthermore distribute unevenly in the culturing vessel. Point measurements in these vessels took place at several (in our case: 5) spots of each 1 mm radius to not overlap with each other. This led to many blind spots in the culturing vessels. Unevenly distributed algae (e.g. diatom aggregates or cyanobacterial colonies) therefore showed a very high deviation between the individual measurements, depending on the spatial position of these aggregates (Gregor and Maršálek 2005). Again, a technical approach consisting of vessels (or at least a larger part of the vessel) integrating emission measurements could overcome this biooptical problem. However, self-shading by multiple cell layers would still be problematic. Besides colonial algae, some filamentous ones (e.g. members of the streptophyte Klebsormidium sp.) also could be difficult to handle as these strains form bigger aggregates in the culturing vessel and sometimes even grow at the sidewalls outside the measuring area. Attempts to avoid this obstacle led to a growing and measuring IVCF of these algae on thin agar layers, which gave reproducible results (Kitzing et al. 2014; Donner et al. 2017; Borchhardt and Gründling-Pfaff 2020). However, the log phase growth was not cross-checked with cell counts in these publications. The growth rates in the log phase reported using that setup still were reproducible and comparable to each other as well as to other studies in liquid cultures (Karsten et al. 2010) although the algae often grew in many layers towards the end of the measurement period.
Suitability
Chlorella vulgaris
Chlorella vulgaris was chosen as a model organism to test for the linear range between cell numbers and RFUs. First, it does not form aggregates, second, it is growing fast, and third, it has with up to 3% a very high chlorophyll-a content per cell (Kong et al. 2014). The detection and determination limits were calculated by the median signal per cell in the growth experiment (~ 16,000 and ~ 3,700 cells mL−1 for top and bottom, respectively), and are comparable to values that have been reported by (Gustavs et al. 2009; Skjelbred et al. 2012).
Chlorella vulgaris exhibited growth rates in the IVCF approach only half as high as based on cell numbers. Growth rates of up to 1.1 day−1 under optimal growth conditions for C. vulgaris were reported in the literature (Blair et al. 2014). Therefore, the growth rate derived from cell numbers (1.25 day−1) in the present study fully confirms the literature and, hence, can be considered as reliable. (Skjelbred et al. 2012) reported low absolute growth rates due to limited gas exchange in the wells. This effect does not explain the discrepancy between the growth rates of IVCF and cell number, as both were determined from similar microtiter plate wells. Most probably, a reduced chlorophyll-a content at increased irradiance due to very fast growth caused the differences as suggested by (Seyfabadi et al. 2011), which could result in a prolonged chromatic acclimation phase of cells inside the microtiter plates.
The growth experiment IVCF signals were always below the point of self-shading. That is confirmed by the linearity of cell number and RFU signal during exponential growth (Fig. 2). We started another experimental run and analyzed the extracted chlorophyll-a content, and from this proxy calculated µ, which was slightly lower (1.08 day−1) than the cell based growth (1.25 day−1) but not as strongly reduced as with IVCF (0.58 day−1). This result has to be treated cautiously as the chlorophyll-a growth experiment took place in much larger disposable cell culture flasks with more air above. Also, cells can divide into two or more daughter cells, which depends on the growth conditions, e.g. light regime (Sorokin and Krauss 1959; Zachleder et al. 2021). This divergence of cell number increase and chlorophyll-a increase can only be resolved by other methods as with flow cytrometry pulse width measurements (Chioccioli et al. 2014). IVCF may lead to an underestimation of cell number growth rates in C. vulgaris.
Synechococcus elongatus
The RFU per cell ratio is very low in cyanobacteria due to their generally small cell size and very low excitability of their main accessory pigments with this setting (e.g. (Gustavs et al. 2009). Further, the main light-harvesting pigments are phycobiliproteins, which mainly absorb green, yellow or red light and are not well excited by blue light (MacColl and Guard-Friar 1987). Therefore, cell number limits of detection and quantification were at least 24 times higher compared to C. vulgaris. Again, the bottom read mode performed much better in terms of signal to noise, LOD as well as LOQ. Growth rates and doubling times were equal between the IVCF and cell number approach, and suggest the suitability of this method for S. elongatus.
However, the absolute growth rate is very low compared to other studies (e.g. Binder and Chisholm 1990; Sakamoto and Bryant 1999). Lowered growth rates for cyanobacteria in microtiter plates have already been reported and explained by the missing aeration and mixture of the culture (Skjelbred et al. 2012), which is usually done when working with this strain and other related cyanobacteria. Despite their small size and potentially high scattering, the high cell numbers (up to 6 × 106 cells mL−1) found in our study did not affect the scattering and, thus, this aspect can be neglected. ICVF-signal per cell remained constant over the complete experiment time. Although chromatic acclimation of cyanobacteria and several Synechococcus strains is well documented (e.g. MacColl and Guard-Friar 1987; Apt et al. 1995; MacColl 1998; Stadnichuk et al. 2015; Śliwińska-Wilczewska et al. 2020) it did not seem to play a major role in our study. That indicates the proportionality of the signal up to the maximum tested cell number (7.8 × 106 cell mL−1). Thus the IVCF method is in principle suitable for quantification of growth also of this strain but the cultivation in microtiter plates seems to have tremendous negative effects on cyanobacterial growth rates.
Rhodella sp.
Rhodella sp. has much larger cells than S. elongatus but the RFU signal per cell ratio is also low in this red alga due to its pigmentation (French and Young 1951). Comparable to cyanobacteria, the phycobilins are the main light-harvesting antennae in red algae, which are not excitable by blue light (Gantt et al. 2003). Therefore, the cell number LOD of Rhodella sp. was approx. 10 times higher than that of C. vulgaris. Rhodella sp. exhibited a LOD only half of the value determined for S. elongatus, despite a cell volume roughly 90 times larger. However, the LODs ratio of S. elongatus and Rhodella sp. well resembled the per cell chlorophyll-a contents (Desquilbet et al. 2003; Śliwińska-Wilczewska et al. 2020). On a volume basis, Rhodella sp. has about 50 times less chlorophyll-a per µm3 cell volume than S. elongatus.
The signal of the experimental cultures dropped during the first days although the pre-cultures were already acclimated in small vessels (20 mL) under the same incident light conditions as the microtiter plates. Within the first 24 h, the signals even declined below the medium blank values in all 48 parallels. Perhaps the absorption of the cells exceeds the fluorescence signal at low cell densities and at unintended shifts in the light climate from pre-culture to the experimental setup. Further, a chromatic acclimation towards increased PFD could be induced and, thus, a reduction in pigment content per cell (Gantt et al. 2003). Other strains of Rhodella sp. have strongly varying (factor 6) chlorophyll-a contents depending on the quantitative light conditions (Ritz et al. 2000). Even though Rhodella sp. was grown in low-density culture, we assume that self-shading already occurred in the pre-culture, which led to the drop in IVCF-signal.
Rhodella sp. gave dissimilar growth rate results applying the different IVCF and cell count measurements. The IVCF-signals increased about one-third less than the cell numbers. The growth rate determined by the IVCF, which was taken from day 6 on, remained low and could not parallel with the growth of cell numbers pointing to a reduction in chlorophyll-a content per cell instead of recovering over time. In red micro-algae, the acclimation towards shifted light regimes can take as long as 8 to 10 days (Ritz et al. 2000), which was achieved only at the end of our experiment.
The consequences of these findings are that the IVCF method is not suited for the determination of growth in the red alga Rhodella sp. and probably also not suitable for other Rhodophyta. The pigmentation and the strong response of the chlorophyll-a IVCF-signals to slightly changed light conditions make them prone to self-shading effects already at low cell densities. Much lower cell densities are necessary, which results in signals below the determination limit of this method. Therefore, IVCF cannot be recommended for absolute growth determination in red algae.
Cylindrotheca closterium
Cylindrotheca closterium has a high and stable RFU per cell ratio (Fig. 3) that resulted in a high signal-to-noise ratio already at low cell densities. Furthermore, the cell-related LOD was as low as that of C. vulgaris with only a few thousand cells per ml. Diatoms contain very efficient light-harvesting complexes with fucoxanthin as accessory pigment and, therefore, are easily excited by blue light (Kuczynska et al. 2015). The low scattering and high signal per cell provided the possibility to start growth experiments with very low cell numbers, which generally allows for more days of measurement as self-shading occurs later. In our study, the stable signal per cell ratio indicated no self-shading over the complete growth measurement period. Cylindrotheca closterium also exhibited the highest IVCF-signals per cell, which were about 1.2 times higher than those of C. vulgaris.
The IVCF measurements showed an approx. 13% higher growth rate compared to the cell counts. The latter is in accordance with (Liang et al. 2005) but the overestimation with IVCF is relatively low and the method is suitable for growth rate determination of this particular diatom. This needs to be tested and confirmed for oder diatoms, especially ones that are more silicified or exhibit other morphologies (e.g. members of the Centrales). Karsten et al. (1996) have reported that the IVCF approach is generally well suited for diatoms in general because they efficiently absorb blue light not only with chlorophyll-a but also with fucoxanthin, which results in a strong fluorescence signal.
In summary, the IVCF measurements in microtiter plates for the four tested algal taxa were only suitable for absolute growth determination in the cyanobacterium S. elongatus and the diatom C. closterium but not for the green alga C. vulgaris and the red alga Rhodella sp. The latter two gave very different results in their growth rates based on IVCF or cell numbers. Few studies looked at the relation of IVCF to cell numbers either with dilution series or as a measure over time in microtiter plates (Eisentraeger et al. 2003; Skjelbred et al. 2012; Van Wagenen et al. 2014). Gustavs et al. (2009) provided such data for the larger-vessel growth fluorimeter, but for microtiter plate IVCF these results were mostly missing and proved the need for individual cross-checks for each strain. The time-intensive controls still are outcompeted by the possibility to test multiple parallels at the same time in experiments.
All results and considerations led to several conclusive recommendations: (1) pre-experiments are essential to conduct quality measures for every new algal strain to check the growth over time to assure signal stability. (2) the method is well suitable for uni-cellular pelagic or adhering unicellular or filamentous benthic algae. Severe quality management is necessary when working with green algae and we do not recommend applying our settings of IVCF for Rhodophyta and aggregate- or colony-forming strains. (3) However, the IVCF in microtiter plates can be applied for all algal groups for relative growth comparison. Absolute growth seems to be underestimated in some cases. (4) scattering by the cell walls does not show a major impact, irrespective of the cell wall material, and can be neglected. (5) for measurements, we recommend using no sealing foils nor lids. Between measurements, lids or seals should be applied to prevent media contamination and evaporation. (6) The IVCF in microtiter-plates is best suitable for repeated measurements on quality checked organisms (otherwise: pre-experiments) to determine the lag and exponential growth phase as well as the stationary phase (capacity) under different culturing conditions (temperature, salinity, nutrients, light). Caution is necessary when it comes to changes in the light regime as this factor directly and strongly influences the pigmentation with consequences for the fluorescence signal. (7) low cell density is crucial to avoid IVCF signal reduction during measurements. Very dense cultures taken from large growth vessels have to be carefully diluted before measurement.
If these recommendations are carefully addressed the IVCF approach is a minituarized, non-invasive and suitable method which allows to screen numerous new algal strains for their ecophysiological responses and hence their biotechnological potential.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Apt KE, Collier JL, Grossman AR (1995) Evolution of the phycobiliproteins. J Mol Biol 248:79–96
Aruoja V, Dubourguier HC, Kasemets K, Kahru A (2009) Toxicity of nanoparticles of CuO, ZnO and TiO2 to microalgae Pseudokirchneriella subcapitata. Sci Total Environ 407:1461–1468
Belnap J, Büdel B (2016) Biological soil crusts as soil stablizers. In: Weber B, Büdel B, Belnap J (eds) Biological soil crusts: An organizing pricinple in Drylands. Springer, Cham, pp 305–320
Berthold M, Schumann R (2020) Phosphorus dynamics in a eutrophic lagoon: Uptake and utilization of nutrient pulses by phytoplankton. Front Mar Sci 7:1–15
Bi Fai P, Grant A, Reid B (2007) Chlorophyll a fluorescence as a biomarker for rapid toxicity assessment. Environ Toxicol Chem 26:1520–1531
Binder BJ, Chisholm SW (1990) Relationship between DNA cycle and growth rate in Synechococcus sp. strain PCC 6301. J Bact 172:2313–2319
Blair MF, Kokabian B, Gude VG (2014) Light and growth medium effect on Chlorella vulgaris biomass production. J Environ Chem Eng 2:665–674
Borchhardt N, Gründling-Pfaff S (2020) Ecophysiological response against temperature in Klebsormidium (Streptophyta) strains isolated from biological soil crusts of Arctic and Antarctica indicate survival during global warming. Front Ecol Evol 8:153
Borowitzka MA (1998) Limits to growth. In: Wong YS, Tam NFY (eds) Wastewater treatment with algae. Springer, Berlin, pp 203–226
Brand LE, Guillard RRL, Murphy LS (1981) A method for the rapid and precise determination of acclimated phytoplankton reproduction rates. J Plankton Res 3:193–201
Bryant FD, Latimer P, Seiber BA (1969) Changes in total light scattering and absorption caused by changes in particle conformation-A test of theory. Arch Biochem Biophys 135:109–117
Butterwick C, Heaney SI, Talling JF (1982) A comparison of eight methods for estimating the biomass and growth of planktonic algae. Brit Phycol J 17:69–79
Chen M, Mertiri T, Holland T, Basu AS (2012) Optical microplates for high-throughput screening of photosynthesis in lipid-producing algae. Lab Chip 12:3870–3874
Chioccioli M, Hankamer B, Ross IL (2014) Flow cytometry pulse width data enables rapid and sensitive estimation of biomass dry weight in the microalgae Chlamydomonas reinhardtii and Chlorella vulgaris. PLoS ONE 9:e97269
Desquilbet TE, Duval JC, Robert B, Houmard J, Thomas JC (2003) In the unicellular red alga Rhodella violacea iron deficiency induces an accumulation of uncoupled LHC. Plant Cell Physiol 44:1141–1151
Domozych DS (2016) Biosynthesis of the cell walls of the algae. In: Borowitzka MA, Beardall J, Raven JA (eds) The physiology of microalgae. Springer, Cham, pp 47–63
Donner A, Glaser K, Borchhardt N, Karsten U (2017) Ecophysiological response on dehydration and temperature in terrestrial Klebsormidium (Streptophyta) isolated from biological soil crusts in Central European grasslands and forests. Microb Ecol 73:850–864
Eisentraeger A, Dott W, Klein J, Hahn S (2003) Comparative studies on algal toxicity testing using fluorometric microplate and Erlenmeyer flask growth-inhibition assays. Ecotoxicol Environ Saf 54:346–354
Elbert W, Weber B, Burrows S, Steinkamp J, Büdel B, Andreae MO, Pöschl U (2012) Contribution of cryptogamic covers to the global cycles of carbon and nitrogen. Nat Geosci 5:459–462
Esteves AF, Pires JCM, Gonçalves AL (2021) Current utilization of microalgae in the food industry beyond direct human consumption. In: Lafarga T, Acien G (eds) Cultured microalgae for the food industry. Academic Press, London, pp 199–248
French CS, Young VK (1951) The fluorescence spectra of red algae and the transfer of energy from phycoerythrin to phycocyanin and chlorophyll. J Gen Physiol 35:873–890
Fritzsche M, Mandenius CF (2010) Fluorescent cell-based sensing approaches for toxicity testing. Anal Bioanal Chem 398:181–191
Gantt E, Grabowski B, Cunningham FX (2003) Antenna systems of red algae: Phycobilisomes with Photosystem ll and chlorophyll complexes with Photosystem I. In: Green BR, Parson WW (eds) Light harvesting antennas in photosynthesis. Kluwer, Dordrecht, pp 307–322
Geis SW, Fleming KL, Korthals ET, Searle G, Reynolds L, Karner DA (2000) Modifications to the algal growth inhibition test for use as a regulatory assay. Environ Toxicol Chem 19:36–41
Giovagnetti V, Brunet C, Conversano F, Tramontano F, Obernosterer I, Ridame C, Guieu C (2013) Assessing the role of dust deposition on phytoplankton ecophysiology and succession in a low-nutrient low-chlorophyll ecosystem: A mesocosm experiment in the mediterranean sea. Biogeosciences 10:2973–2991
Gregor J, Maršálek B (2004) Freshwater phytoplankton quantification by chlorophyll a: A comparative study of in vitro, in vivo and in situ methods. Water Res 38:517–522
Gregor J, Maršálek B (2005) A simple in vivo fluorescence method for the selective detection and quantification of freshwater cyanobacteria and eukaryotic algae. Acta Hydrochim Hydrobiol 33:142–148
Griffiths MJ, Garcin C, van Hille RP, Harrison STL (2011) Interference by pigment in the estimation of microalgal biomass concentration by optical density. J Microbiol Meth 85:119–123
Guillard RRL (1973) Division rates. In: Stein JR (ed) Handbook of phycological methods: Culture methods and growth measurements, vol 1. Cambridge University Press, Cambridge, pp 289–311
Guillard RRL (1975) Culture of phytoplankton for fedding marine invertebrates. In: Smith WL, Chanley MH (eds) Culture of marine invertebrates. Springer, Boston, pp 29–60
Gustavs L, Schumann R, Eggert A, Karsten U (2009) In vivo growth fluorometry: Accuracy and limits of microalgal growth rate measurements in ecophysiological investigations. Aquat Microb Ecol 55:95–104
Haire TC, Bell C, Cutshaw K, Swiger B, Winkelmann K, Palmer AG (2018) Robust microplate-based methods for culturing and in vivo phenotypic screening of Chlamydomonas reinhardtii. Front Plant Sci 9:1–10
Hannan PJ, Patouillet C (1972) Effect of mercury on algal growth rates. Biotechnol Bioeng 14:93–101
Jezberová J, Komárková J (2007) Morphometry and growth of three Synechococcus-like picoplanktic cyanobacteria at different culture conditions. Hydrobiologia 578:17–27
Jørgensen BB, Des Marais DJ (1988) Optical properties of benthic photosynthetic communities: Fiber-optic studies of cyanobacterial mats. Limnol Oceanogr 33:99–113
Karsten U, Klimant I, Holst G (1996) A new in vivo fluorimetric technique to measure growth of adhering phototrophic microorganisms. Appl Environ Microbiol 62:237–243
Karsten U, Lütz C, Holzinger A (2010) Ecophysiological performance of the aeroterrestrial green alga Klebsormidium crenulatum (Charophyceae, Streptophyta) isolated from an alpine soil crust with an emphasis on desiccation stress. J Phycol 46:1187–1197
Khan MI, Shin JH, Kim JD (2018) The promising future of microalgae: Current status, challenges, and optimization of a sustainable and renewable industry for biofuels, feed, and other products. Microb Cell Fact 17:36
Kitzing C, Pröschold T, Karsten U (2014) UV-induced effects on growth, photosynthetic performance and sunscreen contents in different populations of the green alga Klebsormidium fluitans (Streptophyta) from Alpine soil crusts. Microb Ecol 67:327–340
Kong W, Liu N, Zhang J, Yang Q, Hua S, Song H, Xia C (2014) Optimization of ultrasound-assisted extraction parameters of chlorophyll from Chlorella vulgaris residue after lipid separation using response surface methodology. J Food Sci Technol 51:2006–2013
Kuczynska P, Jemiola-Rzeminska M, Strzalka K (2015) Photosynthetic pigments in diatoms. Mar Drugs 13:5847–5881
Lehmuskero A, Skogen Chauton M, Boström T (2018) Light and photosynthetic microalgae: A review of cellular- and molecular-scale optical processes. Progr Oceanogr 168:43–56
Liang Y, Mai K, Sun S (2005) Differences in growth, total lipid content and fatty acid composition among 60 clones of Cylindrotheca fusiformis. J Appl Phycol 17:61–65
Longhurst A, Sathyendranath S, Platt T, Caverhill C (1995) An estimate of global primary production in the ocean from satellite radiometer data. J Plankton Res 17:1245–1271
MacColl R (1998) Cyanobacterial phycobilisomes. J Struct Biol 124:311–334
MacColl R, Guard-Friar D (1987) Phycobiliproteins. CRC Press, Boca Raton
Millie D, Schofield O, Kirkpatrick G, Johnson G, Evens T (2002) Using absorbance and fluorescence spectra to discriminate microalgae. Eur J Phycol 37:313–322
Nagai T, Taya K, Yoda I (2016) Comparative toxicity of 20 herbicides to 5 periphytic algae and the relationship with mode of action. Environ Toxicol Chem 35:368–375
Pálmai T, Szabó B, Kotut K, Krienitz L, Padisák J (2020) Ecophysiology of a successful phytoplankton competitor in the African flamingo lakes: the green alga Picocystis salinarum (Picocystophyceae). J Appl Phycol 32:1813–1825
Papageorgiou GC (2004) Fluorescence of Photosynthetic pigments in vitro and in vivo. In: Papageorgiou GC, Govindjee (eds) Chlorophyll a Fluorescence. Advances in Photosynthesis and Respiration. Springer, Dordrecht, pp 43–58
Podevin M, De Francisci D, Holdt SL, Angelidaki I (2015) Effect of nitrogen source and acclimatization on specific growth rates of microalgae determined by a high-throughput in vivo microplate autofluorescence method. J Appl Phycol 27:1415–1423
Prelle LR, Albrecht M, Karsten U, Damer P, Giese T, Jähns J, Müller S, Schulz L, Viertel L, Glaser K (2021) Ecophysiological and cell biological traits of benthic diatoms from coastal wetlands of the southern Baltic Sea. Front Microbiol 12:642811
Provasoli L (1968) Media and prospects for the cultivation of marine algae. In: Watanabe A, Hattori A (eds) Cultures and Collections of Algae. Japanese Society Plant Physiology, Hakone, pp 63–75
Raven JA, Falkowski PG (1999) Oceanic sinks for atmospheric CO2. Plant Cell Environ 22:741–755
Ritchie RJ (2008) Universal chlorophyll equations for estimating chlorophylls a, b, c, and d and total chlorophylls in natural assemblages of photosynthetic organisms using acetone, methanol, or ethanol solvents. Photosynthetica 46:115–126
Ritz M, Thomas JC, Spilar A, Etienne AL (2000) Kinetics of photoacclimation in response to a shift to high light of the red alga Rhodella violacea adapted to low irradiance. Plant Physiol 123:1415–1425
Roche KR, Drummond JD, Boano F, Packman AI, Battin TJ, Hunter WR (2017) Benthic biofilm controls on fine particle dynamics in streams. Water Resour Res 53:222–236
Sakamoto T, Bryant DA (1999) Nitrate transport and not photoinhibition limits growth of the freshwater cyanobacterium Synechococcus species PCC 6301 at low temperature. Plant Physiol 119:785–794
Schubert H, Schiewer U, Tschirner E (1989) Fluorescence characteristics of cyanobacteria (blue-green algae). J Plankton Res 11:353–359
Seyfabadi J, Ramezanpour Z, Khoeyi ZA (2011) Protein, fatty acid, and pigment content of Chlorella vulgaris under different light regimes. J Appl Phycol 23:721–726
Skjelbred B, Edvardsen B, Andersen T (2012) A high-throughput method for measuring growth and loss rates in microalgal cultures. J Appl Phycol 24:1589–1599
Śliwińska-Wilczewska S, Konarzewska Z, Wiśniewska K, Konik M (2020) Photosynthetic pigments changes of three phenotypes of picocyanobacteria Synechococcus sp. under different light and temperature conditions. Cells 9:2030
Sorokin C, Krauss RW (1959) Maximum growth rates of Chlorella in steady-state and in synchronized cultures. Proc Nat Acad Sci USA 45:1740–1744
Stadnichuk IN, Krasilnikov PM, Zlenko DV (2015) Cyanobacterial phycobilisomes and phycobiliproteins. Microbiology 84:101–111
Starr RC, Zeikus JA (1993) UTEX—The culture collection of algae at the University of Texas at Austin 1993. J Phycol 29:1–106
Sukenik A (1991) Ecophysiological considerations in the optimization of eicosapentaenoic acid production by Nannochloropsis sp. (Eustigmatophyceae). Bioresour Technol 35:263–269
Suresh Kumar K, Dahms HU, Lee JS, Kim HC, Lee WC, Shin K-H (2014) Algal photosynthetic responses to toxic metals and herbicides assessed by chlorophyll a fluorescence. Ecotoxicol Environ Saf 104:51–71
Van Wagenen J, Holdt SL, De Francisci D, Valverde-Pérez B, Plósz BG, Angelidaki I (2014) Microplate-based method for high-throughput screening of microalgae growth potential. Bioresour Technol 169:566–572
Wood A, Everroad R, Wingard L (2005) Measuring growth rates in microalgal cultures. In: Andersen A (ed) Algal culturing techniques. Elsevier Academic Press, Burlington, pp 269–282
Zachleder V, Ivanov IN, Kselíková V, Bialevich Vítová M, Ota S, Takeshita T, Kawano S, Bišová K (2021) Characterization of growth and cell cycle events affected by light intensity in the green alga Parachlorella kessleri: A new model for cell cycle research. Biomolecules 11:891
Zehr JP (2011) Nitrogen fixation by marine cyanobacteria. Trends Microbiol 19:162–173
Zhao M, Heinsch FA, Nemani RR, Running SW (2005) Improvements of the MODIS terrestrial gross and net primary production global data set. Remote Sens Environ 95:164–176
Acknowledgements
We want to thank Franziska Gladis-Schmacka, Lydia Gustavs, Henriette Häuser and further working group members who contributed by working on the pre-experiments to this publication.
Funding
Open Access funding enabled and organized by Projekt DEAL. This work was funded by the Deutsche Forschungsgemeinschaft (grant: DFG (KA899/16) and by scholarships from the Ministry of Education, Science, and Culture of Mecklenburg-Vorpommern, Germany.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Laura Fuchs, Samira Khanipour-Roshan and Martin Albrecht. The draft of this manuscript was written by Martin Albrecht and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Albrecht, M., Khanipour Roshan, S., Fuchs, L. et al. Applicability and limitations of high-throughput algal growth rate measurements using in vivo fluorescence in microtiter plates. J Appl Phycol 34, 2037–2049 (2022). https://doi.org/10.1007/s10811-022-02778-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-022-02778-z