Abstract
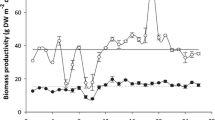
The kelp Saccharina latissima is an economically important edible seaweed with great potential for use in other applications. The goal of this study was to identify locations in a coastal bay with better conditions for commercial cultivation of this species. We compared the biomass yield and morphological features of S. latissima cultivated at two locations in a bay of Galicia (northwest Spain): in an external moderately exposed site and in an internal sheltered site. Biomass yield was significantly higher at the moderately exposed site than at the sheltered site (16 fresh kg vs. 12 fresh kg per linear meter of rope). Fronds were significantly larger at the moderately exposed site compared to that at the sheltered site. However, the “substantiality value” (around 40 mg cm−2), which is an index used to assess blade quality for human consumption, did not differ significantly between the two locations. The effect of light exposure and water velocity seemed to be the most important environmental factors that explain the differences in the results obtained at both culture sites. In summary, both the moderately exposed site and the sheltered site were suitable for cultivation of S. latissima, as indicated by the high yields and similar blade quality obtained at both locations.





Similar content being viewed by others
References
Abe E, Kakiuchi M, Matsuyama K, Kaneko T (1984) On the estimating method of the blade area in Laminaria religiosa Miyabe, Oshoro Bay, Hokkaido. Bull Hokkaido Reg Fish Res Lab 25:47–60 (in Japanese with English abstract)
Bartsch I, Wiencke C, Bischof K, Buchholz CM, Buck BH, Eggert A, Feuerpfeil P, Hanelt D, Jacobsen S, Karez R, Karsten U, Molis M, Roleda MY, Schubert H (2008) The genus Laminaria sensu lato: recent insights and developments. Eur J Phycol 43:1–86
Bolton JJ, Lüning K (1982) Optimal growth and maximal survival temperatures of Atlantic Laminaria species (Phaeophyta) in culture. Mar Biol 66:89–94
Borum J, Pedersen MF, Krause-Jensen D, Christensen PB, Nielsen K (2002) Biomass, photosynthesis and growth of Laminaria saccharina in a high-arctic fjord, NE Greenland. Mar Biol 141:11–19
Brinkhuis BH, Breda VA, Tobin S, Macler BA (1983) New York marine biomass program-culture of Laminaria saccharina. J World Maricul Soc 14:360–379
Brinkhuis BH, Mariani EC, Breda VA, Brady-Campbell MM (1984) Cultivation of Laminaria saccharina in the New York Marine Biomass Program. Hydrobiologia 116/117:266–277
Buck BH, Buchholz CM (2004) The offshore-ring: a new system design for the open ocean aquaculture of macroalgae. J Appl Phycol 16:355–368
Buck BH, Buchholz CM (2005) Response of offshore cultivated Laminaria saccharina to hydrodynamic forcing in the North Sea. Aquaculture 250:674–691
Chugaynova VA, Gorennikov SP (1996) Mariculture of Laminaria saccharina in the White Sea. Hydrobiol J 32:63–67
Cremades J, Freire Ó, Baamonde S, Salinas JM, Fuertes C (2007) Nuevo método para el cultivo industrial de Laminaria saccharina (Laminariales, Phaeophyta) en las costas gallegas. In: Cerviño Eiroa A, Guerra Díaz A, Pérez Acosta C (eds) XI Congreso Nacional de Acuicultura. Consellería de Pesca e Asuntos Marítimos, Xunta de Galicia, Vigo, Spain, pp 559–562 (in Spanish with English abstract)
Dring MJ, Luning K (1994) Influence of spring-neap tidal cycles on the light available for photosynthesis by benthic marine plants. Mar Ecol Prog Ser 104:131–137
Druehl LD, Baird R, Lindwall A, Lloyd KE, Pakula S (1988) Longline cultivation of some Laminariaceae in British Columbia, Canada. Aquacult Fish Manag 19:253–263
Fortes MD, Lüning K (1980) Growth rates of North Sea macroalgae in relation to temperature, irradiance and photoperiod. Helgoländer Meeresunters 34:15–29
Fujita RM, Goldman JC (1985) Nutrient flux and growth of the red alga Gracilaria tikvahiae McLachlan (Rhodophyta). Bot Mar 28:265–268
Gao K (1991) Effects of seawater current speed on the photosynthetic oxygen evolution of Sargassum thunbergii (Phaeophyta). Jpn J Phycol 39:291–293
Gao K, Aruga Y, Asada K, Ishihara T, Akano T, Kiyohara M (1992) Enhancement of photosynthetic CO2 fijation of the red alga Porphyra yezoensis Ueda in flowing seawater. Jpn J Phycol 40:377–400
Gerard V (1982) In situ water motion and nutrient uptake by the giant kelp Macrocystis pyrifera. Mar Biol 69:51–54
Gerard VA (1987) Hydrodynamic streamlining of Laminaria saccharina Lamour. in response to mechanical stress. J Exp Mar Biol Ecol 107:237–244
Gerard V, Mann KH (1979) Growth and production of Laminaria longicruris (Phaeophyta) populations exposed to different intensities of water movement. J Phycol 15:33–41
Gerard VA, DuBois K, Greene R (1987) Growth responses of two Laminaria saccharina populations to environmental variation. Hydrobiologia 151/152:229–232
Gevaert F, Davoult D, Creach A, Kling R, Janquin MA, Seuront L, Lemoine Y (2001) Carbon and nitrogen content of Laminaria saccharina in the eastern English Channel: biometrics and seasonal variations. J Mar Biol Assoc UK 81:727–734
Glenn EP, Doty MS (1992) Water motion affects the growth rates of Kappaphycus alvarezii and related red seaweeds. Aquaculture 108:233–246
Gonen Y, Kimmel E, Friedlander M (1993) Effect of relative water motion on photosynthetic rate of red alga Gracilaria conferta. Hydrobiologia 261:493–498
Han T, Kain (Jones) JM (1996) Effect of photon irradiance and photoperiod on young sporophytes of four species of the Laminariales. Eur J Phycol 31:233–240
Hurd CL (2000) Water motion, marine macroalgal physiology and production. J Phycol 36:453–472
Jackson GA (1997) Currents in the high drag environment of a coastal kelp stand off California. Cont Shef Res 17:1913–1928
Johnson AS (2001) Drag, drafting, and mechanical interactions in canopies of the red alga Chondrus crispus. Biol Bull 201:126–135
Kain (Jones) JM, Holt TJ, Dawes CP (1990) European Laminariales and their cultivation. In: Yarish C, Penniman CA, Van Petten P (eds) Economically important plants of the Atlantic: their biology and cultivation. Connecticut Sea Grant College Program, University of Connecticut, Groton, pp 95–111
Karsten U (2007) Salinity tolerance of Arctic kelps from Spitsbergen. Phycol Res 55:257–262
Katz S, Kizner Z, Dubinsky Z, Friedlander M (2000) Responses of Porphyra linearis (Rhodophyta) to environmental factors under controlled culture conditions. J Appl Phycol 12:535–542
Kawamata S (2001) Adaptive mechanical tolerance and dislodgement velocity of the kelp Laminaria japonica in wave-induced water motion. Mar Ecol Prog Ser 211:89–104
Kawashima S (1984) Kombu cultivations in Japan for human foodstuff. Jpn J Phycol 32:379–394
Kawashima S (1993) Cultivation of the brown alga, Laminaria “kombu”. In: Ohno M, Critchley AT (eds) Seaweed cultivation and marine ranching. Japan International Cooperation Agency (JICA), Jokosuka, pp 25–40
Koch EW (1993) The effect of water flow on photosynthetic processes of the alga Ulva lactuca L. Hydrobiologia 260(261):457–462
Koehl MAR, Alberte RS (1988) Flow, flapping, and photosynthesis of Nereocystis luetkeana: a functional comparison of undulate and flat blade morphologies. Mar Biol 99:435–444
Koehl MAR, Silk WK, Liang H, Mahadevan L (2008) How kelp produce blade shapes suited to different flow regimes: a new wrinkle. Integr Comp Biol 48:834–851
Kubler JE, Raven JA (1996a) Inorganic carbon acquisition by red seaweeds grown under dynamic light regimes. Hydrobiologia 327:401–406
Kubler JE, Raven JA (1996b) Nonequilibrium rates of photosynthesis and respiration under dynamic light supply. J Phycol 32:963–969
Lapointe BE, Ryther JH (1979) The effects of nitrogen and seawater flow rate on the growth and biochemical composition of Gracilaria foliifera var. angustissima in mass outdoor cultures. Bot Mar 22:529–538
Leigh EG, Paine RT, Quinn JF, Suchanek TH (1987) Wave energy and intertidal productivity. Proc Natl Acad Sci USA 84:1314–1318
Lüning K (1981) Light. In: Lobban CS, Wynne MJ (eds) The biology of seaweeds. Blackwell Scientific Publications, Oxford, pp 326–355
Lüning K, Freshwater W (1988) Temperature tolerance of Northeast Pacific marine algae. J Phycol 24:310–315
Mairh OP, Ohno M, Matsuoka M (1991) Culture of brown alga Laminaria japonica (Phaeophyta, Laminariales) in warm waters of Shikoku, Japan. Indian J Mar Sci 20:55–60
Mariño J, Campos MJ, Nunes MT, Iglesias MT (1985) Variación estacional de los factores ambientales y del fitoplancton en la zona de Lorbé (Ría de Ares y Betanzos) en 1978. Bol Inst Esp Oceanogr 2(1):88–99 (in Spanish with English abstract)
Molloy FJ, Bolton JJ (1996) The effects of wave exposure and depth on the morphology of inshore populations of the Namibian kelp, Laminaria schinzii Foslie. Bot Mar 39:525–531
Nanba N, Fujiwara T, Kuwano K, Ishikawa Y, Ogawa H, Kado R (2011) Effect of water flow velocity on growth and morphology of cultured Undaria pinnatifida sporophytes (Laminariales, Phaeophyceae) in Okirai Bay on the Sanriku coast, Northeast Japan. J Appl Phycol 23:1023–1030
Ozaki A, Mizuta H, Yamamoto H (2001) Physiological differences between the nutrient uptakes of Kjellmaniella crassifolia and Laminaria japonica (Phaeophyceae). Fish Sci 67:415–419
Palmisano JF, Sheng YC (1977) Blade width of Laminaria longipes (Phaeophyceae, Laminariales) as an indicator of wave exposure. Syesis 10:53–52
Parke M (1948) Studies on British Laminariaceae: I. Growth in Laminaria saccharina (L.) Lamour. J Mar Biol Assoc UK 27:651–709
Parker HS (1981) Influence of relative water motion on the growth, ammonium uptake and carbon and nitrogen composition of Ulva lactuca (Chlorophyta). Mar Biol 63:309–318
Parker HS (1982) Effects of simulated current on the growth rate and nitrogen metabolism of Gracilaria tikvahiae (Rhodophyta). Mar Biol 69:137–145
Pérez-Villar V, Gómez-Gesteira M, Pérez-Muñuzuri V (2000) Modelización informática. In: Ordenación integral del espacio marítimo-terrestre de Galicia. Fundación Empresa-Universidad Galega, Santiago de Compostela, pp 41–46 (in Spanish)
Peteiro C, Freire Ó (2009) Effect of outplanting time on the commercial cultivation of the kelp Laminaria saccharina at the southern limit in the Atlantic Coast (N.W. Spain). Chin J Oceanol Limnol 27:54–60
Peteiro C, Freire Ó (2011a) Effect of water motion on the cultivation of the commercial seaweed Undaria pinnatifida in a coastal bay of Galicia, Northwest Spain. Aquaculture 314:269–276
Peteiro C, Freire Ó (2011b) Offshore cultivation methods affects blade features of the edible seaweed Saccharina latissima in a bay of Galicia, Northwest Spain. Russ J Mar Biol 37:319–323
Peteiro C, Sánchez N (2012) Comparing salinity tolerance in early stages of the sporophytes of a non-indigenous kelp (Undaria pinnatifida) and a native kelp (Saccharina latissima). Russ J Mar Biol 38(2):197–200
Peteiro C, Salinas JM, Freire Ó, Fuertes C (2006) Cultivation of the autoctonous seaweed Laminaria saccharina off the galician coast (NW Spain): production and features of the sporophytes for an annual and biennial harvest. Thalassas 22:45–52
Ryder E, Nelson SG, McKeon GEP, Fitzsimmons K, Napolean S (2004) Effect of water motion on the cultivation of the economic seaweed Gracilaria parvispora (Rhodophyta) on Molokai, Hawaii. Aquaculture 238:207–219
Sanbonsuga Y (1984) Studies of the growth of forced Laminaria. Bull Hokkaido Reg Fish Res Lab 49:1–78
Sjøtun K, Fredriksen S (1995) Growth allocation in Laminaria hyperborea in relation to age and wave exposure. Mar Ecol Prog Ser 126:213–222
Stevens CL, Hurd CL (1997) Boundary-layers around bladed aquatic macrophytes. Hydrobiologia 346:119–128
Stewart HL (2006) Hydrodynamic consequences of flexural stiffness and buoyancy for seaweeds: a study using physical models. J Exp Biol 209:2170–2181
Stewart HL, Carpenter RC (2003) The effects of morphology and water flow on photosynthesis of marine macroalgae. Ecol 84:2999–3012
Stewart HL, Payri CE, Koehl MAR (2007) The role of buoyancy in mitigating reduced light in macroalgal aggregations. J Exp Mar Biol Ecol 343:11–20
Sundene O (1962) Growth in the sea of Laminaria digitata sporophytes from culture. Nytt Mag Bot 11:5–26
Wheeler WN (1980) Effect of boundary layer transport on the fixation of carbon by the giant kelp Macrocystis pyrifera. Mar Biol 56:103–110
Wheeler WN (1982) Nitrogen nutrition of Macrocystis pyrifera. In: Srivastava LM (ed) Synthetic and degradative processes in marine macrophytes. Walter de Gruyter, Berlin, pp 121–137
Wing SR, Patterson MR (1993) Effects of wave-induced light flecks in the intertidal zone on photosynthesis in the macroalgae Postelsia palmaeformis and Hedophyllum sessile (Phaeophyceae). Mar Biol 116:519–525
Wing SR, Leichter JJ, Denny MW (1993) A dynamic model for wave-induced light fluctuations in a kelp forest. Limnol Oceanogr 38:396–407
Acknowledgments
This research has been developed in the framework of the projects AGL2002-01285 funded by the Ministerio de Ciencia y Tecnologia of Spanish government. Thanks to the University of A Coruña and Provincial Council of A Coruña for awarding research fellowships to support this study. Gratitude is extended to PORTO-MUIÑOS edible seaweed selling company (A Coruña, Spain) and to all the members of the project. Also, special thanks to Dr N. Sánchez for her time and advice that greatly improved the manuscript and to Dr A. Secilla for his assistance in elaboration of the figures. Finally, we acknowledge the reviewers' suggestions which greatly improved this paper. The English language has been edited by International Science Editing, Ireland.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Peteiro, C., Freire, Ó. Biomass yield and morphological features of the seaweed Saccharina latissima cultivated at two different sites in a coastal bay in the Atlantic coast of Spain. J Appl Phycol 25, 205–213 (2013). https://doi.org/10.1007/s10811-012-9854-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-012-9854-9