Abstract
Purpose
To introduce a new method for estimation of the target intraocular pressure (TIOP) in naïve eyes with early primary open-angle glaucoma (POAG) using corneal hysteresis (CH) and corneal resistance factor (CRF).
Methods
A prospective quasi-experimental study was conducted on naïve 90 eyes of 45 patients who were newly diagnosed with early primary open-angle glaucoma (POAG). They were compared to 72 eyes of 36 normal subjects. The TIOP was determined for each eye. The IOP Goldmann (IOPg), IOP corneal-compensated (IOPcc), CH and CRF were estimated by ocular response analyzer (ORA, Reichert) device. Measurements were taken for each patient prior to treatment and after 1, 3, 6, 9 and 12 months of receiving medications; either monotherapy or combination therapy.
Results
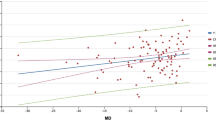
For all patients, there was a significant negative correlation (p < 0.05) between IOP, either IOPg or IOPcc, and CH, while a significant positive relationship (p < 0.05) existed between IOP and CRF. For patients with early POAG, the CH was significantly increased (p ≤ 0.001), while CRF was significantly decreased (p ≤ 0.001) when TIOP was achieved. At IOP levels higher than TIOP, CH value was lower than CRF with a significant negative correlation between them in contrast to controls. This correlation was reversed on reaching TIOP and CH values became higher than CRF similar to controls.
Conclusion
CH, CRF and IOP measured by ORA can be used for TIOP estimation. This provides us with a guide for assessing the effectiveness of medications introduced to patients with POAG.




Similar content being viewed by others
References
Sihota R, Angmo D, Ramaswamy D, Dada T (2018) Simplifying “target” intraocular pressure for different stages of primary open-angle glaucoma and primary angle-closure glaucoma. Indian J Ophthalmol 66:495–505. https://doi.org/10.4103/ijo.IJO_1130_17
Prum BE, Rosenberg LF, Gedde SJ et al (2016) Primary open-angle glaucoma preferred practice pattern® guidelines. Ophthalmology 123:P41–P111
Quaranta L, Riva I, Gerardi C et al (2016) Quality of life in glaucoma: a review of the literature. Adv Ther 33:959–981. https://doi.org/10.1007/s12325-016-0333-6
Parikh RS, Parikh SR, Navin S et al (2008) Practical approach to medical management of glaucoma. Indian J Ophthalmol 56:223–230. https://doi.org/10.4103/0301-4738.40362
Heijl A, Leske MC, Bengtsson B et al (2002) Reduction of intraocular pressure and glaucoma progression: results from the early manifest glaucoma trial. Arch Ophthalmol (Chicago, Ill 1960) 120:1268–1279. https://doi.org/10.1001/archopht.120.10.1268
Yaoeda K, Fukushima A, Shirakashi M, Fukuchi T (2016) Comparison of intraocular pressure adjusted by central corneal thickness or corneal biomechanical properties as measured in glaucomatous eyes using noncontact tonometers and the Goldmann applanation tonometer. Clin Ophthalmol 10:829–834. https://doi.org/10.2147/OPTH.S106836
Medeiros FA, Meira-Freitas D, Lisboa R et al (2013) Corneal hysteresis as a risk factor for glaucoma progression: a prospective longitudinal study. Ophthalmology 120:1533–1540. https://doi.org/10.1016/j.ophtha.2013.01.032
Brown KE, Congdon NG (2006) Corneal structure and biomechanics: impact on the diagnosis and management of glaucoma. Curr Opin Ophthalmol 17:338–343. https://doi.org/10.1097/01.icu.0000233951.01971.5b
Medeiros FA, Weinreb RN (2006) Evaluation of the influence of corneal biomechanical properties on intraocular pressure measurements using the ocular response analyzer. J Glaucoma 15:364–370. https://doi.org/10.1097/01.ijg.0000212268.42606.97
Johnson CS, Mian SI, Moroi S et al (2007) Role of corneal elasticity in damping of intraocular pressure. Invest Ophthalmol Vis Sci 48:2540–2544. https://doi.org/10.1167/iovs.06-0719
Congdon NG, Broman AT, Bandeen-Roche K et al (2006) Central corneal thickness and corneal hysteresis associated with glaucoma damage. Am J Ophthalmol 141:868–875. https://doi.org/10.1016/j.ajo.2005.12.007
De Moraes CVG, Hill V, Tello C et al (2012) Lower corneal hysteresis is associated with more rapid glaucomatous visual field progression. Ophthalmology 141:868–875. https://doi.org/10.1097/IJG.0b013e3182071b92
Park JH, Jun RM, Choi K-R (2015) Significance of corneal biomechanical properties in patients with progressive normal-tension glaucoma. Br J Ophthalmol 99:746–751. https://doi.org/10.1136/bjophthalmol-2014-305962
Pillunat KR, Hermann C, Spoerl E, Pillunat LE (2016) Analyzing biomechanical parameters of the cornea with glaucoma severity in open-angle glaucoma. Graefe’s Arch Clin Exp Ophthalmol = Albr von Graefes Arch fur Klin und Exp Ophthalmol 254:1345–1351. https://doi.org/10.1007/s00417-016-3365-3
Sullivan-Mee M, Billingsley SC, Patel AD et al (2008) Ocular response analyzer in subjects with and without glaucoma. Optom Vis Sci 85:463–470. https://doi.org/10.1097/OPX.0b013e3181784673
Thomas R, Thomas S, Chandrashekar G (1998) Gonioscopy. Indian J Ophthalmol 46:255–261
Mundial AM (2013) World medical association declaration of Helsinki ethical principles for medical research involving human subjects. JAMA 310:2191–2194. https://doi.org/10.1001/jama.2013.281053
Hodapp E, Parrish R K, Anderson DR (1993) Clinical decisions in Glaucoma. Mosby Company, Maryland Heights, MO, USA
Bengtsson B, Olsson J, Heijl A, Rootzén H (1997) A new generation of algorithms for computerized threshold perimetry, SITA. Acta Ophthalmol Scand 75:368–375. https://doi.org/10.1111/j.1600-0420.1997.tb00392.x
Ayala M, Chen E (2012) Measuring corneal hysteresis: threshold estimation of the waveform score from the ocular response analyzer. Graefe’s Arch Clin Exp Ophthalmol = Albr von Graefes Arch fur Klin und Exp Ophthalmol 250:1803–1806. https://doi.org/10.1007/s00417-012-2053-1
Kaushik S, Pandav SS (2012) Ocular response analyzer. J Curr glaucoma Pract 6:17–19. https://doi.org/10.5005/jp-journals-10008-1103
(2017) Terminology and guidelines for glaucoma. Br J Ophthalmol 4th edition: https://doi.org/10.1136/bjophthalmol-2016-EGSguideline.01
De Moraes CVG, Hill V, Tello C et al (2012) Lower corneal hysteresis is associated with more rapid glaucomatous visual field progression. J Glaucoma 21:209–213. https://doi.org/10.1097/IJG.0b013e3182071b92
Chandrasekaran S, Uppuluri S, Sadek H et al (2020) Fluctuations of the ocular response analyzer in measuring corneal hysteresis of subjects with and without glaucoma and other systemic conditions. Invest Ophthalmol Vis Sci 61:4710
Ehrlich JR, Haseltine S, Shimmyo M, Radcliffe NM (2010) Evaluation of agreement between intraocular pressure measurements using Goldmann applanation tonometry and Goldmann correlated intraocular pressure by Reichert’s ocular response analyser. Eye (Lond) 24:1555–1560. https://doi.org/10.1038/eye.2010.83
Deol M, Taylor DA, Radcliffe NM (2015) Corneal hysteresis and its relevance to glaucoma. Curr Opin Ophthalmol 26:96–102. https://doi.org/10.1097/ICU.0000000000000130
Mangouritsas G, Morphis G, Mourtzoukos S, Feretis E (2009) Association between corneal hysteresis and central corneal thickness in glaucomatous and non-glaucomatous eyes. Acta Ophthalmol 87:901–905. https://doi.org/10.1111/j.1755-3768.2008.01370.x
Liang L, Zhang R, He L-Y (2019) Corneal hysteresis and glaucoma. Int Ophthalmol 39:1909–1916. https://doi.org/10.1007/s10792-018-1011-2
Agarwal DR, Ehrlich JR, Shimmyo M, Radcliffe NM (2012) The relationship between corneal hysteresis and the magnitude of intraocular pressure reduction with topical prostaglandin therapy. Br J Ophthalmol 96:254–257. https://doi.org/10.1136/bjo.2010.196899
Tsikripis P, Papaconstantinou D, Koutsandrea C et al (2013) The effect of prostaglandin analogs on the biomechanical properties and central thickness of the cornea of patients with open-angle glaucoma: a 3-year study on 108 eyes. Drug Des Devel Ther 7:1149–1156. https://doi.org/10.2147/DDDT.S50622
Bolívar G, Sánchez-Barahona C, Teus M et al (2015) Effect of topical prostaglandin analogues on corneal hysteresis. Acta Ophthalmol 93:e495–e498. https://doi.org/10.1111/aos.12689
Meda R, Wang Q, Paoloni D et al (2017) The impact of chronic use of prostaglandin analogues on the biomechanical properties of the cornea in patients with primary open-angle glaucoma. Br J Ophthalmol 101:120–125. https://doi.org/10.1136/bjophthalmol-2016-308432
Miki A, Medeiros FA, Weinreb RN et al (2014) Rates of retinal nerve fiber layer thinning in glaucoma suspect eyes. Ophthalmology 121:1350–1358. https://doi.org/10.1016/j.ophtha.2014.01.017
Funding
This research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethical approval
This study was approved by our National and Institutional Research Ethics Committee (REC) in accordance with the tenets of the Declaration of Helsinki.
Consent to participate
Informed consent was obtained from all individual participants included in the study. Patients who were unwilling to provide an informed consent were excluded from the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sallam, M.A., Elghareib, M.E. Use of corneal hysteresis and corneal resistance factor in target intraocular pressure estimation in patients with early primary open-angle glaucoma. Int Ophthalmol 42, 891–902 (2022). https://doi.org/10.1007/s10792-021-02070-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-021-02070-y