Abstract
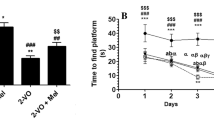
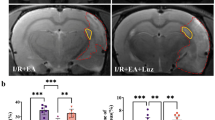
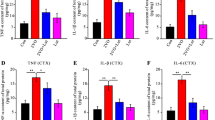
Chronic cerebral hypoxia (CCH) is caused by a reduction in cerebral blood flow, and cognitive impairment has been the predominant feature that occurs after CCH. Recent reports have revealed that melatonin is proficient in neurodegenerative diseases. However, the molecular mechanism by which melatonin affects CCH remains uncertain. In this study, we aimed to explore the role and underlying mechanism of melatonin in inflammation and blood‒brain barrier conditions in rats with CCH. Male Wistar rats were subjected to permanent bilateral common carotid artery occlusion (BCCAO) to establish the VAD model. Rats were randomly divided into four groups: Sham, BCCAO, BCCAO treated with melatonin (10 mg/kg), and BCCAO treated with resveratrol (20 mg/kg). All drugs were administered once daily for 4 weeks. Our results showed that melatonin attenuated cognitive impairment, as demonstrated by the Morris water maze tests. Furthermore, melatonin reduced the activation of inflammation by attenuating the phosphorylated nuclear factor of kappa light polypeptide gene enhancer in B cells inhibitor alpha (pIκBα), causing the suppression of proteins related to inflammation and inflammasome formation. Moreover, immunohistochemistry revealed that melatonin reduced glial cell activation and proliferation, which were accompanied by Western blotting results. Additionally, melatonin also promoted the expression of sirtuin-1 (SIRT1), peroxisome proliferator-activated receptor-gamma coactivator 1-alpha (PGC-1α), and peroxisome proliferator-activated receptor-gamma (PPARγ), causing attenuated blood‒brain barrier (BBB) disruption by increasing tight junction proteins. Taken together, our results prove that melatonin treatment modulated inflammation and BBB disruption and improved cognitive function in VaD rats, partly by activating the SIRT1/PGC-1α/PPARγ signaling pathway.







Similar content being viewed by others
Availability of data and materials
All data are available on request from authors.
References
Amaral FGd, Cipolla-Neto J (2018) A brief review about melatonin, a pineal hormone. Arch Endocrinol Metab 62:472–479. https://doi.org/10.20945/2359-3997000000066
Braidy N, Guillemin GJ, Mansour H, Chan-Ling T, Poljak A, Grant R (2011) Age related changes in NAD+ metabolism oxidative stress and Sirt1 activity in wistar rats. PLoS One 6:e19194. https://doi.org/10.1371/journal.pone.0019194
Cantó C, Auwerx J (2009) PGC-1alpha, SIRT1 and AMPK, an energy sensing network that controls energy expenditure. Curr Opin Lipidol 20:98. https://doi.org/10.1097/MOL.0b013e328328d0a4
Cekanaviciute E, Buckwalter MS (2016) Astrocytes: integrative regulators of neuroinflammation in stroke and other neurological diseases. Neurotherapeutics 13:685–701. https://doi.org/10.1007/s13311-016-0477-8
Corona JC, Duchen MR (2015) PPARγ and PGC-1α as therapeutic targets in Parkinson’s. Neurochem Res 40:308–316. https://doi.org/10.1007/s11064-014-1377-0
Cummins EP, Berra E, Comerford KM, Ginouves A, Fitzgerald KT, Seeballuck F, Godson C, Nielsen JE, Moynagh P, Pouyssegur J (2006) Prolyl hydroxylase-1 negatively regulates IκB kinase-β, giving insight into hypoxia-induced NFκB activity. Proc Natl Acad Sci 103:18154–18159. https://doi.org/10.1073/pnas.0602235103
Daulatzai MA (2017) Cerebral hypoperfusion and glucose hypometabolism: Key pathophysiological modulators promote neurodegeneration, cognitive impairment, and Alzheimer’s disease. J Neurosci Res 95:943–972. https://doi.org/10.1002/jnr.23777
Dehne N, Brüne B (2009) HIF-1 in the inflammatory microenvironment. Exp Cell Res 315:1791–1797. https://doi.org/10.1016/j.yexcr.2009.03.019
Ding Y, Kang J, Liu S, Xu Y, Shao B (2020) The protective effects of peroxisome proliferator-activated receptor gamma in cerebral ischemia-reperfusion injury. Front Neurol 11:1469. https://doi.org/10.3389/fneur.2020.588516
Du SQ, Wang XR, Xiao LY, Tu JF, Zhu W, He T, Liu CZ (2017) Molecular mechanisms of vascular dementia: what can be learned from animal models of chronic cerebral hypoperfusion? Mol. Neurobiol 54:3670–3682. https://doi.org/10.1007/s12035-016-9915-1
Fang Y, Gao S, Wang X, Cao Y, Lu J, Chen S, Zhang J (2020) Programmed cell deaths and potential crosstalk with blood–brain barrier dysfunction after hemorrhagic stroke. Front Cell Neurosci 14:68. https://doi.org/10.3389/fncel.2020.00068
Govindarajulu M, Pinky PD, Bloemer J, Ghanei N, Suppiramaniam V, Amin R (2018) Signaling mechanisms of selective PPARγ modulators in Alzheimer’s disease. PPAR Res. https://doi.org/10.1155/2018/2010675
Huang JJ, Xia J, Huang LL, Li YC (2019) HIF-1α promotes NLRP3 inflammasome activation in bleomycin-induced acute lung injury. Mol Med Rep 20:3424–3432. https://doi.org/10.3892/mmr.2019.10575
Hubbard BP, Gomes AP, Dai H, Li J, Case AW, Considine T, Riera TV, Lee JE, Lamming DW, Pentelute BL (2013) Evidence for a common mechanism of SIRT1 regulation by allosteric activators. Science 339:1216–1219. https://doi.org/10.1126/science.1231097
Hugo J, Ganguli M (2014) Dementia and cognitive impairment: epidemiology, diagnosis, and treatment. Clin Geriatr Med 30:421–442. https://doi.org/10.1016/j.cger.2014.04.001
Imai S-i (2011) Dissecting systemic control of metabolism and aging in the NAD World: the importance of SIRT1 and NAMPT-mediated NAD biosynthesis. FEBS Lett 585:1657–1662. https://doi.org/10.1016/j.febslet.2011.04.060
Kleszcz R, Paluszczak J, Baer-Dubowska W (2015) Targeting aberrant cancer metabolism–The role of sirtuins. Pharmacol Rep 67:1068–1080. https://doi.org/10.1016/j.pharep.2015.03.021
Lee CH, Park JH, Ahn JH, Won MH (2016) Effects of melatonin on cognitive impairment and hippocampal neuronal damage in a rat model of chronic cerebral hypoperfusion. Exp Ther Med 11:2240–2246. https://doi.org/10.3892/etm.2016.3216
Li X (2013) SIRT1 and energy metabolism. Acta Biochim Biophys Sin 45:51–60. https://doi.org/10.1093/abbs/gms108
Li Y, Wang P, Yang X, Wang W, Zhang J, He Y, Zhang W, Jing T, Wang B, Lin R (2016) SIRT1 inhibits inflammatory response partly through regulation of NLRP3 inflammasome in vascular endothelial cells. Mol Immunol 77:148–156. https://doi.org/10.1016/j.molimm.2016.07.018
Li J, Zheng M, Shimoni O, Banks WA, Bush AI, Gamble JR, Shi B (2021) Development of novel therapeutics targeting the blood-brain barrier: from barrier to carrier. Adv Sci. https://doi.org/10.1002/advs.202101090
Liang H, Ward WF (2006) PGC-1α: a key regulator of energy metabolism. Adv Physiol Educ. https://doi.org/10.1152/advan.00052.2006
Lim J-H, Lee Y-M, Chun Y-S, Chen J, Kim J-E, Park J-W (2010) Sirtuin 1 modulates cellular responses to hypoxia by deacetylating hypoxia-inducible factor 1α. Mol Cell 38:864–878. https://doi.org/10.1016/j.molcel.2010.05.023
Ma J, Yang H, Liu L, Wan Y, Wang F (2021) Melatonin alleviated oxidative stress induced by energy restriction on sheep Leydig cells through Sirt1/Sod2 pathway. Theriogenology 173:83–92. https://doi.org/10.1016/j.theriogenology.2021.07.011
Mojsilovic-Petrovic J, Callaghan D, Cui H, Dean C, Stanimirovic DB, Zhang W (2007) Hypoxia-inducible factor-1 (HIF-1) is involved in the regulation of hypoxia-stimulated expression of monocyte chemoattractant protein-1 (MCP-1/CCL2) and MCP-5 (Ccl12) in astrocytes. J Neuroinflammation 4:1–15. https://doi.org/10.1186/1742-2094-4-12
Permpoonputtana K, Tangweerasing P, Mukda S, Boontem P, Nopparat C, Govitrapong P (2018) Long-term administration of melatonin attenuates neuroinflammation in the aged mouse brain. EXCLI J 17:634. https://doi.org/10.17179/excli2017-654
Price NL, Gomes AP, Ling AJ, Duarte FV, Martin-Montalvo A, North BJ, Agarwal B, Ye L, Ramadori G, Teodoro JS (2012) SIRT1 is required for AMPK activation and the beneficial effects of resveratrol on mitochondrial function. Cell Metab 15:675–690. https://doi.org/10.1016/j.cmet.2012.04.003
Ren Z, He H, Zuo Z, Xu Z, Wei Z, Deng J (2019) The role of different SIRT1-mediated signaling pathways in toxic injury. Cell Mol Biol Lett 24:1–10. https://doi.org/10.1186/s11658-019-0158-9
Sanderson TH, Wider JM (2013) 2-vessel occlusion/hypotension: a rat model of global brain ischemia. J vis Exp. https://doi.org/10.3791/50173
Scirpo R, Fiorotto R, Villani A, Amenduni M, Spirli C, Strazzabosco M (2015) Stimulation of nuclear receptor PPAR-γ limits NF-kB-dependent inflammation in mouse cystic fibrosis biliary epithelium. Hepatology 62:1551. https://doi.org/10.1002/hep.28000
Shen D, Tian X, Sang W, Song R (2016) Effect of melatonin and resveratrol against memory impairment and hippocampal damage in a rat model of vascular dementia. NeuroImmunoModulation 23:318–331. https://doi.org/10.1159/000454681
Thangwong P, Jearjaroen P, Govitrapong P, Tocharus C, Tocharus J (2022) Melatonin improves cognitive impairment by suppressing endoplasmic reticulum stress and promoting synaptic plasticity during chronic cerebral hypoperfusion in rats. Biochem Pharmacol 198:114980. https://doi.org/10.1016/j.bcp.2022.114980
Tsai TH, Lin CJ, Chua S, Chung SY, Yang CH, Tong MS, Hang CL (2017) Melatonin attenuated the brain damage and cognitive impairment partially through MT2 melatonin receptor in mice with chronic cerebral hypoperfusion. Oncotarget 8:74320. https://doi.org/10.18632/oncotarget.20382
Venkat P, Chopp M, Chen J (2015) Models and mechanisms of vascular dementia. Exp Neurol 272:97–108. https://doi.org/10.1016/j.expneurol.2015.05.006
Vorhees CV, Williams MT (2006) Morris water maze: procedures for assessing spatial and related forms of learning and memory. Nat Protoc 1:848–858. https://doi.org/10.1038/nprot.2006.116
Watts ER, Walmsley SR (2019) Inflammation and hypoxia: HIF and PHD isoform selectivity. Trends Mol Med 25:33–46. https://doi.org/10.1016/j.molmed.2018.10.006
Weiss N, Miller F, Cazaubon S, Couraud P-O (2009) The blood-brain barrier in brain homeostasis and neurological diseases. Biochim Et Biophys Acta Biomembr 1788:842–857. https://doi.org/10.1016/j.bbamem.2008.10.022
Wicha P, Tocharus J, Janyou A, Jittiwat J, Chaichompoo W, Suksamrarn A, Tocharus C (2020) Hexahydrocurcumin alleviated blood–brain barrier dysfunction in cerebral ischemia/reperfusion rats. Pharmacol Rep. https://doi.org/10.1007/s43440-019-00050-9
Xue Y, Tuipulotu DE, Tan WH, Kay C, Man SM (2019) Emerging activators and regulators of inflammasomes and pyroptosis. Trends Immunol 40:1035–1052. https://doi.org/10.1016/j.it.2019.09.005
Yang CC, Wu CH, Lin TC, Cheng YN, Chang CS, Lee KT, Tsai PJ, Tsai YS (2021) Inhibitory effect of PPARγ on NLRP3 inflammasome activation. Theranostics 11:2424. https://doi.org/10.7150/thno.46873
Yawoot N, Sengking J, Wicha P, Govitrapong P, Tocharus C, Tocharus J (2022) Melatonin attenuates reactive astrogliosis and glial scar formation following cerebral ischemia and reperfusion injury mediated by GSK-3β and RIP1K. J Cell Physiol 237:1818–1832. https://doi.org/10.1002/jcp.30649
Yeleswaram K, McLaughlin LG, Knipe JO, Schabdach D (1997) Pharmacokinetics and oral bioavailability of exogenous melatonin in preclinical animal models and clinical implications. J Pineal Res 22:45–51. https://doi.org/10.1111/j.1600-079x.1997.tb00302.x
Zhao L, Liu H, Yue L, Zhang J, Li X, Wang B, Lin Y, Qu Y (2017) Melatonin attenuates early brain injury via the melatonin receptor/Sirt1/NF-κB signaling pathway following subarachnoid hemorrhage in mice. Mol Neurobiol 54:1612–1621. https://doi.org/10.1007/s12035-016-9776-7
Zhou Y, Wang S, Li Y, Yu S, Zhao Y (2018) SIRT1/PGC-1α signaling promotes mitochondrial functional recovery and reduces apoptosis after intracerebral hemorrhage in rats. Front Mol Neurosci 10:443. https://doi.org/10.3389/fnmol.2017.00443
Funding
This work was supported by grants from Functional Food Research Center for Wellbeing, Chiang Mai University, Thailand Science Research and Innovation (TSRI) (grant number 64/004) and the Development and Promotion of Science and Technology Talents Project Royal Government of Thailand scholarship (grant number 572109).
Author information
Authors and Affiliations
Contributions
The authors declare that all data were generated in-house and that no paper mill was used. All authors were involved in the study design. PT performed writing–original draft, methodology, investigation, and formal analysis. PJ performed the investigation. PG performed resources. CT performed methodology, writing–review, and editing. JT performed writing–review and editing, conceptualization, supervision, project administration, and funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Compliance with ethical standards
All experimental procedures were approved by the Institutional Animal Care and Use Committee at the Faculty of Medicine, Chiang Mai University, (Permit number: 32/2563) and performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Thangwong, P., Jearjaroen, P., Tocharus, C. et al. Melatonin suppresses inflammation and blood‒brain barrier disruption in rats with vascular dementia possibly by activating the SIRT1/PGC-1α/PPARγ signaling pathway. Inflammopharmacol 31, 1481–1493 (2023). https://doi.org/10.1007/s10787-023-01181-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-023-01181-5