Abstract
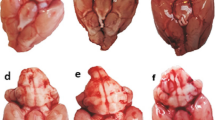
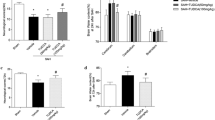
Melatonin (Mel) has been reported to alleviate early brain injury (EBI) following subarachnoid hemorrhage (SAH). The activation of silent information regulator 1 (Sirt1), a histone deacetylase, has been suggested to be beneficial in SAH. However, the precise role of Sirt1 in Mel-mediated protection against EBI following SAH has not been elucidated. The present study aims to evaluate the role of melatonin receptor/Sirt1/nuclear factor-kappa B (NF-κB) in this process. The endovascular perforation SAH model was used in male C57BL/6J mice, and melatonin was administrated intraperitoneally (150 mg/kg). The mortality, SAH grade, neurological score, brain water content, and neuronal apoptosis were evaluated. The expression of Sirt1, acetylated-NF-κB (Ac-NF-κB), Bcl-2, and Bax were detected by western blot. To study the underlying mechanisms, melatonin receptor (MR) antagonist luzindole and Sirt1 small interfering RNA (siRNA) were administrated to different groups. The results suggest that Mel improved the neurological deficits and reduced the brain water content and neuronal apoptosis. In addition, Mel enhanced the expression of Sirt1 and Bcl-2 and decreased the expression of Ac-NF-κB and Bax. However, the protective effects of Mel were abolished by luzindole or Sirt1 siRNA. In conclusion, our results demonstrate that Mel attenuates EBI following SAH via the MR/Sirt1/NF-κB signaling pathway.







Similar content being viewed by others
References
van Gijn J, Kerr RS, Rinkel GJ (2007) Subarachnoid haemorrhage. Lancet 369(9558):306–318. doi:10.1016/S0140-6736(07)60153-6
Chen S, Feng H, Sherchan P, Klebe D, Zhao G, Sun X, Zhang J, Tang J et al (2014) Controversies and evolving new mechanisms in subarachnoid hemorrhage. Prog Neurobiol 115:64–91. doi:10.1016/j.pneurobio.2013.09.002
Sehba FA, Hou J, Pluta RM, Zhang JH (2012) The importance of early brain injury after subarachnoid hemorrhage. Prog Neurobiol 97(1):14–37. doi:10.1016/j.pneurobio.2012.02.003
Altay O, Suzuki H, Hasegawa Y, Caner B, Krafft PR, Fujii M, Tang J, Zhang JH (2012) Isoflurane attenuates blood–brain barrier disruption in ipsilateral hemisphere after subarachnoid hemorrhage in mice. Stroke J Cereb Circ 43(9):2513–2516. doi:10.1161/STROKEAHA.112.661728
Nornes H, Magnaes B (1972) Intracranial pressure in patients with ruptured saccular aneurysm. J Neurosurg 36(5):537–547. doi:10.3171/jns.1972.36.5.0537
Asano T, Sano K (1977) Pathogenetic role of no-reflow phenomenon in experimental subarachnoid hemorrhage in dogs. J Neurosurg 46(4):454–466. doi:10.3171/jns.1977.46.4.0454
Friedrich V, Flores R, Muller A, Bi W, Peerschke EI, Sehba FA (2011) Reduction of neutrophil activity decreases early microvascular injury after subarachnoid haemorrhage. J Neuroinflammation 8:103. doi:10.1186/1742-2094-8-103
Li J, Chen J, Mo H, Chen J, Qian C, Yan F, Gu C, Hu Q et al (2015) Minocycline protects against NLRP3 inflammasome-induced inflammation and P53-associated apoptosis in early brain injury after subarachnoid hemorrhage. Mol Neurobiol. doi:10.1007/s12035-015-9318-8
Cai J, Cao S, Chen J, Yan F, Chen G, Dai Y (2015) Progesterone alleviates acute brain injury via reducing apoptosis and oxidative stress in a rat experimental subarachnoid hemorrhage model. Neurosci Lett 600:238–243. doi:10.1016/j.neulet.2015.06.023
Zhang ZY, Yang MF, Wang T, Li DW, Liu YL, Zhang JH, Sun BL (2015) Cysteamine alleviates early brain injury via reducing oxidative stress and apoptosis in a rat experimental subarachnoid hemorrhage model. Cell Mol Neurobiol 35(4):543–553. doi:10.1007/s10571-014-0150-x
Yang YQ, Li H, Zhang XS, Li W, Huang LT, Yu Z, Jiang TW, Chen Q et al (2015) Inhibition of SENP3 by lentivirus induces suppression of apoptosis in experimental subarachnoid hemorrhage in rats. Brain Res 1622:270–278. doi:10.1016/j.brainres.2015.06.032
Liu F, Chen Y, Hu Q, Li B, Tang J, He Y, Guo Z, Feng H et al (2015) MFGE8/Integrin beta3 pathway alleviates apoptosis and inflammation in early brain injury after subarachnoid hemorrhage in rats. Exp Neurol. doi:10.1016/j.expneurol.2015.04.016
Li JG, Lin JJ, Wang ZL, Cai WK, Wang PN, Jia Q, Zhang AS, Wu GY et al (2015) Melatonin attenuates inflammation of acute pulpitis subjected to dental pulp injury. Am J Transl Res 7(1):66–78
Shim DW, Shin HJ, Han JW, Ji YE, Jang CH, Koppula S, Kang TB, Lee KH (2015) A novel synthetic derivative of melatonin, 5-hydroxy-2′-isobutyl-streptochlorin (HIS), inhibits inflammatory responses via regulation of TRIF-dependent signaling and inflammasome activation. Toxicol Appl Pharmacol. doi:10.1016/j.taap.2015.02.006
Galano A, Tan DX, Reiter RJ (2011) Melatonin as a natural ally against oxidative stress: a physicochemical examination. J Pineal Res 51(1):1–16. doi:10.1111/j.1600-079X.2011.00916.x
Amin AH, El-Missiry MA, Othman AI (2015) Melatonin ameliorates metabolic risk factors, modulates apoptotic proteins, and protects the rat heart against diabetes-induced apoptosis. Eur J Pharmacol 747:166–173. doi:10.1016/j.ejphar.2014.12.002
El-Missiry MA, Othman AI, Al-Abdan MA, El-Sayed AA (2014) Melatonin ameliorates oxidative stress, modulates death receptor pathway proteins, and protects the rat cerebrum against bisphenol-A-induced apoptosis. J Neurol Sci 347(1–2):251–256. doi:10.1016/j.jns.2014.10.009
Luchetti F, Canonico B, Betti M, Arcangeletti M, Pilolli F, Piroddi M, Canesi L, Papa S et al (2010) Melatonin signaling and cell protection function. FASEB J Off Publ Fed Am Soc Exp Biol 24(10):3603–3624. doi:10.1096/fj.10-154450
Ayer RE, Sugawara T, Chen W, Tong W, Zhang JH (2008) Melatonin decreases mortality following severe subarachnoid hemorrhage. J Pineal Res 44(2):197–204. doi:10.1111/j.1600-079X.2007.00508.x
Ersahin M, Toklu HZ, Cetinel S, Yuksel M, Yegen BC, Sener G (2009) Melatonin reduces experimental subarachnoid hemorrhage-induced oxidative brain damage and neurological symptoms. J Pineal Res 46(3):324–332. doi:10.1111/j.1600-079X.2009.00664.x
Aladag MA, Turkoz Y, Parlakpinar H, Ozen H, Egri M, Unal SC (2009) Melatonin ameliorates cerebral vasospasm after experimental subarachnoidal haemorrhage correcting imbalance of nitric oxide levels in rats. Neurochem Res 34(11):1935–1944. doi:10.1007/s11064-009-9979-7
Fang Q, Chen G, Zhu W, Dong W, Wang Z (2009) Influence of melatonin on cerebrovascular proinflammatory mediators expression and oxidative stress following subarachnoid hemorrhage in rabbits. Mediat Inflamm 2009:426346. doi:10.1155/2009/426346
Chen J, Qian C, Duan H, Cao S, Yu X, Li J, Gu C, Yan F et al (2015) Melatonin attenuates neurogenic pulmonary edema via the regulation of inflammation and apoptosis after subarachnoid hemorrhage in rats. J Pineal Res. doi:10.1111/jpi.12278
Rogina B, Helfand SL (2004) Sir2 mediates longevity in the fly through a pathway related to calorie restriction. Proc Natl Acad Sci U S A 101(45):15998–16003. doi:10.1073/pnas.0404184101
Wang Z, Zhang L, Liang Y, Zhang C, Xu Z, Zhang L, Fuji R, Mu W et al (2015) Cyclic AMP mimics the anti-ageing effects of calorie restriction by up-regulating sirtuin. Sci Rep 5:12012. doi:10.1038/srep12012
Hernandez-Jimenez M, Hurtado O, Cuartero MI, Ballesteros I, Moraga A, Pradillo JM, McBurney MW, Lizasoain I et al (2013) Silent information regulator 1 protects the brain against cerebral ischemic damage. Stroke J Cereb Circ 44(8):2333–2337. doi:10.1161/STROKEAHA.113.001715
Zheng Y, Hu Q, Manaenko A, Zhang Y, Peng Y, Xu L, Tang J, Tang J et al (2015) 17beta-Estradiol attenuates hematoma expansion through estrogen receptor alpha/silent information regulator 1/nuclear factor-kappa b pathway in hyperglycemic intracerebral hemorrhage mice. Stroke J Cereb Circ 46(2):485–491. doi:10.1161/STROKEAHA.114.006372
Zhou XM, Zhang X, Zhang XS, Zhuang Z, Li W, Sun Q, Li T, Wang CX et al (2014) SIRT1 inhibition by sirtinol aggravates brain edema after experimental subarachnoid hemorrhage. J Neurosci Res 92(6):714–722. doi:10.1002/jnr.23359
Chen J, Wang L, Wu C, Hu Q, Gu C, Yan F, Li J, Yan W et al (2014) Melatonin-enhanced autophagy protects against neural apoptosis via a mitochondrial pathway in early brain injury following a subarachnoid hemorrhage. J Pineal Res 56(1):12–19. doi:10.1111/jpi.12086
Wang Z, Ma C, Meng CJ, Zhu GQ, Sun XB, Huo L, Zhang J, Liu HX et al (2012) Melatonin activates the Nrf2-ARE pathway when it protects against early brain injury in a subarachnoid hemorrhage model. J Pineal Res 53(2):129–137. doi:10.1111/j.1600-079X.2012.00978.x
Hasegawa Y, Suzuki H, Altay O, Zhang JH (2011) Preservation of tropomyosin-related kinase B (TrkB) signaling by sodium orthovanadate attenuates early brain injury after subarachnoid hemorrhage in rats. Stroke J Cereb Circ 42(2):477–483. doi:10.1161/STROKEAHA.110.597344
Yu L, Sun Y, Cheng L, Jin Z, Yang Y, Zhai M, Pei H, Wang X et al (2014) Melatonin receptor-mediated protection against myocardial ischemia/reperfusion injury: role of SIRT1. J Pineal Res 57(2):228–238. doi:10.1111/jpi.12161
Ma Q, Chen S, Hu Q, Feng H, Zhang JH, Tang J (2014) NLRP3 inflammasome contributes to inflammation after intracerebral hemorrhage. Ann Neurol 75(2):209–219. doi:10.1002/ana.24070
Liu L, Fujimoto M, Kawakita F, Nakano F, Imanaka-Yoshida K, Yoshida T, Suzuki H (2015) Anti-vascular endothelial growth factor treatment suppresses early brain injury after subarachnoid hemorrhage in mice. Mol Neurobiol. doi:10.1007/s12035-015-9386-9
Sugawara T, Ayer R, Jadhav V, Zhang JH (2008) A new grading system evaluating bleeding scale in filament perforation subarachnoid hemorrhage rat model. J Neurosci Methods 167(2):327–334. doi:10.1016/j.jneumeth.2007.08.004
Garcia JH, Liu KF, Ho KL (1995) Neuronal necrosis after middle cerebral artery occlusion in Wistar rats progresses at different time intervals in the caudoputamen and the cortex. Stroke J Cereb Circ 26(4):636–642, discussion 643
Zhao L, An R, Yang Y, Yang X, Liu H, Yue L, Li X, Lin Y et al (2015) Melatonin alleviates brain injury in mice subjected to cecal ligation and puncture via attenuating inflammation, apoptosis, and oxidative stress: the role of SIRT1 signaling. J Pineal Res 59(2):230–239. doi:10.1111/jpi.12254
Leibowitz A, Volkov A, Voloshin K, Shemesh C, Barshack I, Grossman E (2015) Melatonin prevents kidney injury in a high salt diet induced hypertension model by decreasing oxidative stress. J Pineal Res. doi:10.1111/jpi.12287
Liu S, Guo Y, Yuan Q, Pan Y, Wang L, Liu Q, Wang F, Wang J et al (2015) Melatonin prevents neural tube defects in the offspring of diabetic pregnancy. J Pineal Res. doi:10.1111/jpi.12282
Yang Y, Jiang S, Dong Y, Fan C, Zhao L, Yang X, Li J, Di S et al (2015) Melatonin prevents cell death and mitochondrial dysfunction via a SIRT1-dependent mechanism during ischemic-stroke in mice. J Pineal Res 58(1):61–70. doi:10.1111/jpi.12193
Chen J, Chen G, Li J, Qian C, Mo H, Gu C, Yan F, Yan W et al (2014) Melatonin attenuates inflammatory response-induced brain edema in early brain injury following a subarachnoid hemorrhage: a possible role for the regulation of pro-inflammatory cytokines. J Pineal Res 57(3):340–347. doi:10.1111/jpi.12173
Reppert SM (1997) Melatonin receptors: molecular biology of a new family of G protein-coupled receptors. J Biol Rhythm 12(6):528–531
Hardeland R (2009) Melatonin: signaling mechanisms of a pleiotropic agent. BioFactors 35(2):183–192. doi:10.1002/biof.23
Bouatia-Naji N, Bonnefond A, Cavalcanti-Proenca C, Sparso T, Holmkvist J, Marchand M, Delplanque J, Lobbens S et al (2009) A variant near MTNR1B is associated with increased fasting plasma glucose levels and type 2 diabetes risk. Nat Genet 41(1):89–94. doi:10.1038/ng.277
Bonnefond A, Clement N, Fawcett K, Yengo L, Vaillant E, Guillaume JL, Dechaume A, Payne F, Roussel R, Czernichow S, Hercberg S, Hadjadj S, Balkau B, Marre M, Lantieri O, Langenberg C, Bouatia-Naji N, Meta-Analysis of G, Insulin-Related Traits C, Charpentier G, Vaxillaire M, Rocheleau G, Wareham NJ, Sladek R, McCarthy MI, Dina C, Barroso I, Jockers R, Froguel P (2012) Rare MTNR1B variants impairing melatonin receptor 1B function contribute to type 2 diabetes. Nature genetics 44 (3):297–301. doi:10.1038/ng.1053
Wu YH, Ursinus J, Zhou JN, Scheer FA, Ai-Min B, Jockers R, van Heerikhuize J, Swaab DF (2013) Alterations of melatonin receptors MT1 and MT2 in the hypothalamic suprachiasmatic nucleus during depression. J Affect Disord 148(2–3):357–367. doi:10.1016/j.jad.2012.12.025
Sande PH, Dorfman D, Fernandez DC, Chianelli M, Dominguez Rubio AP, Franchi AM, Silberman DM, Rosenstein RE et al (2014) Treatment with melatonin after onset of experimental uveitis attenuates ocular inflammation. Br J Pharmacol 171(24):5696–5707. doi:10.1111/bph.12873
Ozdemir G, Ergun Y, Bakaris S, Kilinc M, Durdu H, Ganiyusufoglu E (2014) Melatonin prevents retinal oxidative stress and vascular changes in diabetic rats. Eye 28(8):1020–1027. doi:10.1038/eye.2014.127
Dubocovich ML, Delagrange P, Krause DN, Sugden D, Cardinali DP, Olcese J (2010) International Union of Basic and Clinical Pharmacology. LXXV. Nomenclature, classification, and pharmacology of G protein-coupled melatonin receptors. Pharmacol Rev 62(3):343–380. doi:10.1124/pr.110.002832
Baba K, Benleulmi-Chaachoua A, Journe AS, Kamal M, Guillaume JL, Dussaud S, Gbahou F, Yettou K et al (2013) Heteromeric MT1/MT2 melatonin receptors modulate photoreceptor function. Sci Signal 6(296):ra89. doi:10.1126/scisignal.2004302
Brydon L, Roka F, Petit L, de Coppet P, Tissot M, Barrett P, Morgan PJ, Nanoff C et al (1999) Dual signaling of human Mel1a melatonin receptors via G(i2), G(i3), and G(q/11) proteins. Mol Endocrinol 13(12):2025–2038. doi:10.1210/mend.13.12.0390
Nosjean O, Nicolas JP, Klupsch F, Delagrange P, Canet E, Boutin JA (2001) Comparative pharmacological studies of melatonin receptors: MT1, MT2 and MT3/QR2. Tissue distribution of MT3/QR2. Biochem Pharmacol 61(11):1369–1379
Karbownik M, Reiter RJ (2000) Antioxidative effects of melatonin in protection against cellular damage caused by ionizing radiation. Proceed Soc Exp Biol Med Soc Exp Biol Med 225(1):9–22
Reiter RJ, Tan DX, Mayo JC, Sainz RM, Leon J, Czarnocki Z (2003) Melatonin as an antioxidant: biochemical mechanisms and pathophysiological implications in humans. Acta Biochim Pol 50(4):1129–1146
Cardinali DP, Pagano ES, Scacchi Bernasconi PA, Reynoso R, Scacchi P (2013) Melatonin and mitochondrial dysfunction in the central nervous system. Horm Behav 63(2):322–330. doi:10.1016/j.yhbeh.2012.02.020
Takuma K, Yan SS, Stern DM, Yamada K (2005) Mitochondrial dysfunction, endoplasmic reticulum stress, and apoptosis in Alzheimer’s disease. J Pharmacol Sci 97(3):312–316
Schwartz C, Ballinger MA, Andrews MT (2015) Melatonin receptor signaling contributes to neuroprotection upon arousal from torpor in thirteen-lined ground squirrels. Am J Physiol Regul Integ Comp Physiol 00292:02015. doi:10.1152/ajpregu.00292.2015
Tocharus C, Puriboriboon Y, Junmanee T, Tocharus J, Ekthuwapranee K, Govitrapong P (2014) Melatonin enhances adult rat hippocampal progenitor cell proliferation via ERK signaling pathway through melatonin receptor. Neuroscience 275:314–321. doi:10.1016/j.neuroscience.2014.06.026
Wongprayoon P, Govitrapong P (2015) Melatonin attenuates methamphetamine-induced neuroinflammation through the melatonin receptor in the SH-SY5Y cell line. Neurotoxicology 50:122–130. doi:10.1016/j.neuro.2015.08.008
Wu YH, Zhou JN, Van Heerikhuize J, Jockers R, Swaab DF (2007) Decreased MT1 melatonin receptor expression in the suprachiasmatic nucleus in aging and Alzheimer’s disease. Neurobiol Aging 28(8):1239–1247. doi:10.1016/j.neurobiolaging.2006.06.002
Etain B, Dumaine A, Bellivier F, Pagan C, Francelle L, Goubran-Botros H, Moreno S, Deshommes J et al (2012) Genetic and functional abnormalities of the melatonin biosynthesis pathway in patients with bipolar disorder. Hum Mol Genet 21(18):4030–4037. doi:10.1093/hmg/dds227
Adi N, Mash DC, Ali Y, Singer C, Shehadeh L, Papapetropoulos S (2010) Melatonin MT1 and MT2 receptor expression in Parkinson’s disease. Med Sci Monit Int Med J Exp Clin Res 16(2):BR61–67
Mitchell SJ, Martin-Montalvo A, Mercken EM, Palacios HH, Ward TM, Abulwerdi G, Minor RK, Vlasuk GP et al (2014) The SIRT1 activator SRT1720 extends lifespan and improves health of mice fed a standard diet. Cell reports 6(5):836–843. doi:10.1016/j.celrep.2014.01.031
Ran M, Li Z, Yang L, Tong L, Zhang L, Dong H (2015) Calorie restriction attenuates cerebral ischemic injury via increasing SIRT1 synthesis in the rat. Brain Res 1610:61–68. doi:10.1016/j.brainres.2015.03.043
Cho SH, Chen JA, Sayed F, Ward ME, Gao F, Nguyen TA, Krabbe G, Sohn PD et al (2015) SIRT1 deficiency in microglia contributes to cognitive decline in aging and neurodegeneration via epigenetic regulation of IL-1beta. J Neurosci Off J Soc Neurosci 35(2):807–818. doi:10.1523/JNEUROSCI.2939-14.2015
Bederson JB, Connolly ES Jr, Batjer HH, Dacey RG, Dion JE, Diringer MN, Duldner JE Jr, Harbaugh RE et al (2009) Guidelines for the management of aneurysmal subarachnoid hemorrhage: a statement for healthcare professionals from a special writing group of the Stroke Council, American Heart Association. Stroke J Cereb Circ 40(3):994–1025. doi:10.1161/STROKEAHA.108.191395
Broderick JP, Brott TG, Duldner JE, Tomsick T, Leach A (1994) Initial and recurrent bleeding are the major causes of death following subarachnoid hemorrhage. Stroke J Cereb Circ 25(7):1342–1347
Claassen J, Carhuapoma JR, Kreiter KT, Du EY, Connolly ES, Mayer SA (2002) Global cerebral edema after subarachnoid hemorrhage: frequency, predictors, and impact on outcome. Stroke J Cereb Circ 33(5):1225–1232
Cheng G, Wei L, Zhi-Dan S, Shi-Guang Z, Xiang-Zhen L (2009) Atorvastatin ameliorates cerebral vasospasm and early brain injury after subarachnoid hemorrhage and inhibits caspase-dependent apoptosis pathway. BMC Neurosci 10:7. doi:10.1186/1471-2202-10-7
Broughton BR, Reutens DC, Sobey CG (2009) Apoptotic mechanisms after cerebral ischemia. Stroke J Cereb Circ 40(5):e331–339. doi:10.1161/STROKEAHA.108.531632
Kalle AM, Mallika A, Badiger J, Alinakhi, Talukdar P, Sachchidanand (2010) Inhibition of SIRT1 by a small molecule induces apoptosis in breast cancer cells. Biochem Biophys Res Commun 401(1):13–19. doi:10.1016/j.bbrc.2010.08.118
Acknowledgments
This study was supported by grants from the National Natural Science Foundation of China (81222015, 81571215), the New Century Talent Supporting Project supported by Chinese Education Ministry (NCET-12-1004), and Leading Talents of Middle-Age and Young in S&T Innovation supported by Chinese Science and Technology Ministry (2013RA2181).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
None declared
Additional information
Lei Zhao, Haixiao Liu, and Liang Yue contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhao, L., Liu, H., Yue, L. et al. Melatonin Attenuates Early Brain Injury via the Melatonin Receptor/Sirt1/NF-κB Signaling Pathway Following Subarachnoid Hemorrhage in Mice. Mol Neurobiol 54, 1612–1621 (2017). https://doi.org/10.1007/s12035-016-9776-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-016-9776-7