Abstract
Chronic inflammation is pathologically associated with various clinical conditions such as rheumatoid arthritis. Several anti-inflammatory and analgesic drugs currently available in market presents a wide range of problems. Therefore, the current study was aimed to evaluate anti-inflammatory and analgesic activities of newly synthesized compound 2-(5-mercapto-1,3,4-oxadiazol-2-yl)-N-propylbenzenesulphonamide (MOPBS). Carrageenan and CFA-induced models were developed for evaluation of anti-inflammatory and analgesic activity. Quantitative real-time PCR (qRT-PCR) was performed to determine the mRNA expression levels of inflammatory mediators. Pain behaviours were evaluated by performing Von Frey, Randall Selitto, cold acetone and hot plate test respectively. X-ray imaging and haematoxylin and eosin (H&E) staining were performed for examination of soft tissues of treated mice paw. Additionally, Kodzeila’s screen test and weight test were performed for determination of any side effects on motor function and muscle strength. Acute pretreatment of animals with MOPBS (1, 10, 50 and 100 mg/kg, i.p.) produced a significant reduction of paw oedema against carrageenan-induced acute inflammation as well as notable inhibition of mechanical hyperalgesia, allodynia and thermal hyperalgesia. Similarly, in chronic inflammation model, administration of MOPBS (50 mg/kg, i.p.) produced a remarkable reduction of paw oedema. Additionally, MOPBS pretreatment showed a significant inhibition of thermal hyperalgesia, mechanical allodynia, and mechanical hyperalgesia in chronic arthritis model. Several pro-inflammatory mediators such as nitric oxide (NO), vascular endothelial growth factor (VEGF), interleukins (IL-1β, IL-6) and tumor necrosis factor-α (TNF-α) were inhibited by MOPBS treatment in blood plasma and paw tissues, respectively. MOPBS also enhanced the mRNA expression levels of nuclear factor (erythroid-derived 2)-like 2 (Nrf2), superoxide dismutase (SOD2) and heme oxygenase (HO-1) and in turn reduced arthritis severity and inflammation. Furthermore, anti-inflammatory data were confirmed by X-rays and histological analysis. MOPBS pretreatment did not produce any apparent toxic effect on gastric, kidney and liver function and on muscle strength and motor function. Hence, the present data suggest that MOPBS might be a candidate for several chronic inflammatory diseases such RA and other auto-immune diseases.








Similar content being viewed by others
Change history
10 March 2018
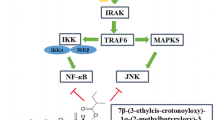
Unfortunately, Fig. 1 was incorrectly published in the original publication. The corrected Fig. 1 is given as below
References
Amarasekara DS, Yu J, Rho J (2015) Bone loss triggered by the cytokine network in inflammatory autoimmune diseases bone. J Immunol Res 150:3
Arora R, Kuhad A, Kaur I, Chopra K (2015) Curcumin loaded solid lipid nanoparticles ameliorate adjuvant-induced arthritis in rats. Eur J Pain 19:940–952
Barksby H, Lea S, Preshaw P, Taylor J (2007) The expanding family of interleukin-1 cytokines and their role in destructive inflammatory disorders. Clin Exp Immunol 149:217–225
Bertorelli R, Corradini L, Rafiq K, Tupper J, Calò G, Ongini E (1999) Nociceptin and the ORL-1 ligand [Phe1ψ (CH2-NH) Gly2] nociceptin (1–13) NH2 exert anti-opioid effects in the Freund’s adjuvant-induced arthritic rat model of chronic pain. Br J Pharmacol 128:1252–1258
Bölcskei K et al (2005) Investigation of the role of TRPV1 receptors in acute and chronic nociceptive processes using gene-deficient mice. Pain 117:368–376
Brennan FM, Hayes AL, Ciesielski CJ, Green P, Foxwell BM, Feldmann M (2002) Evidence that rheumatoid arthritis synovial T cells are similar to cytokine-activated T cells: involvement of phosphatidylinositol 3-kinase and nuclear factor κB pathways in tumor necrosis factor α production in rheumatoid arthritis. Arthritis Rheum 46:31–41
Brzozowski T, Konturek PC, Konturek S, Sliwowski Z, Drozdowicz D, Kwiecień S (2000) Gastroprotective and ulcer healing effects of nitric oxide-releasing non-steroidal anti-inflammatory drugs. Dig Liver Dis 32(7):583–594
Cai X et al (2006) The comparative study of Sprague-Dawley and Lewis rats in adjuvant-induced arthritis. Naunyn-Schmiedeberg’s Arch Pharmacol 373:140
Contet C, Rawlins JNP, Deacon RM (2001) A comparison of 129S2/SvHsd and C57BL/6JOlaHsd mice on a test battery assessing sensorimotor, affective and cognitive behaviours: implications for the study of genetically modified mice. Behav Brain Res 124:33–46
Costa B, Colleoni M, Conti S, Parolaro D, Franke C, Trovato AE, Giagnoni G (2004) Oral anti-inflammatory activity of cannabidiol, a non-psychoactive constituent of cannabis, in acute carrageenan-induced inflammation in the rat paw. Naunyn-Schmiedeberg’s Arch Pharmacol 369:294–299
da Silva AJ et al (2016) Anti-nociceptive, anti-hyperalgesic and anti-arthritic activity of amides and extract obtained from Piper amalago in rodents. J Ethnopharmacol 179:101–109
Deacon RM, Croucher A, Rawlins JNP (2002) Hippocampal cytotoxic lesion effects on species-typical behaviours in mice. Behav Brain Res 132:203–213
El-Sabbagh OI (2013) Synthesis of some new benzisothiazolone and benzenesulfonamide derivatives of biological interest starting from saccharin sodium. Arch Pharm 346:733–742
Feldman M, Maini R (2008) Role of cytokines in rheumatoid arthritis: an education in pathophysiology ant therapeutics. Immunol Rev 223:7–19
Gibofsky A (2014) Current therapeutic agents and treatment paradigms for the management of rheumatoid arthritis. Am J Manag Care 20:S136–S144
Hernstadt H, Wang S, Lim G, Mao J (2009) Spinal translocator protein (TSPO) modulates pain behavior in rats with CFA-induced monoarthritis. Brain Res 1286:42–52
Hess A et al (2011) Blockade of TNF-α rapidly inhibits pain responses in the central nervous system. Proc Natl Acad Sci USA 108:3731–3736
Hitchon CA, El-Gabalawy HS (2004) Oxidation in rheumatoid arthritis. Arthritis Res Ther 6:265
Horváth Á et al (2016) Transient receptor potential ankyrin 1 (TRPA1) receptor is involved in chronic arthritis: in vivo study using TRPA1-deficient mice. Arthr Res Ther 18:6
Jia P, Chen G, Qin W-Y, Zhong Y, Yang J, Rong X-F (2016) Xitong Wan attenuates inflammation development through inhibiting the activation of nuclear factor-κB in rats with adjuvant-induced arthritis. J Ethnopharmacol 193:266–271
Kang SY et al (2008) The anti-arthritic effect of ursolic acid on zymosan-induced acute inflammation and adjuvant-induced chronic arthritis models. J Pharm Pharmacol 60:1347–1354
Keffer J, Probert L, Cazlaris H, Georgopoulos S, Kaslaris E, Kioussis D, Kollias G (1991) Transgenic mice expressing human tumour necrosis factor: a predictive genetic model of arthritis. EMBO J 10:4025
Khan S, Shin EM, Choi RJ, Jung YH, Kim J, Tosun A, Kim YS (2011) Suppression of LPS-induced inflammatory and NF-κB responses by anomalin in RAW 264.7 macrophages. J Cell Biochem 112:2179–2188
Khan S, Choi RJ, Shehzad O, Kim HP, Islam MN, Choi JS, Kim YS (2013a) Molecular mechanism of capillarisin-mediated inhibition of MyD88/TIRAP inflammatory signaling in in vitro and in vivo experimental models. J Ethnopharmacol 145:626–637
Khan S, Shehzad O, Chun J, Kim YS (2013b) Mechanism underlying anti-hyperalgesic and anti-allodynic properties of anomalin in both acute and chronic inflammatory pain models in mice through inhibition of NF-κB, MAPKs and CREB signaling cascades. Eur J Pharmacol 718:448–458
Khan S et al (2014) Anti-hyperalgesic and anti-allodynic activities of capillarisin via suppression of inflammatory signaling in animal model. J Ethnopharmacol 152:478–486
Koenders MI et al (2005) Induction of cartilage damage by overexpression of T cell interleukin-17A in experimental arthritis in mice deficient in interleukin-1. Arthritis Rheum 52:975–983
Longo G, Osikowicz M, Ribeiro-da-Silva A (2013) Sympathetic fiber sprouting in inflamed joints and adjacent skin contributes to pain-related behavior in arthritis. J Neurosci 33:10066–10074
Lu J et al (2000) Vascular endothelial growth factor expression and regulation of murine collagen-induced arthritis. J Immunol 164:5922–5927
MacNaul K, Hutchinson N, Parsons J, Bayne E, Tocci M (1990) Analysis of IL-1 and TNF-alpha gene expression in human rheumatoid synoviocytes and normal monocytes by in situ hybridization. J Immunol 145:4154–4166
Mohammadi S, Christie MJ (2014) α9-nicotinic acetylcholine receptors contribute to the maintenance of chronic mechanical hyperalgesia, but not thermal or mechanical allodynia. Mol Pain 10:64
Schaible HG, Ebersberger A, Banchet GS (2002) Mechanisms of pain in arthritis. Ann N Y Acad Sci 966:343–354
Siebert S, Tsoukas A, Robertson J, McInnes I (2015) Cytokines as therapeutic targets in rheumatoid arthritis and other inflammatory diseases. Pharmacol Rev 67:280–309
Spurlock CF, Tossberg JT, Matlock BK, Olsen NJ, Aune TM (2014) Methotrexate inhibits NF-κB activity via long intergenic (noncoding) RNA–p21 induction. Arthr Rheumatol 66:2947–2957
Syed T, Akhtar T, Al-Masoudi NA, Jones PG, Hameed S (2011) Synthesis, QSAR and anti-HIV activity of new 5-benzylthio-1, 3, 4-oxadiazoles derived from α-amino acids. J Enzyme Inhib Med Chem 26:668–680
Talalay P, Dinkova-Kostova AT, Holtzclaw WD (2003) Importance of phase 2 gene regulation in protection against electrophile and reactive oxygen toxicity and carcinogenesis. Adv Enzyme Regul 43:121–134
Terker AS et al (2015) Potassium modulates electrolyte balance and blood pressure through effects on distal cell voltage and chloride. Cell Metab 21:39–50
Walz D, DiMartino M, Misher A (1971) Adjuvant-induced arthritis in rats. II. Drug effects on physiologic, biochemical and immunologic parameters. J Pharmacol Exp Ther 178:223–231
Yu Y, Xiong Z, Lv Y, Qian Y, Jiang S, Tian Y (2006) In vivo evaluation of early disease progression by X-ray phase-contrast imaging in the adjuvant-induced arthritic rat. Skeletal Radiol 35:156–164
Zuo J, Xia Y, Li X, J-w Chen (2014) Therapeutic effects of dichloromethane fraction of Securidaca inappendiculata on adjuvant-induced arthritis in rat. J Ethnopharmacol 153:352–358
Acknowledgements
This work was supported by the SRGP start-up Grant (21-357/SRGP/R&D/HEC/2014) supported by HEC, Government of Pakistan. The authors are thankful to National Research Foundation (NRF), South Korea, Seoul National University, Grant funded by the Korean Government (MSIP) (No. 2009-0083533).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rasheed, H., Afridi, R., Khan, A.U. et al. Anti-inflammatory, anti-rheumatic and analgesic activities of 2-(5-mercapto-1,3,4-oxadiazol-2-yl)-N-propylbenzenesulphonamide (MOPBS) in rodents. Inflammopharmacol 26, 1037–1049 (2018). https://doi.org/10.1007/s10787-018-0446-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-018-0446-4