Abstract
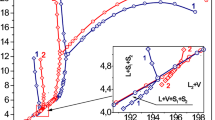
It is of much interest to study the heavy hydrocarbon impact on the phase behavior of hydrocarbons. Using a method of the precision adiabatic calorimetry the phase equilibria in quaternary mixtures of methane, propane, octane, and nonane have been studied. The phase diagrams for investigated mixtures have been plotted based on the experimental data. The phase transitions were localized by the finite discontinuities in temperature derivatives of the thermodynamic potentials. The heat capacity, internal energy, pressure, and temperature derivative of pressure at constant volume were measured in the range 170–320 K, up to 40 MPa. Quaternary mixtures as quasi-binary mixtures are presented. The first quasi-component of the quasi-binary mixture is the binary mixture with the constant methane/propane ratio. The second quasi-component of the quasi-binary mixture is the binary mixture of octane and nonane. Investigated hydrocarbon system is the combination of the simple hydrocarbon mixtures for the low concentration of octane and nonane. In the earlier papers we presented the simple hydrocarbon mixtures for the low concentration of octane, nonane, and decane, and quaternary mixtures of methane, propane, octane, and decane for the low concentration of octane and decane, which are the combination of the simple hydrocarbon mixtures for the low concentration of octane and decane. Our investigations show that quaternary hydrocarbon mixture for the low concentration of octane and nonane as well as the simple hydrocarbon mixtures for the low concentration of octane, nonane, and decane split into three phases, the macrophase composed of methane, propane, octane, and nonane, and two microphases formed by octane and nonane. In this system octane and nonane dissolved in the macrophase partly. Besides, the heavy components provoke a split of the liquid part of the quaternary mixture into two liquid phases.







Similar content being viewed by others
Data Availability
Not applicable.
Abbreviations
- p :
-
Pressure [MPa]
- T :
-
Temperature [K]
- U :
-
Internal energy [J]
- x :
-
Concentration [mole fraction]
- ρ :
-
Density [kg·m−3]
- C V = (∂U/∂T)V :
-
Heat capacity at constant volume [kJ·kg−1·K−1]
- (∂p/∂T)V :
-
Temperature derivative of pressure at constant volume (the thermal pressure coefficient) [MPa·K−1]
- V:
-
Vapor phase
- L(S):
-
Single-phase macrophase composed of methane, propane, and partly dissolved octane and nonane
- LV(S):
-
Two-phase macrophase composed of methane, propane, and partly dissolved octane and nonane
- L(SC9):
-
Single-phase macrophase composed of methane, propane, octane, and partly dissolved nonane
- LV(SC9):
-
Two-phase macrophase composed of methane, propane, octane, and partly dissolved nonane
- L1 :
-
Liquid phase of the quaternary mixture (octane-, nonane-lean phase)
- L2 :
-
Liquid phase of the quaternary mixture (octane-, nonane-rich phase)
- SC8 :
-
Microphase formed by octane
- SC9 :
-
Microphase formed by nonane
- SC8+C9 :
-
Microphase formed by octane and nonane
References
A. Tarek, Equations of State and PVT Analysis, 2nd edn. (Gulf Professional Publishing, Elsevier, 2016)
N.B. Vargaftic, Reference Book on Gas-Liquid Thermophysical Properties, (Moscow, 1972), p. 720. [In Russian]
K.S. Pedersen, P.L. Christensen, J.A. Shaikh, Phase Behavior of Petroleum Reservoir Fluids (Taylor & Francis Group, LLC, Milton Park, 2015)
S.M. Walas, Phase Equilibria in Chemical Engineering, Butterworth publishers. Department of Chemical and Petroleum Engineering. University of Kansas and the C.W. Nofsinger Company, p. 664.
V.M. Buleiko, D.V. Buleiko, Int. J. Thermophys. 41, 27 (2020). https://doi.org/10.1007/s10765-020-2602-5
V.M. Buleiko, D.V. Buleiko, Int. J. Thermophys. 42, 85 (2021). https://doi.org/10.1007/s10765-021-02835-8
V.M. Buleiko, D.V. Buleiko, Int. J. Thermophys. 43, 60 (2022). https://doi.org/10.1007/s10765-022-02988-0
V.M. Buleiko, D.V. Buleiko, Int. J. Thermophys. 44, 77 (2023). https://doi.org/10.1007/s10765-023-03193-3
V.M. Buleiko, B.A. Grigoriev, V.A. Istomin, Fluid Phase Equilib. 441, 64 (2017)
V.M. Buleiko, B.A. Grigoriev, J. Mendoza, Fluid Phase Equilib. 462, 14 (2018)
H.E. Stanley, Introduction to Phase Transitions and Critical Phenomena (Clarendon Press, Oxford, 1971)
M.A. Anisimov, A.V. Voronel, T.M. Ovodova, Sov. Phys.—JETP 34, 583 (1972)
A.V. Voronel, Thermal Measuremts and Critical Phenomena in Liquids, 12th School of Modern Physics on Phase Transitions and Critical Phenomena, (Tel Aviv U., 1976), p. 343
Acknowledgments
This work was supported by the Russian Science Foundation under Grant No. 23-29-00088, https://rscf.ru/en/project/23-29-00088/.
Funding
Funding was provided by Poccийcкий Hayчный Фoнд (PHФ) (Grant Number 23-29-00088), https://rscf.ru/en/project/23-29-00088/.
Ethics declarations
Conflict of interest
I declare that the authors have no competing interests as defined by Springer, or other interests, that might be perceived to influence the results and/or discussion reported in this paper.
Ethical Approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Buleiko, V.M., Buleiko, D.V. An Experimental Study of the Heavy Hydrocarbon Impact on the Phase Behavior of Hydrocarbons. Int J Thermophys 44, 125 (2023). https://doi.org/10.1007/s10765-023-03234-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10765-023-03234-x