Abstract
Group-living in primates provides benefits but also entails competition and aggression. To retain the positive fitness-enhancing advantages of social bonding, individuals can reconcile past aggression. Reconciliation has been extensively studied in haplorrhine primates and occurs more frequently in more tolerant species, but whether this pattern extends to strepsirrhine primates, including lemurs, remains unclear, particularly given the differences in behavioral biology between the two primate suborders. We examined the occurrence of reconciliation behavior and its relationship with dominance hierarchy characteristics in wild populations of Propithecus diadema and Eulemur fulvus. We collected behavioral data on dominance and affiliation behaviors in the Maromizaha Protected Area, Madagascar, focusing on three P. diadema groups and two E. fulvus groups over 14 months between 2015 and 2020. The dominance hierarchies differed between species, with P. diadema exhibiting steeper within-group hierarchies than E. fulvus. We found evidence of reconciliation behavior in P. diadema, as affiliation was more likely to follow conflicts than during control periods, but we did not find this effect in E. fulvus. Furthermore, reconciliation tendencies in P. diadema were higher in male–male dyads and those with disparate ranks. These findings suggest that dominance hierarchy structure may be an important factor influencing reconciliation behavior across primates, although the nuances of the relationship seem to vary between our two strepsirrhine study species and traditional haplorrhine patterns. Further research is needed to determine whether these patterns are species-, site-, or group-specific. In conclusion, these results further our understanding of the differences between strepsirrhine and haplorrhine social evolution.
Abstract in French
Chez les primates, vivre en groupes offre des avantages mais cause aussi compétition et agressions. Pour retenir des avantages positifs des liens sociales pour la valeur sélective, des individus peuvent réconcilier des agressions passées. Le comportement de réconciliation a été bien étudié chez les haplorrhiniens et se passent plus dans des espèces tolérantes, mais si ces tendances s’appliquer chez les primates strepsirrhiniens n’est pas bien compris, notamment à cause des différences dans leur biologie comportemental entre les deux sous-ordres. Cette étude examine le comportement de réconciliation en relation avec des caractéristiques des hiérarchies de dominance chez deux de lémuriens : Propithecus diadema et Eulemur fulvus. Des données ont été collectées dans trois groupes de P. diadema et deux groupes de E. fulvus vivant dans l’aire protégée de Maromizaha à Madagascar, pendant une période de 14 mois entre 2015 et 2020. Les hiérarchies de dominance intra-groupe ont été différentes selon les espèces. La pente de la hiérarchie est plus marquée chez P. diadema que chez E. fulvus. Nous avons démontré l’existence d’un comportement de réconciliation chez P. diadema, chez qui la probabilité d’occurrence de comportements affiliatifs est plus élevée après les conflits que dans la période contrôle, mais nous n’avons pas observée cet effet chez E. fulvus. De plus, la fréquence de comportements de réconciliation chez P. diadema a été élevée dans des dyades avec deux males ou dyades plus différentes dans les rangs. Ces résultats suggèrent que la structures des hiérarchies de dominances peuvent être un facteur important qui influence le comportement de réconciliation, mais les nuances dans cette relation peuvent être variées entre notre deux espèces d’études strepsirrhiniens et des tendances traditionnelles haplorrhiniens. Des recherches supplémentaires seront nécessaires pour déterminer si ces résultats sont révélateurs de différences entre espèces, entre sites, ou entres groupes. En conclusion, ces résultats approfondissent notre compréhension des différences dans l'évolution sociale pour les strepsirrhiniens et les haplorrhiniens. *The publisher did not copy edit the translated abstract.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Living in groups can increase interspecific competition over food, space, mates, and other resources (Arnold et al., 2011; Sterck et al., 1997; Wrangham, 1980). Consequently, recurring, intragroup aggressive behaviors such as threats, displays, and physical confrontations, lead to the establishment of dominance hierarchies (de Waal, 1986; de Waal & Luttrell, 1985; Drews, 1993; Lewis, 2019). These hierarchies are an emergent phenomenon due to the individual’s consistent competitiveness for resources (Tibbetts et al., 2022; Wittig & Boesch, 2003). In addition to engaging in dominance behaviors, individuals living in groups spend time participating in affiliative behaviors to enhance social bonds, where bondedness is associated with higher fitness (Campos et al., 2020; Ellis et al., 2019; Feldblum et al., 2021; McFarland et al., 2017; Schulke et al., 2010; Silk et al., 2003, 2009). This creates a tradeoff between competing with an individual for resources and enhancing the social bond with that individual through affiliative behaviors such as grooming and spending time in contact.
Given this tradeoff, conflict mediation is critical for maintaining strong social bonds when conflicts over resources occur. Reconciliation is one such mechanism that dyads can use to directly mediate any negative consequences of aggression on their relationship (Aureli & van Schaik, 1991; Das et al., 1998; Kutsukake & Castles, 2001; Silk, 2002; Sussman et al., 2005). Reconciliation is defined as affiliation between former opponents that takes place soon after a conflict to restore tolerance and reduce chances of future conflict (Cords, 1992). Determining what qualifies as ‘soon enough’ for reconciliation after a conflict requires a thorough understanding of the typical social dynamics of a species, or even more specifically of a group or dyad, in the absence of conflict. Given the interspecies variability in sociality, simple time-based definitions for reconciliation are insufficient (Sussman et al., 2005). One solution to identify reconciliation behavior is to compare the latency to affiliate after a conflict with the latency to affiliate during control periods without confict (Arnold et al., 2011).
A prominent hypothesis for the evolutionary function of reconciliation is the valuable relationship hypothesis, which suggests that reconciliation serves to settle conflicts between combatants whose relationship generates mutual fitness benefits (de Waal & Aureli, 1997). Thus, according to the hypothesis, reconciliation is expected to occur more frequently between close affiliative partners. This prediction has been supported in a variety of species, especially in catarrhine monkeys (as reviwed in Arnold et al., 2011). Many reconciliation studies have focused on Macaca, a genus known for its interspecific variation in the style of dominance hierarchies. Macaques exhibit a range of dominance hierarchies from highly despotic to highly tolerant (Balasubramaniam et al., 2012; Thierry, 2000). In groups with highly despotic hierarchies, social bonding opportunities are limited due to the high selectivity for kin in affiliative social interactions (Amici et al., 2020; Aureli et al., 1997), leading to relatively weak social bonds and low social tolerance. If strong despotism limits social bonding, and social bonding is dependent on repair from reconciliation, it follows that despotic species should have lower rates of reconciliation. Indeed, macaque species that are considered more tolerant tend to reconcile more than despotic species (Berman et al., 2004; Cooper & Bernstein, 2002; de Waal & Luttrell, 1985; de Waal & Ren, 1988; Demaria & Thierry, 2001; Thierry, 2000).
Due to the differences in primate societies across species and the ubiquity of affiliative and agonistic behaviors, understanding the evolutionary function of reconciliation requires investigations of the behavior across a wide range of taxa, social systems, and ecological conditions. However, studies of reconciliation are not evenly distributed across the order Primates. Reconciliation behavior has been studied extensively in catarrhine monkeys and other haplorhine primates (as reviewed in Arnold et al., 2011). Haplorhines tend to engage in affiliative behaviors, such as playing, handholding, grooming, and initiating physical contact, sooner after a conflict than in control periods (Arnold et al., 2011). Various demographic factors (such as kinship, rank, and sex) and ecological factors (such as seasonality) influence the probability of reconciliation, sometimes in species-specific ways (Kin: Aureli et al., 1997; Castles & Whiten, 1998; Judge, 1991; Kutsukake & Castles, 2001; Majolo et al., 2009; Palagi & Norscia, 2011; Rank: Aureli et al., 1993; Judge, 1991; Sex: Cordoni et al., 2006; Leca et al., 2002; Palagi et al., 2005; Penate et al., 2009; Schino et al., 1998; Season: Majolo & Koyama, 2006). Across species, dyads that are close social affiliates outside of the conflict are also more likely to reconcile (Castles et al., 1996; de Waal & Yoshihara, 1983; Kappeler & van Schaik, 1992), which supports the valuable relationship hypothesis (de Waal & Aureli, 1997).
Investigations of potential reconciliation behavior are much rarer in strepsirrhine primates (as reviewed in Arnold et al., 2011; Fig. 1). Most of the published studies focused on Lemuridae (20/22 studied groups) or groups living in captivity (13/22 studied groups). The presence and characteristics of reconciliation appear to vary within and across species of strepsirrhines, and do not cluster in the same clade (Fig. 1). For example, in Lemur catta, reconciliation was observed in some circumstances (Palagi et al., 2005; Palagi & Norscia, 2015; Rolland & Roeder, 2000), but not all (Kappeler, 1993a). Furthermore, in closely related Eulemur species, some species show reconciliation behavior (Kappeler, 1993a; Norscia & Palagi, 2011; Roeder et al., 2002), and others do not (Roeder et al., 2002). Factors that explain this apparent variation in reconciliation in lemurs may be connected to aspects of the dominance hierarchy, group demographics, seasonality, and intensity of fights. While the current literature does not suggest that phylogenetic relatedness among species predicts the presence or absence of reconciliation behavior, the lack of phylogenetic diversity in the currently published studies (Fig. 1) makes it difficult to fully understand how these patterns are organized across strepsirrhine species.
Phylogenetic tree showing the results of reconciliation studies in Lemuridae and Indriidae (Eulemur fulvus: Roeder et al., 2002; Eulemur macaco: Roeder et al., 2002; Eulemur fulvus rufus: Kappeler, 1993a; Palagi & Norscia, 2011; Lemur catta: Kappeler, 1993a, Rolland & Roeder, 2000, Palagi & Norscia, 2015, Palagi et al., 2005; Propithecus verreauxi: Lewis, 2019; Palagi et al., 2008). Phylogenetic data from 10K Trees v3 (Arnold et al., 2010).
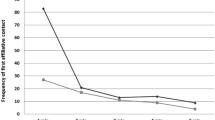
One challenge of the lack of phylogenetic diversity in the study of reconciliation across primate species is that strepsirrhines and haplorrhines fundamentally differ in several aspects of behavior and biology (Simmen et al., 2021) that may be salient to reconciliation behavior. For example, the social behavior of strepsirrhines and haplorrhines differs (van Schaik & Kappeler, 1996). Specifically, strepsirrhines tend to have low levels of affiliative contact (Jolly, 1998; Sussman et al., 2005), and grooming plays a less crucial role in their social networks in comparision to non-grooming behaviors (Kulachi et al., 2015, 2018; Lewis, 2019 — although see Lewis, 2010). In contrast, haplorrhines tend to affiliate by physical contact, making grooming and time spent in contact good measures for reconciliation after a conflict (McFarland & Majolo, 2013). The generally low levels of affiliative contact in strepsirrhines combined with their generally small group sizes compared to haplorrhines (Wright, 1999) makes triadic interactions (Jolly, 1966) such as polyadic conflicts (Kappeler, 1993b), coalitions, or alliances less common. In other words, individual strepsirrhines in smaller groups with fewer affiliative interactions have fewer social relationships to manage, without many chances for triadic or more complex relationships to form. This could impact reconciliation by reducing the necessity of and opportunities for complex social strategies to repair relationships after conflicts. Furthermore, strepsirrhines rely more on olfactory communication than haplorrhines (Fichtel & Kappeler, 2022). In fact, sizes of the accessory olfactory bulb, which processes social information and pheromones (Keverne, 1999), systematically vary with the social system in strepsirrhines (Barton, 2006). This may influence reconciliation behavior because individuals can assess conspecifics without any physical contact, lessening the chance of conflicts over space. Lastly, lemurs are seasonal breeders, and most show female dominance (Kappeler et al., 2022; van Schaik & Kappeler, 1996; Wright, 1999). These two concepts together can limit the importance of some social bond types and can also increase competition during the short mating season (Wright, 1999). Therefore, it is possible that conflicts during the mating season are too important to reconcile. In fact, reconciliation in Lemur catta is less common during the mating season than in the non-mating season (Schino et al., 1998; Palagi & Norscia, 2015). Thus, due to these fundamental differences between strepsirrhines and haplorrhines, fully understanding the purpose and structure of reconciliation behavior requires studying it across taxa of diverse social systems, including strepsirrhines.
Propithecus diadema and Eulemur fulvus live in sympatry in the central-eastern rainforests of Madagascar yet show notable contrasts in their respective dominance hierarchies (Kappeler et al., 2022). P. diadema lives in stable groups that are usually between two and 11 individuals and include one or more adult males and one or more adult females (Lutz et al., 2019; Powzyk & Mowry, 2003; Rasolonjatovo & Irwin, 2020). Similarly, E. fulvus lives in multi-male/multi-female social groups that can range between three and ten individuals (Ganzhorn, 1988). While Propithecus species generally show female dominance (Irwin, 2006; Pochron et al., 2003; Ramanamisata et al., 2014; Richard & Nicoll, 1987), with a higher tendency for female aggression towards males (Rasolonjatovo & Irwin, 2020), in Eulemur fulvus, females and males share near equal status (Kappeler, 1993a; Kappeler et al., 2022), and sex does not have a significant effect on the distribution of grooming and agonistic interactions (Kappeler, 1993a; Pereira et al., 1990). In fact, both male and female E. fulvus can obtain low and high dominance ranks when resources are limited (Roeder & Fornasieri, 1995). Reconciliation has been observed in captive E. fulvus (Roeder et al., 2002), but to our knowledge, it has not been studied in wild P. diadema or E. fulvus. Thus, in the present study, we investigate whether these two lemur species show reconciliation behavior in the wild.
Using five years of observational data that we collected on three Propithecus diadema groups and two Eulemur fulvus groups, we aimed to provide a more comprehensive understanding of reconciliation behavior, particularly by examining intraspecies variation across groups in strepsirrhines. To that end, we analyzed the dominance hierarchies and reconciliatory tendencies of both species. By combining the valuable relationship hypothesis and the differences between strepsirrhines and haplorrhines, we hypothesize that differences in dominance structures will correlate with reconciliatory tendencies. Specifically, we predict that the species with steeper dominance hierarchies, as a proxy for the despotism present in a group, will exhibit lower reconciliatory tendencies. Lastly, given that reconciliation in haplorrhines is often dependent on demographic attributes, we investigate whether our two strepsirrhine species follow similar demographic patterns of sex and rank impacting reconciliation rates.
Methods
Study Site
We collected all data in the Maromizaha Protected Area (S18°58.6'; E48°27.9'). This 21.5 km2 protected rainforest is in the corridor between Ankeniheny and Zahamena (CAZ; Andasibe commune, Alaotra-Mangoro region) in Madagascar’s eastern mountains and is managed by Le Groupe d’Etudes et de Recherche sur les Primates de Madagascar (GERP).
Study Groups
We collected observational data on three groups of Propithecus diadema and two groups of Eulemur fulvus across 14 months between 2015 and 2020 (Table I; full subject table available as Table S1 in the electronic supplementary material). Previous researchers habituated the P. diadema groups in 2010, and we habituated the E. fulvus groups in 2019 prior to starting data collection.
Behavioral Data Collection
We collected observational data on affiliative and agonistic behavior using continuous recording focal sampling (Altmann, 1974). All group members served as focal animals, and the observational order was chosen according to a pre-randomized list. Our focal samples varied in length over the course of the study, since we originally collected the observational data used in this study for other related purposes. While focal samples from 2015 and 2016 were 30 minutes long, we increased focal sample length to 60 minutes starting in 2018. The last author collected data from 2015–2020 and was aided in 2019–2020 by a team of seven additional, trained observers. From 2015–2018, a team of two to three people followed a single study group. From 2019–2020, three observation teams (of two to three people each) rotated across the four study groups to ensure relatively even sampling across study groups.
We used a consistent ethogram across time when collecting the original data that expanded over time. From 2015–2016, we collected data on only affiliative (social play, contact, and allogrooming) and aggressive behaviors (threat). Beginning in 2018, we expanded the set of aggressive behaviors to include biting, chasing, displacing, hitting, and lunging. Full ethogram definitions of all behaviors are available in the electronic supplementary material (Table S2). For each social behavior in which the focal animal was involved, we recorded the start time, end time, initiator, and recipient. The final data set included 1422.9 hours of observations on Propithecus diadema and 631 hours on Eulemur fulvus (Table I).
Post Conflict Matching
Following methodology by Palagi et al. (2008) for studying reconciliation in Propithecus verreauxi, we defined the post-conflict (PC) period as the 15 minutes after a dyadic aggressive behavior (i.e., a conflict) took place. In post-conflict studies, it is common to collect matched control data (MC) 24 hours following the aggressive behavior (e.g., Palagi et al., 2008). This was not possible here, since the data we used were not originally collected for this purpose. Rather, we assigned 15-min matched control periods from collected focal observations using the following two criteria: (1) same focal animal and (2) within 3 hours of the time of day that the conflict happened in order to control for diel cycles. We picked the closest date that matched these conditions (median number of days between PC and MC — 2 days, interquartile range = 4 days, 20.2% (17 out of 84) of intervals were longer than 7 days). If an MC was not possible within 3 hours we eliminated the PC from the data (median interval between PC and MC time = 60.2 minutes, interquartile range = 60.3 minutes). We chose the exact start time of the MC such that the latency to conflict from the start of the focal in the PC was the same as the latency to MC start. Importantly, this procedure ensured that the PC and MC were additionally matched on the duration of observation. Twenty of the 84 PC/MC pairs were less than 15 minutes in duration because the focal observation time ended before the 15 minutes were reached (mean duration = 13.2 minutes, standard deviation = 3.9 minutes).
Statistical Analysis
We performed all statistical analyses for this study using R (R Core Team, 2023), and all code and data are available on FigShare (https://doi.org/10.6084/m9.figshare.23293043).
We analyzed dominance hierarchies using Elo ratings (Neumann et al., 2011). The Elo method is useful in cases where aggression and submission data are limited and when group membership changes over time (Neumann et al., 2011), both of which occurred in our study groups. Each individual in a group was assigned an Elo rating based on their observed aggressive and submissive interactions with other individuals. The Elo rating of an individual was then updated after each dominance interaction involving the individual. We measured the strictness of the dominance hierarchy by calculating the steepness of the dominance hierarchies using a Bayesian framework (Neumann & Fischer, 2023), which offers several advantages over the more commonly used David’s scores method, which defines steepness as the absolute slope of the straight line fitted to the normalized David’s scores (De Vries et al., 2006). The Bayesian procedure is similar to the original in that steepness is defined as the slope of a best-fit line; however, the line regresses the summed winning probability on the mean ordinal rank across iterations (Neumann & Fischer, 2023), which keeps the new value on the same scale as the original measure. As it is a Bayesian procedure, it generates distributions of plausible steepness values, rather than point estimates (Neumann & Fischer, 2023). This Bayesian procedure also eliminates the need to assign arbitrary start values and a k value (a scaling factor for the amount to which Elo ratings change after an interaction), but rather implements them as distributions that are influenced by the data itself (Neumann & Fischer, 2023). Importantly, the scores are robust to the density of interactions (Neumann & Fischer, 2023), meaning that large or small datasets are not more likely to have a particular steepness value. This allows the analysis of smaller datasets than is typical with David’s scores steepness. We used the ‘EloSteepness’ package in R to calculate steepness values (Neumann, 2022).
Many analyses of reconciliation have used latency to affiliation as a metric (e.g., de Waal & Yoshihara, 1983; Kutsukake & Castles, 2001; Majolo & Koyama, 2006). However, due to the sparsity of our data, we opted to analyze the occurrence of any affiliation as a binary variable. We modeled the occurrence of affiliation in an observation period with a logit link function as a multi-membership model implemented in brms (Bürkner, 2017, 2018). This type of model allowed us to account for repeated observations on dyads while recognizing that individuals are also a part of multiple dyads. Our predictor of interest was the interaction between the type of observation period (PC vs MC) and species. We also included an offset of the natural logarithm of observation time in hours. We included random intercepts for study group and dyad ID as well as the conflict ID, to account for the matching between PC and MC. Lastly, to control for the fact that some animals engaged in repeated conflicts across the dataset, we also included a random intercept for the dyad members. This was implemented using a multi-membership approach rather than two separate random intercepts for the aggressor and victim, because the multi-membership approach accounts for the fact that animal A is the same animal A whether they were the aggressor or victim of a particular dyadic interaction. Two separate random intercepts would not treat animal A as the same individual when they were in different roles in the interaction. We used default priors for the random effects, and the analyses presented were robust to making these priors less informative.
Additionally, we wanted to understand the demographic factors which predicted reconciliation bouts. We coded the post-conflict interval of the PC/MC pair as reconciled if affiliation occurred in the post-conflict period but not in the matched-control period. We used a multi-membership model with a binary outcome and logit link function to analyze the odds of reconciliation. Our fixed-effect predictors included the sex combination of the dyad (male–male, male–female, or female–female) and the absolute difference in rank between the individuals, as measured from their mean rank order from the Bayesian Elo analysis. We also included a random intercept for group, dyad, and a multi-membership term for the aggressor and victim, as in the previous analysis. We did not include an offset in this model, as by design, the members of the PC/MC pair were of the same duration. We would not expect affiliation to be earlier or later in the post-conflict interval compared to control depending on the observation length, meaning an offset was unnecessary for this model.
For all analyses, we also ran the analyses excluding data prior to 2018, due to the change of ethogram in 2018. We compared nested models with and without a fixed effect of interest using Bayes factors to evaluate the weight of evidence for including the fixed effect. We use Kass & Raftery’s (1995) scale for the interpretation of the Bayes factors.
Ethical Statement
Research adhered to the legal requirements of Madagascar and was approved by Madagascar’s CAFF/CORE committee. Research was conducted as educational training and under permit numbers N°127/16/MEEF/SG/DGF/DAPT/SCBT.Re, N°68/18/MEEF/SG/DGF/DSAP/SCB, N°105/19/MEDD/SG/DGF/DSAP/SCB.Re, and N°58/19/MEDD/SG/DGEF/DGRNE. The authors declare that they have no conflict of interest.
Data Availability
The datasets analyzed during the current study and code used for analysis are available on FigShare, (https://doi.org/10.6084/m9.figshare.23293043).
Results
Dominance Hierarchies
Across the entire dataset, we observed 559 dominance interactions, 494 in Propithecus diadema (105, 172, and 217 interactions for groups 1, 2, and 3 respectively) and 65 in Eulemur fulvus (19 and 46 interactions for groups 2 and 3 respectively). Aggression was initiated by a female in 76% of the interactions (212 out of 278 aggressive interactions), by a male 23% of the interactions (65 out of 278), and by an infant of undetermined sex 0.4% of the interactions (one out of 278). Contrastingly, submission was initiated by a female in 32% of interactions (91 out of 281 submissive interactions), a male in 66% of interactions (185 out of 281), and an infant of undetermined sex in 2% of interactions (five out of 281).
For Propithecus diadema, the top-ranked animal was always the dominant female (Fig. S1 available in the electronic supplementary material). In P. diadema groups, all adult females outranked males, except for group 1 from 2015–2016, where the second adult female ranked third behind the top-ranked female and top-ranked male (who was a subadult). The identities of the lowest ranked animals varied, and included a subadult female nearing dispersal age (group 1 2015–2016), juveniles (group 1 2018), a dispersing subadult female (group 2), and an adult male (group 3). Hierarchies for Eulemur fulvus were less clear (Fig. S1 available in the electronic supplementary material). In one group (group 2), the most dominant animal was an adult male, followed closely by an adult female. Younger males fell at the low end of this hierarchy. Contrastingly, in the other group (group 3), there was a clear dominant female, a clear lowest ranked male, and the other individuals were relatively close to each other in the middle.
Dominance hierarchy steepness was generally higher for Propithecus diadema than for Eulemur fulvus (Fig. 2). The median steepness value for P. diadema over the years was between 0.70 and 0.83. For E. fulvus the median steepness value was between 0.60 and 0.66.
Distribution of dominance hierarchy steepness values for each Propithecus diadema and Eulemur fulvus group in the Maromizaha Protected Area, Madagascar in various data collection periods between 2015 and 2020. Data for Propithecus diadema group 1 are split between 2015–2016 and 2018 due to our expansion of the ethogram in 2018. Dashed vertical lines represent the most frequent steepness value for the study group.
Conflict Behavior
We recorded a total of 84 paired PC/MC observations across species. Of the 84 conflicts, 68 were observed in the Propithecus diadema groups (approximately one conflict every 20.9 hours), and 16 were observed in the Eulemur fulvus groups (approximately one conflict every 39.4 hours). The conflicts for P. diadema were most common in group 1 (31 conflicts) and more evenly spread across the remaining groups (18 and 19 conflicts in groups 2 and 3 respectively), but the E. fulvus conflicts were highly skewed towards a single group (2 and 14 pairs respectively in groups 2 and 3). Most dyads did not engage in conflicts. Within our sample, 35% of P. diadema dyads (43 out of 123 possible dyads) and 20% of E. fulvus dyads (11 out of 56 possible dyads) engaged in a conflict. Between dyads that had a conflict, there was a median of one conflict per dyad (Interquartile range = 1, range = 1–4).
Post-Conflict Affiliation
We found very strong evidence that affiliation patterns differed by species (Bayes factor in favor of model accounting for species = 673.79; see full model results in Table S3). For Propithecus diadema, 95% of the generated posterior distribution for the log odds ratio (\(\text{log}(\frac{Odds\; of\; affliation\; PC}{Odds\; of\; affliation\; MC})\), where log odds ratio of 0 indicates equal probabilities of affiliation in post conflict and matched control) fell between 0.76 and 4.49 (median estimate for log odds ratio = 2.35; Fig. 3), providing support for the prediction that the odds of affiliation were higher in post-conflict periods compared to control periods for P. diadema. However, we did not find a difference in the log odds of affiliation for Eulemur fulvus between post-conflict periods compared to control periods (median estimate for log odds ratio = −0.85; 95% credible interval: [−2.65, 0.92]; Fig. 3). These results did not change when we excluded data gathered before 2018 due to the change in data recording (full results in Table S4).
Posterior distribution of the log odds ratio (\(\text{log}(\frac{Odds\;of\;affliation\;PC}{Odds\;of\;affliation\;MC})\)) for Propithecus diadema and Eulemur fulvus, Maromizaha Protected Area, Madagascar (2015–2020). The black, vertical dotted lines represent the median log odds ratios for the two species. The red, vertical dashed line at x = 0 represents the value if the odds of affiliation were the same in PC and MC. Posterior distributions that overlap less with 0 offer more support for the prediction that the odds of affiliation differ in PC and MC. Based on 95% credible intervals, the odds of affiliation for P. diadema were higher in post-conflict periods compared to matched control; however, we detected no difference between the conditions for E. fulvus.
Which Propithecus diadema Dyads Reconcile?
We only analyzed demographic trends in reconciliation in Propithecus diadema, since Eulemur fulvus did not show reconciliation behavior. Out of a total of 43 P. diadema dyads who interacted, ten dyads showed reconciliation behavior. We found very strong support that sex combinations varied in how frequently they reconciled (Bayes factor comparing models with and without sex combination = 5773.07; see full model results in Table S5). Specifically, male–male dyads were more likely to reconcile than other sex combinations (Fig. 4left; 95% CI for log odds ratio of reconciliation given no difference in rank for: male–male = [0.06, 10.25]; male–female = [−12.91, −1.27]; female–female = [−9.84, 2.99]). Additionally, we found very strong evidence that the odds of reconciliation varied by absolute rank difference (Bayes factor comparing models with and without rank difference = 5903.13). Specifically, dyads where the two individuals were further apart in ranking had higher log odds of reconciliation (Fig. 4right; 95% CI for log odds ratio of reconciliation for rank difference for a male–female dyad = [−0.11, 2.38]). These results were generally consistent when we analyzed data only from 2018 and beyond (Table S6), except that male-male dyads reconciled less frequently in the smaller dataset.
Posterior distribution of the log odds ratio for reconciliation in Propithecus diadema, Maromizaha Protected Area, Madagascar (2015–2020) broken down by sex classes (left) and rank difference (right). The black, vertical dotted lines represent the median log odds ratios for each distribution. The red, vertical dashed line at x = 0 represents the value of no effect of the demographic variable on the log odds ratio of reconciliation. Based on 95% credible intervals, the odds of reconciliation were higher for male–male dyads than other sex classes and for dyads where the difference in rank was greater.
Discussion
Propithecus diadema had more frequent paired post-conflict/matched-control intervals than Eulemur fulvus, happening approximately twice as frequently. The dominant female in P. diadema groups consistently held the highest rank, with adult females outranking adult males. However, we observed one exception in a case where the second adult female ranked third behind the top-ranked female and male. In E. fulvus, dominance hierarchies were less clear, with one group showing a dominant adult male followed closely by an adult female, while in another group, only a dominant female and a lowest-ranked male were evident. We observed reconciliation behavior only in P. diadema, in which the odds of affiliation were higher in post-conflict periods compared to matched control. We detected no difference between the conditions for E. fulvus. While 40% of P. diadema dyads who interacted reconciled (23 out of 58 dyads who interacted), various demographic classes did not reconcile at the same rates. Male–male dyads reconciled more than male–female or female–female dyads. Dyads of more disparate ranks also reconciled more.
Differences in Dominance and Post-Conflict Behavior Across Species
Our finding of steep and well-defined hierarchies with alpha females in Propithecus diadema and less defined hierarchies not based on sex in Eulemur fulvus is consistent with other findings in these species and their congeners. For example, P. diadema in Tsinjoarivo generally show female dominance hierarchies as measured by female leadership of group movements and displacements of males during feeding events (Rasolonjatovo & Irwin, 2020). Although data from additional P. diadema populations is lacking, both rainforest-living and dry forest-living Propithecus show similar patterns (Pochron et al., 2003; Ramanamisata et al., 2014; Richard & Nicoll, 1987). Data on dominance patterns are more varied among Eulemur species, both in captivity and the wild (Kappeler et al., 2022). How dominance is expressed (Erhart & Overdorff, 1999; Jacobs et al., 2008) and which sex is dominant (Digby & Kahlenberg, 2002; Marolf et al., 2007) can both vary among Eulemur species. This species-level variability in dominance patterns can trickle down within populations to group-level differences (Marolf et al., 2007; Ostner & Kappeler, 1999; Pereira & McGlynn, 1997; Roeder & Fornasieri, 1995), which aligns with our finding of group-specific hierarchies.
Our post-conflict reconciliation results across species did not support our original prediction generated by the valuable relationships hypothesis and differences between strepsirrhines and haplorrhines. While we predicted that steeper dominance hierarchies would be associated with decreased reconciliation of conflicts, we observed the exact opposite. Propithecus diadema (whose groups showed steeper dominance hierarchies) showed evidence of reconciliation while Eulemur fulvus (whose groups showed less steep (i.e., more shallow) dominance hierarches) did not show evidence of reconciliation. These results diverged from previous work in catarrhine monkeys, particularly Macaca, that suggests that despotic hierarchies are associated with lower reconciliation tendencies (Berman et al., 2004; Cooper & Bernstein, 2002; de Waal & Luttrell, 1985; de Waal & Ren, 1988; Demaria & Thierry, 2001; Thierry, 2000). Without further data on additional strepsirrhine species and populations, it is difficult to pinpoint a specific cause leading to this contrast between the haplorrhine literature and our results on two strepsirrhine species. While it could reflect a true difference in the social behavior of the two primate suborders, it is also possible that there are more nuanced relationships between reconciliation behavior, despotism, and hierarchy steepness that have yet to be explored.
In our case, it may be that dominance hierarchies control behavior less in Eulemur fulvus, particularly due to the low rate of aggression. Thus, there would be less selective pressure on the evolution of reconciliation behavior. In species with a higher rate of aggression, reconciliation behavior may evolve as a mechanism to reduce the cost of aggression. Without frequent aggression, there may not be enough of a selective pressure on individuals to evolve reconciliation. Further investigation into the rate of aggression, rather than the steepness of the hierarchy alone, is needed to fully understand the lack of evidence of reconciliation observed in the study.
Our results for the two species were partially consistent with the existing literature on reconciliation in Propithecus and Eulemur. While reconciliation behavior has not been previously reported in Propithecus diadema to our knowledge, it aligns with results in other congener species (Propithecus verreauxi: Lewis, 2019; Palagi et al., 2008). For example, P. verreauxi living in southeastern Madagascar reconciled their conflicts, particularly those of low intensity or in non-feeding contexts (Palagi et al., 2008). Similarly, P. verreauxi living in western Madagascar engaged in affiliative interactions within 15 minutes of a conflict (Lewis, 2019). Our results, however, diverged from past research on E. fulvus, where affiliative contacts occurred sooner in PC periods than MC periods (Roeder et al., 2002). Lemurs in these previous studies lived in captivity, which has been proposed as a source of increased reconciliation due to the space constraints and inability for individuals to move away from others (Judge, 2000). However, reviews of the literature in this area have suggested that this is not a consistent driving factor in the presence or absence of reconciliation behavior in a particular group of primates (Aureli et al., 2002; Colmenares, 2006). Thus, it seems unlikely that the difference in captivity versus wild is the sole explaining factor in the difference of results. Other research on Eulemur has shown mixed results across species, including reports of reconciliation and reports of no reconciliation (Kappeler, 1993a; Palagi & Norscia, 2011; Roeder et al., 2002), so further investigation into their social dynamics within and across species is warranted.
Differences Across Dyads in Reconciliation Rates in Propithecus d iadema
Within our observations, less than one-fourth of the Propithecus diadema dyads who engaged in dominance interactions reconciled, with uneven distribution across demographic variables. In P. diadema we found that male–male dyads reconciled more than any dyads involving females. In related species, such as P. verreauxi, rates of male–male grooming are higher than male–female or female–female grooming, and males do not discourage multimale groups from forming (Lewis, 2008). While the benefits of living in a multimale group for males are not well understood, if they exist at all (Ostner & Schulke, 2014; Port et al., 2012), further research should be conducted to identify possible aspects of value in the relationship (for example, coalitionary support: Leimberger & Lewis, 2017; Arnold et al., 2011), as would be suggested by the valuable relationship hypothesis.
Interestingly, in terms of Bayes factors, the effect of rank difference was approximately equal to the effect of sex in our dataset, supporting the inclusion of the parameter. While this corroborates much of the literature on rank difference and reconciliation patterns where individuals of disparate ranks are more likely to reconcile (Judge, 1991; Silk et al., 1996; Palagi et al., 2008), the parity in the size of effects for rank and sex in Propithecus diadema may reflect the female dominant social structure that manifests as sex-biased aggression (Rasolojavato & Irwin, 2020). An interesting consideration for future work is the effect of rank difference and individual rank on reconciliation behavior. For example, the relationship value may not be the same between a low- and mid-ranking animal versus a mid- and high-ranking animal, despite having similar rank differences due to the resource holding potential of top-ranked animals. The valuable-relationships hypothesis would then predict differences in the probability of reconciliation based on dominance rank alone regardless of rank difference. In this case, investigating these concepts in a species that lives in larger groups with frequent conflicts may be necessary to generate the data richness necessary to differentiate between ordinal rank and rank difference.
Caveats and Ideas for Further Study
While interpreting these results, it is important to consider that there is currently no consensus on what duration should be used for PC/MC observations. A common duration is between 5 and 15 minutes (for examples see: Arnold, 1997; Castles & Whiten, 1998; Kutsukake & Castles, 2001; Leca et al., 2002; Matsumura, 1996; Norscia & Palagi, 2011; Palagi & Norscia, 2011), but some studies have used durations up to 70 minutes resulting in conflicting results (Rolland & Roeder, 2000). For this study, we decided to use 15-minute intervals to align with previous research on Propithecus and Eulemur (Norscia & Palagi, 2011; Palagi et al., 2008; Palagi & Norscia, 2011). Given that E. fulvus has long periods of inactivity during the day due to its cathemeral activity pattern (Razanaparany & Sato, 2020), it is possible that this species reconciles but just on a much longer time-scale that our analysis fails to capture.
Additionally, the data used in this study, while not originally collected with the specific aim of investigating lemur reconciliation, provided a valuable resource for addressing these research questions. One notable advantage of this dataset was the possible inclusion of matched controls that preceded the conflicts, which is typically not feasible in traditionally collected data where conflicts cannot be anticipated. Thus, the dataset presented opportunities to analyze control data without any influence of the conflict, even if it is long-lasting.
Several other sociodemographic factors beyond the scope of the study are important in structuring reconciliation patterns across dyads, and may have impacted the results reported here. For example, across primate species, reconciliation is generally more common in close genetic kin than non-kin (as reviewed in Silk, 2002; Arnold et al., 2011). At this time, the relatedness among group members in our population is unknown, so we were unable to directly test this hypothesis. Interestingly, among eastern sifakas (Propithecus diadema, P. edwardsi, P. candidus, P. perrieri, P. tattersalli), both sexes regularly disperse (Irwin, 2006; Irwin et al., 2019; King et al., 2009), meaning that both males and females regularly live in groups that vary in relatedness to them (including groups where a focal member has close genetic kin and groups where a focal member is relatively unrelated to the rest of the group). This forms an interesting system for evaluating how reconciliation functions dependent on the kin relations present in the group, and deserves further study combining genetic relatedness estimates with behavioral data. Additionally, social bonding is also known to influence reconciliation patterns across species (Castles et al., 1996; Cooper et al., 2005; Pereira et al., 2000; Romero et al., 2009; Schino et al., 1998), including in Propithecus (Palagi et al., 2008). This aspect supports the valuable relationships hypothesis (de Waal & Aureli, 1997), which suggests that reconciliation serves to repair relationships that were damaged by conflict. Although beyond the scope of this manuscript, previous work in the population has suggested that sifaka play partner choices that are correlated with grooming partner choice (Lutz et al., 2019). Sifaka tended to play with like-aged, like-ranked, and same sex animals, so these preferences in social bonding may also influence the distribution of reconciliation behavior within the group.
Temporal differences in reconciliation may be expected, especially considering lemurs’ strict seasonal breeding (Wright, 1999). These temporal variations could be due to changes in resource availability (Verbeek & de Waal, 1997) and/or reproductive seasonality (Majolo & Koyama, 2006; Palagi et al., 2005; Palagi & Norscia, 2015; Schino et al., 1998; Silk, 2002; but see Aureli et al., 1993). While changes in resource availability are known to alter many aspects of primates’ behavior, this may not drive seasonal patterns of reconciliation patterns, as feeding-related conflicts are not reconciled as frequently (Aureli et al., 2002; Castles & Whitten, 1998; Schaffner et al., 2005; Westlund et al., 2000). However, in terms of the seasonality in mating, our data are aggregated across multiple seasons and did not include data from the gestation period. This means that it may not fully capture the nuances of time-sensitive variables that influence reconciliation behavior such as seasonality in mating. Interestingly, Lemur catta reconcile the least during the mating season, a season that is represented in our dataset (Palagi & Norscia, 2015). Therefore, further investigation is warranted to examine the potential seasonal distribution and its impact on the observed patterns.
We chose to focus on dominance hierarchies as a proxy of the differences between species, given the pervasiveness of female dominance in the lineage and the relationships between dominance, power, conflict, and affiliation. Dominance hierarchies can influence who in the group has access to food resources and mating opportunities, with dominant individuals having priority access. In lemurs, for example, dominant females have been found to have greater access to food resources (Overdorff et al., 2005; Rasamimanana, 1999; Rasolonjatovo & Irwin, 2020; White et al., 2007), which may be an avenue that leads to higher reproductive success for these dominant animals (Gouzoules et al., 1982; Pusey et al., 1997). Additionally, dominance can influence patterns of aggression and conflict, with dominant individuals being more likely to win fights and maintain social order (Kappeler et al., 2022; Seex et al., 2022). Lemur social affiliation patterns are also governed by differences in dominance rank in many species (Erhart & Overdorff, 2008; Jolly, 1998; Lewis, 2010; Lutz et al., 2019; Port et al., 2009; Waeber & Hemelrijk, 2003). However, the studied species differ in other aspects, even though they were studied sympatrically. Specifically, factors such as group size, diel period, mating competition, diet, and territoriality can differ between these genera. For example, mating competition and reproductive strategies can shape dominance interactions (Gachot-Neveu et al., 1999; Sauther, 1993). Furthermore, dietary variations stemming from differences in resource distribution can alter social dynamics (Gemmill & Gould, 2008; Janson, 1985). Without further data on additional populations of these species and/or more longitudinal data on this population, it is difficult to tease apart these effects from differences in dominance hierarchies.
Conclusion
We had evidence of reconciliation in Propithecus diadema, but not in Eulemur fulvus. This finding did not support our hypothesis that strict dominance hierarchies would be associated with lower rates of reconciliation, and may suggest that frequent aggressive bouts are needed to support the evolution of reconciliation. We found that within P. diadema, male–male dyads were more likely to reconcile, and that dyads further apart in rank tended to reconcile more. Further studies on reconciliation in P. diadema are needed to clarify whether our results establish a species-specific pattern or whether they are site- or group-specific. Additional studies on other strepsirrhine species, closely related or not, could lead to further understanding of the evolutionary forces driving reconciliation behavior and how the behavior may or may not function differently in strepsirrhine and haplorrhine societies.
References
Altmann, J. (1974). Observational study of behavior: Sampling methods. Behaviour, 49(3–4), 227–266. https://doi.org/10.1163/156853974X00534
Amici, F., Widdig, A., MacIntosh, A. J., Francés, V. B., Castellano-Navarro, A., Caicoya, A. L., ..., & Majolo, B. (2020). Dominance style only partially predicts differences in neophobia and social tolerance over food in four macaque species. Scientific Reports, 10(1), 22069. https://doi.org/10.1038/s41598-020-79246-6
Arnold, K. (1997). Post-conflict behaviour of spectacled langurs (Trachypithecus obscurus). Durham theses, Durham University. Available at Durham E-Theses Online: http://etheses.dur.ac.uk/5461/
Arnold, C., Matthews, L. J., & Nunn, C. L. (2010). The 10kTrees website: A new online resource for primate phylogeny. Evolutionary Anthropology, 19(3), 114–118. https://doi.org/10.1002/evan.20251
Arnold, K., Fraser, O. N., & Aureli, F. (2011). Postconflict reconciliation. In C. Campbell, A. Fuentes, K. C. MacKinnon, S. K. Bearder, & R. M. Stumpt (Eds.), Primates in Perspective (pp. 608–625). Oxford University Press.
Aureli, F., & van Schaik, C. P. (1991). Post-conflict behaviour in long-tailed macaques (Macaca fascicularis): I The social events. Ethology, 89(2), 89–100. https://doi.org/10.1111/j.1439-0310.1991.tb00296.x
Aureli, F., Veenema, H. C., Panthaleon, Van, van Eck, C. J., & van Hooff, J. A. R. A. M. (1993). Reconciliation, consolation, and redirection in Japanese macaques (Macaca fuscata). Behaviour, 124(1–2), 1–21. https://doi.org/10.1163/156853993X00470
Aureli, F., Das, M., & Veenema, H. C. (1997). Differential kinship effect on reconciliation in three species of macaques (Macaca fascicularis, M. fuscata, and M. sylvanus). Journal of Comparative Psychology, 111(1), 91–99. https://doi.org/10.1037/0735-7036.111.1.91
Aureli, F., Cords, M., & van Schaik, C. P. (2002). Conflict resolution following aggression in gregarious animals: A predictive framework. Animal Behaviour, 64, 325–341. https://doi.org/10.1006/anbe.2002.3071
Balasubramaniam, K. N., Dittmar, K., Berman, C. M., Butovskaya, M., Cooper, M. A., Majolo, B., ... & De Waal, F. B. (2012). Hierarchical steepness, counter‐aggression, and macaque social style scale. American Journal of Primatology, 74(10), 915–925. https://doi.org/10.1002/ajp.22044
Barton, R. A. (2006). Olfactory evolution and behavioral ecology in primates. American Journal of Primatology, 68(6), 545–558. https://doi.org/10.1002/ajp.20251
Berman, C. M., Ionica, C. S., & Li, J. (2004). Dominance style among Macaca thibetana on Mt. Huangshan. China. International Journal of Primatology, 25, 1283–1312. https://doi.org/10.1023/B:IJOP.0000043963.77801.c3
Bürkner, P.-C. (2017). brms: An R package for Bayesian multilevel models using Stan. Journal of Statistical Software, 80(1), 1–28. https://doi.org/10.18637/jss.v080.i01
Bürkner, P.-C. (2018). Advanced Bayesian multilevel modeling with the R package brms. The R Journal, 10(1), 395–411. https://doi.org/10.32614/RJ-2018-017
Campos, F. A., Villavicencio, F., Archie, E. A., Colchero, F., & Alberts, S. C. (2020). Social bonds, social status and survival in wild baboons: A tale of two sexes. Philosophical Transactions of the Royal Society B, 375(1811), 20190621. https://doi.org/10.1098/rstb.2019.0621
Castles, D. L., Aureli, F., & de Waal, F. B. (1996). Variation in conciliatory tendency and relationship quality across groups of pigtail macaques. Animal Behaviour, 52(2), 389–403. https://doi.org/10.1006/anbe.1996.0183
Castles, D. L., & Whiten, A. (1998). Post-conflict behaviour of wild olive baboons. I. Reconciliation, redirection and consolation. Ethology, 104(2), 126–147. https://doi.org/10.1111/j.1439-0310.1998.tb00057.x
Colmenares, F. (2006). Is post-conflict affiliation in captive nonhuman primates an artifact of captivity? International Journal of Primatology, 27, 1311–1336. https://doi.org/10.1007/s10764-006-9080-x
Cooper, M. A., & Bernstein, I. S. (2002). Counter aggression and reconciliation in Assamese macaques (Macaca assamensis). American Journal of Primatology, 56(4), 215–230. https://doi.org/10.1002/ajp.1076
Cooper, M. A., Bernstein, I. S., & Hemelrijk, C. K. (2005). Reconciliation and relationship quality in Assamese macaques (Macaca assamensis). American Journal of Primatology, 65(3), 269–282. https://doi.org/10.1002/ajp.20114
Cordoni, G., Palagi, E., & Tarli, S. B. (2006). Reconciliation and consolation in captive western gorillas. International Journal of Primatology, 27, 1365–1382.
Cords, M. (1992). Post-conflict reunions and reconciliation in long-tailed macaques. Animal Behaviour, 44(1), 57–61. https://doi.org/10.1016/S0003-3472(05)80754-7
Das, M., Penke, Z., & van Hooff, J. A. R. A. M. (1998). Postconflict affiliation and stress-related behavior of long-tailed macaque aggressors. International Journal of Primatology, 19(1), 53–71. https://doi.org/10.1023/A:1020354826422
De Vries, H., Stevens, J. M. G., & Vervaecke, H. (2006). Measuring and testing the steepness of dominance hierarchies. Animal Behaviour, 71(3), 585–592. https://doi.org/10.1016/J.ANBEHAV.2005.05.015
de Waal, F. B. M. (1986). The integration of dominance and social bonding in primates. Quarterly Review of Biology, 61(4), 459–479. https://doi.org/10.1086/415144
de Waal, F. B. M., & Aureli, F. (1997). Conflict resolution and distress alleviation in monkeys and apes. In C. S. Carter, B. Kirkpatrick, & I. Lenderhendler (Eds.), The Integrative Neurobiology of Affiliation (pp. 317–328). Annals of the New York Academy of Sciences.
de Waal, F. B. M., & Luttrell, L. M. (1985). The formal hierarchy of rhesus macaques: An investigation of the bared-teeth display. American Journal of Primatology, 9(2), 73–85. https://doi.org/10.1002/ajp.1350090202
de Waal, F. B. M., & Yoshihara, D. (1983). Reconciliation and redirected affection in rhesus monkeys. Behaviour, 85(3–4), 224–241. https://doi.org/10.1163/156853983X00237
de Waal, F. B., & Ren, R. (1988). Comparison of the reconciliation behavior of stumptail and rhesus macaques. Ethology, 78(2), 129–142. https://doi.org/10.1111/j.1439-0310.1988.tb00224.x
Demaria, C., & Thierry, B. (2001). A comparative study of reconciliation in rhesus and Tonkean macaques. Behaviour, 138(3), 397–410. https://doi.org/10.1163/15685390152032514
Digby, L. J., & Kahlenberg, S. M. (2002). Female dominance in blue-eyed black lemurs (Eulemur macaco flavifrons). Primates, 43(3), 191–199. https://doi.org/10.1007/BF02629647
Drews, C. (1993). The concept and definition of dominance in animal behaviour. Behaviour, 125(3–4), 283–313. https://doi.org/10.1163/156853993X00290
Ellis, S., Snyder-Mackler, N., Ruiz-Lambides, A., Platt, M. L., & Brent, L. J. (2019). Deconstructing sociality: The types of social connections that predict longevity in a group-living primate. Proceedings of the Royal Society B, 286(1917), 20191991.
Erhart, E. M., & Overdorff, D. J. (1999). Female coordination of group travel in wild Propithecus and Eulemur. International Journal of Primatology, 20, 927–940. https://doi.org/10.1023/A:1020830703012
Erhart, E. M., & Overdorff, D. J. (2008). Rates of agonism by diurnal lemuroids: Implications for female social relationships. International Journal of Primatology, 29, 1227–1247. https://doi.org/10.1007/s10764-008-9287-0
Feldblum, J. T., Krupenye, C., Bray, J., Pusey, A. E., & Gilby, I. C. (2021). Social bonds provide multiple pathways to reproductive success in wild male chimpanzees. Iscience, 24(8), 102864. https://doi.org/10.1016/j.isci.2021.102864
Fichtel, C., & Kappeler, P. M. (2022). Coevolution of social and communicative complexity in lemurs. Philosophical Transactions of the Royal Society B, 377(1860), 20210297. https://doi.org/10.1098/rstb.2021.0297
Gachot-Neveu, H., Petit, M., & Roeder, J. J. (1999). Paternity determination in two groups of Eulemur fulvus mayottensis: Implications for understanding mating strategies. International Journal of Primatology, 20(1), 107–119. https://doi.org/10.1023/A:1020588301716
Ganzhorn, J. U. (1988). Food partitioning among Malagasy primates. Oecologia, 75(3), 436–450. https://doi.org/10.1007/BF00376949
Gemmill, A., & Gould, L. (2008). Microhabitat variation and its effects on dietary composition and intragroup feeding interactions between adult female Lemur catta during the dry season at Beza Mahafaly Special Reserve, southwestern Madagascar. International Journal of Primatology, 29, 1511–1533. https://doi.org/10.1007/s10764-008-9316-z
Gouzoules, H., Gouzoules, S., & Fedigan, L. (1982). Behavioural dominance and reproductive success in female Japanese monkeys (Macaca fuscata). Animal Behaviour, 30(4), 1138–1150. https://doi.org/10.1016/S0003-3472(82)80204-2
Irwin, M. T. (2006). Ecologically enigmatic lemurs: The sifakas of the eastern forests (Propithecus candidus, P. diadema, P. edwardsi, P. perrieri, and P. tattersalli). In L. Gould & M. Sauther (Eds.), lemurs (pp. 305–326). Springer. https://doi.org/10.1007/978-0-387-34586-4
Irwin, M. T., Samonds, K. E., Raharison, J. L., Junge, R. E., Mahefarisoa, K. L., Rasambainarivo, F., ... & Glander, K. E. (2019). Morphometric signals of population decline in diademed sifakas occupying degraded rainforest habitat in Madagascar. Scientific Reports, 9, 14. https://doi.org/10.1038/s41598-019-45426-2
Jacobs, A., Maumy, M., & Petit, O. (2008). The influence of social organisation on leadership in brown lemurs (Eulemur fulvus fulvus) in a controlled environment. Behavioural Processes, 79(2), 111–113. https://doi.org/10.1016/j.beproc.2008.05.004
Janson, C. (1985). Aggresive competition and individual food consumption in wild brown capuchin monkeys (Cebus apella). Behavioral Ecology and Sociobiology, 18(2), 125–138. https://doi.org/10.1007/BF00299041
Jolly, A. (1966). Lemur social behavior and primate intelligence: The step from prosimian to monkey intelligence probably took place in a social context. Science, 153(3735), 501–506. https://doi.org/10.1126/science.153.3735.501
Jolly, A. (1998). Pair-bonding, female aggression and the evolution of lemur societies. Folia Primatologica, 69(7), 1–13. https://doi.org/10.1159/000052693
Judge, P. G. (1991). Dyadic and triadic reconciliation in pigtail macaques (Macaca nemestrina). American Journal of Primatology, 23(4), 225–237. https://doi.org/10.1002/ajp.1350230403
Judge, P. G. (2000). Coping strategies under crowded conditions. In F. Aureli & F. B. M. de Waal (Eds.), Natural Conflict Resolution (pp. 129–154). University of California Press. https://doi.org/10.1525/9780520924932-011
Kappeler, P. M. (1993a). Reconciliation and post-conflict behavior in ringtailed lemurs, Lemur catta and redfronted lemurs, Eulemur fulvus rufus. Animal Behaviour, 45, 901–915. https://doi.org/10.1006/anbe.1993.1110
Kappeler, P. M. (1993b). Variation in social structure: The effects of sex and kinship on social interactions in three lemur species. Ethology, 93(2), 125–145. https://doi.org/10.1111/j.1439-0310.1993.tb00984.x
Kappeler, P. M., & van Schaik, C. P. (1992). Methodological and evolutionary aspects of reconciliation among primates. Ethology, 92(1), 51–69. https://doi.org/10.1111/J.1439-0310.1992.TB00948.X
Kappeler, P. M., Fichtel, C., & Radespiel, U. (2022). The island of female power? Intersexual dominance relationships in the lemurs of Madagascar. Frontiers in Ecology and Evolution, 10, 390. https://doi.org/10.3389/FEVO.2022.858859/XML/NLM
Kass, R. E., & Raftery, A. E. (1995). Bayes factors. Journal of the American Statistical Association, 90(430), 773–795. https://doi.org/10.1080/01621459.1995.10476572
Keverne, E. B. (1999). The vomeronasal organ. Science, 286(5440), 716–720. https://doi.org/10.1126/science.286.5440.716
King, S. J., Pochron, S. T., & Wright, P. C. (2009). The rules of disengagement: takeovers, infanticide, and dispersal in a rainforest lemur, Propithecus edwardsi. Behaviour, 146, 499–523. https://doi.org/10.1163/15683908X399554
Kulahci, I. G., Rubenstein, D. I., & Ghazanfar, A. A. (2015). Lemurs groom-at-a-distance through vocal networks. Animal Behaviour, 110, 179–186. https://doi.org/10.1016/j.anbehav.2015.09.016
Kulahci, I. G., Ghazanfar, A. A., & Rubenstein, D. I. (2018). Knowledgeable lemurs become more central in social networks. Current Biology, 28(8), 1306–1310. https://doi.org/10.1016/j.cub.2018.02.079
Kutsukake, N., & Castles, D. L. (2001). Reconciliation and variation in post-conflict stress in Japanese macaques (Macaca fuscata fuscata): Testing the integrated hypothesis. Animal Cognition, 4(3–4), 259–268. https://doi.org/10.1007/s10071-001-0119-2
Leca, J. B., Fornasieri, I., & Petit, O. (2002). Aggression and reconciliation in Cebus capucinus. International Journal of Primatology, 23(5), 979–998. https://doi.org/10.1023/A:1019641830918
Leimberger, K. G., & Lewis, R. J. (2017). Patterns of male dispersal in Verreaux’s sifaka (Propithecus verreauxi) at Kirindy Mitea National Park. American Journal of Primatology, 79(7), e22455. https://doi.org/10.1002/ajp.22455
Lewis, R. J. (2008). Social influences on group membership in Propithecus verreauxi verreauxi. International Journal of Primatology, 29, 1249–1270. https://doi.org/10.1007/s10764-008-9304-3
Lewis, R. J. (2010). Grooming patterns in Verreaux’s sifaka. American Journal of Primatology, 72(3), 254–261. https://doi.org/10.1002/ajp.20776
Lewis, R. J. (2019). Subordination signals improve the quality of social relationships in Verreaux’s sifaka: Implications for the evolution of power structures and social complexity. American Journal of Physical Anthropology, 169(4), 599–607. https://doi.org/10.1002/ajpa.23876
Lutz, M. C., Ratsimbazafy, J., & Judge, P. G. (2019). Use of social network models to understand play partner choice strategies in three primate species. Primates, 60(3), 247–260. https://doi.org/10.1007/S10329-018-00708-7/TABLES/5
Majolo, B., & Koyama, N. (2006). Seasonal effects on reconciliation in Macaca fuscata yakui. International Journal of Primatology, 27, 1383–1397. https://doi.org/10.1007/s10764-006-9079-3
Majolo, B., Ventura, R., & Koyama, N. F. (2009). A statistical modelling approach to the occurrence and timing of reconciliation in wild Japanese macaques. Ethology, 115(2), 152–166. https://doi.org/10.1111/j.1439-0310.2008.01591.x
Marolf, B., McElligott, A. G., & Müller, A. E. (2007). Female social dominance in two Eulemur species with different social organizations. Zoo Biology, 26(3), 201–214. https://doi.org/10.1002/zoo.20135
Matsumura, S. (1996). Postconflict affiliative contacts between former opponents among wild moor macaques (Macaca maurus). American Journal of Primatology, 38(3), 211–219. https://doi.org/10.1002/(SICI)1098-2345(1996)38:3%3c211::AID-AJP2%3e3.0.CO;2-1
McFarland, R., & Majolo, B. (2013). The importance of considering the behavioral form of reconciliation in studies of conflict resolution. International Journal of Primatology, 34, 15–29. https://doi.org/10.1007/s10764-012-9643-y
McFarland, R., Murphy, D., Lusseau, D., Henzi, S. P., Parker, J. L., Pollet, T. V., & Barrett, L. (2017). The ‘strength of weak ties’ among female baboons: Fitness-related benefits of social bonds. Animal Behaviour, 126, 101–106. https://doi.org/10.1007/s10764-012-9643-y
Neumann, C. (2022). EloSteepness: Bayesian dominance hierarchy steepness via Elo rating and David's scores. R package version 0.4.7. https://CRAN.R-project.org/package=EloSteepness. Accessed 21 June 2024.
Neumann, C., & Fischer, J. (2023). Extending Bayesian Elo-rating to quantify the steepness of dominance hierarchies. Methods in Ecology and Evolution, 14(2), 669–682. https://doi.org/10.1111/2041-210X.14021
Neumann, C., Duboscq, J., Dubuc, C., Ginting, A., Irwan, A. M., Agil, M., Widdig, A., & Engelhardt, A. (2011). Assessing dominance hierarchies: Validation and advantages of progressive evaluation with Elo-rating. Animal Behaviour, 82(4), 911–921. https://doi.org/10.1016/J.ANBEHAV.2011.07.016
Norscia, I., & Palagi, E. (2011). Do wild brown lemurs reconcile? Not always. Journal of Ethology, 29(1), 181–185. https://doi.org/10.1007/s10164-010-0228-y
Ostner, J., & Kappeler, P. M. (1999). Central males instead of multiple pairs in redfronted lemurs, Eulemur fulvus rufus (Primates, Lemuridae)? Animal Behaviour, 58(5), 1069–1078. https://doi.org/10.1006/anbe.1999.1222
Ostner, J., & Schülke, O. (2014). The evolution of social bonds in primate males. Behaviour, 151(7), 871–906. https://doi.org/10.1163/1568539X-00003191
Overdorff, D. J., Erhart, E. M., & Mutschler, T. (2005). Does female dominance facilitate feeding priority in black-and-white ruffed lemurs (Varecia variegata) in southeastern Madagascar? American Journal of Primatology, 66(1), 7–22. https://doi.org/10.1002/AJP.20125
Palagi, E., Antonacci, D., & Norscia, I. (2008). Peacemaking on treetops: First evidence of reconciliation from a wild prosimian (Propithecus verreauxi). Animal Behaviour, 76(3), 737–747. https://doi.org/10.1016/j.anbehav.2008.04.016
Palagi, E., & Norscia, I. (2011). Scratching around stress: Hierarchy and reconciliation make the difference in wild brown lemurs (Eulemur fulvus). Stress, 14(1), 93–97. https://doi.org/10.3109/10253890.2010.505272
Palagi, E., & Norscia, I. (2015). The season for peace: Reconciliation in a despotic species (Lemur catta). PLoS One, 10(11), e0142150. https://doi.org/10.1371/journal.pone.0142150
Palagi, E., Paoli, T., & Tarli, S. B. (2005). Aggression and reconciliation in two captive groups of Lemur catta. International Journal of Primatology, 26(2), 279–294. https://doi.org/10.1007/s10764-005-2925-x
Penate, L., Pelaéz, F., & Sanchez, S. (2009). Reconciliation in captive cotton-top tamarins (Saguinus oedipus), a cooperative breeding primate. American Journal of Primatology, 71(11), 895–900. https://doi.org/10.1002/AJP.20721
Pereira, M. E., & McGlynn, C. A. (1997). Special relationships instead of female dominance for redfronted lemurs, Eulemur fulvus rufus. American Journal of Primatology, 43(3), 239–258. https://doi.org/10.1002/(SICI)1098-2345(1997)43:3%3c239::AID-AJP4%3e3.0.CO;2-Y
Pereira, M. E., Schill, J. L., & Charles, E. P. (2000). Reconciliation in captive Guyanese squirrel monkeys (Saimiri sciureus). American Journal of Primatology, 50(2), 159–167. https://doi.org/10.1002/(SICI)1098-2345(200002)50:2%3c159::AID-AJP6%3e3.0.CO;2-F
Pereira, M. E., Kaufman, R., Kappeler, P. M., & Overdoff, D. J. (1990). Female dominance does not characterize all of the lemuridae. Folia Primatologica, 55(2), 96–103. https://doi.org/10.1159/000156505
Pochron, S. T., Fitzgerald, J., Gilbert, C. C., Lawrence, D., Grgas, M., Rakotonirina, G., Ratsimbazafy, R., Rakotosoa, R., & Wright, P. C. (2003). Patterns of female dominance in Propithecus diadema edwardsi of Ranomafana National Park, Madagascar. American Journal of Primatology, 61(4), 173–185. https://doi.org/10.1002/AJP.10119
Port, M., Clough, D., & Kappeler, P. M. (2009). Market effects offset the reciprocation of grooming in free-ranging redfronted lemurs Eulemur fulvus rufus. Animal Behaviour, 77(1), 29–36. https://doi.org/10.1016/j.anbehav.2008.08.032
Port, M., Johnstone, R. A., & Kappeler, P. M. (2012). The evolution of multimale groups in Verreaux’s sifaka, or how to test an evolutionary demographic model. Behavioral Ecology, 23(4), 889–897. https://doi.org/10.1093/beheco/ars053
Powzyk, J. A., & Mowry, C. B. (2003). Dietary and feeding differences between sympatric Propithecus diadema diadema and Indri indri. International Journal of Primatology, 24(6), 1143–1162. https://doi.org/10.1023/B:IJOP.0000005984.36518.94
Pusey, A., Williams, J., & Goodall, J. (1997). The influence of dominance rank on the reproductive success of female chimpanzees. Science, 277(5327), 828–831. https://doi.org/10.1126/science.277.5327.828
R Core Team. (2023). R: A language and environment for statistical computing. R Foundation for Statistical Computing. URL https://www.R-project.org/. Accessed 21 June 2024.
Ramanamisata, R., Pichon, C., Razafindraibe, H., & Simmen, B. (2014). Social behavior and dominance of the crowned sifaka (Propithecus coronatus) in northwestern Madagascar. Primate Conservation, 2014(28), 93–97. https://doi.org/10.1896/052.028.0117
Rasamimanana, H. (1999). Influence of social organization patterns on food intake of Lemur catta in the Berenty Reserve. In B. Rakotosamimanana, H. Rasamimanana, J. U. Ganzhorn, & S. M. Goodman (Eds.), New Directions in Lemur Studies (pp. 173–188). Springer. https://doi.org/10.1007/978-1-4615-4705-1_10
Rasolonjatovo, S. M., & Irwin, M. T. (2020). Exploring social dominance in wild diademed sifakas (Propithecus diadema): Females are dominant, but it is subtle and the benefits are not clear. Folia Primatologica, 91(4), 385–398. https://doi.org/10.1159/000503345
Razanaparany, P. T., & Sato, H. (2020). Abiotic factors affecting the cathemeral activity of Eulemur fulvus in the dry deciduous forest of north-western Madagascar. Folia Primatologica, 91(5), 463–480. https://doi.org/10.1159/000506128
Richard, A. F., & Nicoll, M. E. (1987). Female social dominance and basal metabolism in a Malagasy primate. Propithecus verreauxi. American Journal of Primatology, 12(3), 309–314. https://doi.org/10.1002/ajp.1350120308
Roeder, J. J., & Fornasieri, I. (1995). Does agonistic dominance imply feeding priority in lemurs? A study in Eulemur fulvus mayottensis. International Journal of Primatology, 15(5), 629–642. https://doi.org/10.1007/BF02735285
Roeder, J. J., Fornasieri, I., & Gosset, D. (2002). Conflict and postconflict behaviour in two lemur species with different social organizations (Eulemur fulvus and Eulemur macaco): A study on captive groups. Aggressive Behavior, 28(1), 62–74. https://doi.org/10.1002/ab.90006
Rolland, N., & Roeder, J. J. (2000). Do ringtailed lemurs (Lemur catta) reconcile in the hour post-conflict?: A pilot study. Primates, 41(2), 223–227. https://doi.org/10.1007/BF02557804
Romero, T., Colmenares, F., & Aureli, F. (2009). Testing the function of reconciliation and third-party affiliation for aggressors in hamadryas baboons (Papio hamadryas hamadryas). American Journal of Primatology, 71(1), 60–69. https://doi.org/10.1002/ajp.20619
Sauther, M. L. (1993). Resource competition in wild populations of ringtailed lemurs (Lemur catta): Implications for female dominance. In P. M. Kappeler & J. U. Ganzhorn (Eds.), Lemur Social Systems and Their Ecological Basis (pp. 135–152). Plenum Press. https://doi.org/10.1007/978-1-4899-2412-4_10
Schaffner, C. M., Aureli, F., & Caine, N. G. (2005). Following the rules: Why small groups of tamarins do not reconcile conflicts. Folia Primatologica, 76(2), 67–76.
Schino, G., Rosati, L., & Aureli, F. (1998). Intragroup variation in conciliatory tendencies in captive Japanese macaques. Behaviour, 135, 897–912. https://doi.org/10.1163/156853998792640314
Schülke, O., Bhagavatula, J., Vigilant, L., & Ostner, J. (2010). Social bonds enhance reproductive success in male macaques. Current Biology, 20(24), 2207–2210. https://doi.org/10.1016/j.cub.2010.10.058
Seex, L., Fichtel, C., Kappeler, P. M., & Hemelrijk, C. K. (2022). Interrelationship among spatial cohesion, aggression rate, counter-aggression and female dominance in three lemur species. Behavioral Ecology and Sociobiology, 76(10), 1–12. https://doi.org/10.1007/s00265-022-03241-2
Silk, J. B. (2002). The form and function of reconciliation in primates. Annual Review of Anthropology, 31, 21–44. https://doi.org/10.1146/annurev.anthro.31.032902.101743
Silk, J. B., Cheney, D. L., & Seyfarth, R. M. (1996). The form and function of post-conflict interactions between female baboons. Animal Behaviour, 52(2), 259–268. https://doi.org/10.1006/ANBE.1996.0171
Silk, J. B., Alberts, S. C., & Altmann, J. (2003). Social bonds of female baboons enhance infant survival. Science, 302(5648), 1231–1234. https://doi.org/10.1126/science.1088580
Silk, J. B., Beehner, J. C., Bergman, T. J., Crockford, C., Engh, A. L., Moscovice, L. R., ..., & Cheney, D. L. (2009). The benefits of social capital: Close social bonds among female baboons enhance offspring survival. Proceedings of the Royal Society B: Biological Sciences, 276(1670), 3099–3104. https://doi.org/10.1098/rspb.2009.0681
Simmen, B., Morino, L., Blanc, S., & Garcia, C. (2021). The energy allocation trade-offs underlying life history traits in hypometabolic strepsirrhines and other primates. Scientific Reports, 11(1), 14196. https://doi.org/10.1038/s41598-021-93764-x
Sterck, E. H. M., Watts, D. P., & van Schaik, C. P. (1997). The evolution of female social relationships in nonhuman primates. Behavioral Ecology and Sociobiology, 41(5), 291–309. https://doi.org/10.1007/S002650050390
Sussman, R. W., Garber, P. A., & Cheverud, J. M. (2005). Importance of cooperation and affiliation in the evolution of primate sociality. American Journal of Physical Anthropology, 128(1), 84–97. https://doi.org/10.1002/AJPA.20196
Thierry, B. (2000). Covariation of conflict management patterns across macaque species. In F. Aureli & F. B. M. de Waal (Eds.), Natural conflict resolution (pp. 106–128). University of California Press. https://doi.org/10.1525/9780520924932-010
Tibbetts, E. A., Pardo-Sanchez, J., & Weise, C. (2022). The establishment and maintenance of dominance hierarchies. Philosophical Transactions of the Royal Society B, 377(1845), 20200450. https://doi.org/10.1098/rstb.2020.0450
van Schaik, C. P., & Kappeler, P. M. (1996). The social systems of gregarious lemurs: Lack of convergence with anthropoids due to evolutionary disequilibrium? Ethology, 102(7), 915–941. https://doi.org/10.1111/j.1439-0310.1996.tb01171.x
Verbeek, P., & de Waal, F. B. M. (1997). Postconflict behavior of captive brown capuchins in the presence and absence of attractive food. International Journal of Primatology, 18(5), 703–725. https://doi.org/10.1023/A:1026391728909
WaeberHemelrijk. (2003). Female dominance and social structure in Alaotran gentle lemurs. Behaviour, 140(10), 1235–1246. https://doi.org/10.1163/156853903771980576
Westlund, K., Ljungberg, T., Borefelt, U., & Abrahamsson, C. (2000). Post-conflict affiliation in common marmosets (Callithrix jacchus jacchus). American Journal of Primatology, 52(1), 31–46. https://doi.org/10.1002/1098-2345(200009)52:1%3c31::AID-AJP3%3e3.0.CO;2-Z
White, F. J., Overdorff, D. J., Keith-Lucas, T., Rasmussen, M. A., Kallam, W. E., & Forward, Z. (2007). Female dominance and feeding priority in a prosimian primate: Experimental manipulation of feeding competition. American Journal of Primatology, 69(3), 295–304. https://doi.org/10.1002/AJP.20346
Wittig, R. M., & Boesch, C. (2003). Food competition and linear dominance hierarchy among female chimpanzees of the Tai National Park. International Journal of Primatology, 24, 847–867. https://doi.org/10.1023/A:1024632923180
Wrangham, R. W. (1980). An ecological model of female-bonded primate groups. Behaviour, 75(3/4), 262–300. https://doi.org/10.1163/156853980X00447
Wright, P. C. (1999). Lemur traits and Madagascar ecology: Coping with an island environment. American Journal of Physical Anthropology, 110(S29), 31–72. https://doi.org/10.1002/(SICI)1096-8644(1999)110:29+<31::AID-AJPA3>3.0.CO;2-00
Acknowledgements
We wish to thank Dr. Jonah RATSIMBAZAFY, Dr. Rose Marie RANDRIANARISON, Le Groupe d’Etudes et de Recherche sur les Primates de Madagascar, and the Malagasy Institute for the Conservation of Tropical Environments (MICET) for logistical support in the field. We would like to thank the government of Madagascar and CAFF/CORE for their permission to conduct the described research. In addition, we would like to thank the following individuals for their invaluable assistance in collecting the data presented in this paper: Herinjetovo MAMINIRINA, Jean de Dieu RAKOTOMALALA, Jean Odilon RAFANOMEZANTSOA, Onja RANDRIAMALALA, Tsinjo Sombiniaina Annick ANDRIATIAVINA, Sahobiarisoa Lalaina RANDRIANALY, Hasinirina RAVAOARISOA, Diary Nandrianina RZAFIMANDIMBY, Lokimonina Nasandratra RAHERIMANDIMBY, Sonne Martin RAKOTOMALALA, Blandine LALAONIRINA, and Elyseé MBOLATIANA. We would like to thank Aileen Cornejo and Michael Ball for their help in assembling the dataset. We would also like to thank Allie Schrock and the Caillaud lab for their comments on previous drafts of the manuscript. We additionally would like to thank Christof Neumann, Onja RAZAFINDRATSIMA, Joanna Setchell, and two anonymous reviewers for their comments which greatly improved the manuscript. This study was funded by NSF GRFP (grant 1650042), Primate Conservation, Inc., Greenville Zoo Conservation Fund, Pittsburgh Zoo & PPG Aquarium Conservation Fund, IDEAWild, the International Primatological Society, Bucknell University Animal Behavior research fellowship, Bucknell University Statistics research fellowship, and the Richard G. Coss Wildlife Research Fund from University of California, Davis.
Author information
Authors and Affiliations
Contributions
SOR and MCL conceptualized the experiments. SOR curated the data. SOR and MCL conducted the data analysis. MCL and DC acquired the necessary funding and provided supervision. SOR wrote the original draft, and all authors participated in the review and editing process.
Corresponding authors
Additional information
Handling Editors: Onja Razafindratsima and Joanna M. Setchell


Badge earned for open practices: Open Data and Open Code Badges. Experiment materials and data are available in the repository at https://doi.org/10.6084/m9.figshare.23293043.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ostad Rahimi, S., Caillaud, D. & Lutz, M.C. Patterns of Conflict and Post-Conflict Affiliation in Propithecus diadema and Eulemur fulvus. Int J Primatol (2024). https://doi.org/10.1007/s10764-024-00461-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10764-024-00461-3