Abstract
Synchrotron based nuclear forward scattering (NFS) experiments using the 193Ir nucleus have been performed for the first time on a dinuclear iridium(I) complex, [IrCl(COD)]2 with COD being cycloocta-1,5-diene. This complex serves as a catalyst for hydrogenation and other chemical reactions. Both, the obtained absolute values of the isomer shift \(\delta =0.87 \ {\text{mm s}}^{-1}\) and the quadrupole splitting \(\varDelta {E}_{Q}=3.82 \ {\text{mm s}}^{-1}\) agree within the experimental error with values obtained via conventional 193Ir Mössbauer spectroscopy reported earlier (Gál M. et al. J. Radioanal. Nucl. Chem., 260 (1) 2004, 133). In addition, we present density functional theory (DFT) calculations of the complex yielding its electronic structure and related Mössbauer parameters.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Mössbauer spectroscopy using the 73 keV transition from the I = 3/2 ground to the I = 1/2 first excited state of 193Ir has been considered as an optimal method to study hyperfine interactions in iridium containing materials because of its low natural line width of \(0.625 \ {\text{mm s}}^{-1}\) [1]. However, 193Ir Mössbauer spectroscopy requires 193Os as a radioactive source which has a half life time of 31d and needs to be prepared via neutron irradiation by a 192Os(n,γ)193Os reaction [2, 3]. This has hampered more widespread applications of 193Ir Mössbauer spectroscopy in the past although the 193Ir isotope has a high natural abundance of 62%.
The high brilliance at modern synchrotron sources and the recent development of monochromator systems for 73 keV with an energy resolution of 160 meV at the Dynamics Beamline P01 (PETRA III, DESY, Hamburg) now enables to excite the 73 keV level from the ground state to perform coherent nuclear forward scattering (NFS) experiments using the 193Ir nucleus. This set-up has been developed and used by Alexeev et al. [4] for the studies of magnetic and electronic properties of iridates which display both strong spin orbit coupling and strongly correlated electron systems.
Iridium containing materials play an important role in chemistry. For example, in photochemistry molecular iridium complexes are used in organic light-emitting diodes (OLEDs) [5, 6], organic solar cells [7], in photocatalysis [8], in car exhaust catalysts but also recently for initiating “water oxidation reactions " [9, 10]. In this process water is catalytically split into hydrogen and oxygen, a prerequisite to enable hydrogen as a sustainable storable energy source.
Here, we report the first 193Ir NFS experiments on a molecular material, namely a dinuclear iridium(I) complex, [IrCl(COD)]2 [11] with COD being cycloocta-1,5-diene (Fig. 1). This complex serves as a catalyst for hydrogenation and other chemical reactions [12,13,14,15,16,17,18,19,20,21,22]. It has been investigated by conventional 193Ir Mössbauer spectroscopy in the past [23, 24] which makes this system ideal for elucidating the potential of 193Ir NFS with respect to its chemical applications. In addition, we present density functional theory (DFT) calculations, which have been used to calculate the isomer shift and the quadrupole splitting of the dinuclear iridium(I) complex.
Structural view of the dinuclear iridium(I) complex [IrCl(COD)]2 investigated in this study. Ir atoms are displayed in dark blue, chloride atoms in green, carbon atoms in dark grey and hydrogens in grey [11]
2 Materials and methods
Di-µ-chlorobis[(1,2,5,6-η)-1,5-cyclooctadiene]diiridium (C16H24Cl2Ir2; CAS No.: 12112-67-3) was synthesized as described in [25]. For 193Ir NFS experiments the sample was filled in a hole with a diameter of 2 mm and a length of 4 mm of a sample holder made of aluminium. The sample was tightly pressed into the sample volume and sealed with aluminium tape.
193Ir NFS experiments were performed at the Dynamics Beamline P01 (PETRA III, DESY, Hamburg) using the 40-bunch mode with a time separation of 192 ns between the electron bunches of the PETRA III storage ring.
The synchrotron radiation (SR) generated by the undulator source was monochromatized with a double-crystal monochromator (DCM) consisting of two Si(311) crystals (see Fig. 2) to about 10 eV. The medium resolution monochromator (MRM) reduced the energy bandwidth to about 160 meV. The MRM consisted of two asymmetric channel-cut silicon crystals, a Si(422) collimator crystal and a Si(800) energy selector crystal. Subsequently, the SR, monochromatized to 73.0 keV, was transmitted through the sample mounted in a He-closed cycle cryostat from Advanced Research Systems, Inc. The delayed resonantly scattered radiation was detected with an avalanche photo diode (APD) detector array. The APD detector allowed a time resolution of \(\sim\)0.6 ns and 193Ir-NFS time spectra could be obtained as early as 3 ns after the excitation by the SR pulses by using time gated electronics.
For the determination of the quadrupole splitting (\(\varDelta {E}_{Q}\)) and the isomer shift (\(\delta\)) the 193Ir-NFS data were analyzed with the CONUSS software [26] as described in Alexeev et al. [4].
DFT calculations were used to calculate the hyperfine parameters \(\delta\) and \(\varDelta {E}_{Q}\) on the basis of the crystal structure of [IrCl(COD)]2 [11]. Structure optimization and Natural bond orbital (NBO) analysis [27] was performed with Gaussian 16 [28] using Grimme’s dispersion with the original D3 damping function [29] for the functional TPSSTPSS and the basis set QZVP [30, 31]. Kohn-Sham Molecular orbitals (MOs) and their energies were calculated and graphically represented by the Gauss View mode.
With the optimized structures, calculations of the hyperfine parameters were performed using the Orca 5.0 programme [32]. For this purpose, all-electron calculations of the SARC (segmented all-electron relativistically contracted) basis sets were used, which have been specially developed for scalar relativistic calculations and have been adapted to the Douglas-Kroll-Hess Hamiltonian of the second order (DKH2) [31]. The DKH-def2-TZVP basis set [30] was used for C, H, F, Br and Cl and the SARC-DKH-TZVP basis set [33] was used for the two Ir atoms. Calculations were performed with both TPSS and B3LYP functionals. The convergence criteria and grid points for the self-consistent field (SCF) calculations were set to Tight SCF (energy change \(1\cdot {10}^{-8} au\)). All SCF calculations were performed with the resolution of the Identity Approximation (RI) [34]. The programme Orca_eprnmr implemented in Orca was used to calculate the electron density \({\rho }_{0}\) and the electric field gradient (EFG) tensor at the iridium core [34].
3 Results and discussion
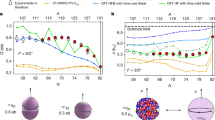
Figure 3 shows a 193Ir NFS spectrum of [IrCl(COD)]2 obtained at T = 10 K. The spectrum shows a beating pattern with a time period of about 5 ns which originates from the non-zero EFG of the two equivalent Ir sites in [IrCl(COD)]2. The beating pattern could be successfully reproduced by a simulation with CONUSS which gave \(\varDelta {E}_{Q}=3.82\left(4\right) {\text{ mm s}}^{-1}\). There are small deviations between experimental and simulated data occurring at > 25 ns which may be due by some spurious bunches of the synchrotron. Nevertheless, the so obtained value of the quadrupole splitting is in excellent agreement with those obtained by conventional 193Ir Mössbauer spectroscopy for this complex at liquid He temperatures (\(3.81\left(2\right) {\text{ mm s}}^{-1}\) [23] and \(3.85\left(2\right) {\text{ mm s}}^{-1}\), respectively [24]).
193Ir NFS spectrum of [IrCl(COD)]2 obtained at 10 K (black circles) (left) with the APD detector mounted behind the sample inside a closed cycle cryostat (right). The red line is a simulation performed with CONUSS [20] yielding \(\varDelta {E}_{Q}=3.82\left(4\right) \text{ mm s}^{-1}\)
For the determination of the isomer shift a data set with a metallic iridium foil as a single-line reference was collected. The corresponding 193Ir NFS spectrum is shown in Fig. 4. The interference of the 73 keV resonantly scattered quanta originating from the iridium foil and the [IrCl(COD)]2 sample leads to the disappearance of the regular beating structure visible in Fig. 3. A simulation with CONUSS using two Ir sites with \(\varDelta {E}_{Q}=3.82 \ {\text{mm s}}^{-1}\) representing [IrCl(COD)]2 and \(\varDelta {E}_{Q}=0 \ {\text{mm s}}^{-1}\) for the metallic Ir foil yields \(\delta =\pm 0.87(4) \ {\text{mm s}}^{-1}\) for the complex. It is important to note that the sign of \(\delta\) cannot be obtained using the set-up displayed in Fig. 4. Indeed, conventional 193Ir Mössbauer spectroscopy showed that the sign of the isomer shift of [IrCl(COD)]2 is negative. Nevertheless, the absolute \(\delta\)-value obtained in this study is in excellent agreement with the reported values of \(-0.88\left(1\right) {\text{ mm s}}^{-1}\) [23] and \(-0.87\left(1\right) {\text{ mm s}}^{-1}\) [24].
193Ir NFS spectrum of [IrCl(COD)]2 obtained at 10 K (black circles) (left) with an iridium foil mounted in the beam path (right). Temperature of the iridium foil was 300 K. The red line is a simulation performed with CONUSS [20] using \(\varDelta {E}_{Q}=3.82(4) \text { mm s}^{-1}\) for the Ir sites of [IrCl(COD)]2 yielding \(\delta =0.87(4) \text { mm s}^{-1}\) for the complex
It has been shown by [35] that experimentally observed isomer shifts are related to the theoretically calculated electron density at the iridium nucleus \({\rho }_{0}^{calc}\) via the following relation:
The parameters \(a\), \(b\) and \(c\) are fit parameters which need to be obtained from a series of complexes with known experimental values of \(\delta\) and calculated \({\rho }_{0}^{calc}\) which can be obtained by DFT methods based on known molecular structures. For 57Fe containing complexes this approach has been shown by various authors to be very successful [36, 37] The same strategy has been used recently within the frame of a DFT study to calculate 193Ir Mössbauer spectroscopic parameters [34].
For the determination of \({\rho }_{0}^{calc}\) of the complex [IrCl(COD)]2 investigated in this study DFT calculations of the various iridium(I), iridium(III) and iridium (IV) complexes listed in ref. [34] were repeated with the Orca 5.0 programme [32] as described in the Materials and Methods section. Table 1 provides a list of the iridium complexes and experimental \(\delta\) values and lists the charge and multiplicity of the complexes used for the calculations. The Cartesian coordinates of the complexes were taken from [34]. Our DFT calculations performed with both, the functional TPSS and B3LYP gave slightly different\({\rho }_{0}^{calc}\) values than reported in ref. [34] (Table 1) and were used to perform a linear regression analysis between \({\rho }_{0}^{calc}\) and measured \(\delta\) values as shown for both functionals in Fig. 5. For [IrCl(COD)]2 we obtain electron densities at the iridium core of 2657326.5773 \({\text{a}\text{u}}^{-3}\) when using TPSS and 2658125.7642 \({\text{a}\text{u}}^{-3}\) when using B3LYP. With the parameters \(a\), \(b\) and \(c\) given in Table 2 the calculated isomer shifts for the complex are \({\delta }_{TPSS}=-0.58 \text{ mm s}^{-1}\) and \({\delta }_{B3LYP}=-0.65 \text{ mm s}^{-1}\). Although the absolute values of the calculated isomer shifts are below the experimental value of \(\delta =-0.87 \text{ mm s}^{-1}\) the DFT calculations also give a negative sign of the isomer shift as has been observed by conventional 193Ir Mössbauer spectroscopy.
Experimental \(\delta\) -values as a function of calculated \({\rho }_{0}\)-values for the TPSS (a) and B3LYP (b) functionals. The black points represent the experimental data (Table 1) and the red line the result of a linear regression analysis with parameters \(a\), \(b\) and \(c\) listed in Table 2
The DFT calculations performed in this study also deliver the main components of the EFG tensor \({V}_{xx}\), \({V}_{yy}\) and \({V}_{zz}\) in its principal axis system (\(\mid {V}_{xx} \mid \le \mid {V}_{yy} \mid \le \mid {V}_{zz} \mid\)). With the asymmetry parameter \(\eta =({V}_{xx}-{V}_{yy})/{V}_{zz}\) and the quadrupole moment Q the quadrupole splitting is given as:
Taking Q = 0.751 b for the first excited nuclear state of 193Ir and the expression \(eQ{V}_{zz}\left[{mm \ s}^{-1}\right]=(eQ{V}_{zz}\left[J\right]\times c\left[{mm \ s}^{-1}\right])/{E}_{\gamma }\left[J\right]\) with the speed of light c in units of \({mm \ s}^{-1}\) allows to obtain \(\varDelta {E}_{Q}\) in its usual units since the DFT package ORCA delivers the EFG tensor components in units of au.
In this way we obtained for both functionals TPSS and B3LYP a positive sign of the quadrupole splitting and slightly different absolute values of \(\varDelta {E}_{Q}^{B3LYP}=+4.70 \ { \text{mm s}}^{-1}\text{; }\eta^{B3LYP}=0.29\) and \(\varDelta {E}_{Q}^{TPSS}=+4.25 \ { \text{mm s}}^{-1}\text{; }\eta^{TPSS}=0.43\). Giving the fact that for quadrupole splittings of 57Fe containing compounds deviations between experimental and DFT calculated \(\varDelta {E}_{Q}\) values in the order of \({ \sim1 \ \text{mm s}}^{-1}\) are not uncommon [39] we consider the agreement with the experimental value of \(\varDelta {E}_{Q}=3.82 \ {\text{mm s}}^{-1}\) at least for the complex [IrCl(COD)]2 investigated here as reasonable.
Gal et al. [24] argued that \(\delta =-0.87 \ \text{mm s}^{-1}\) of [IrCl(COD)]2 is unusually high compared to other Ir(I) complexes which show typically \(\delta \sim-4 \ \text{mm s}^{-1}\). They attributed this to a σ donation into the 6s orbitals as well as hybridization of the 6s with the 5\({d}_{{z}^{2}}\) orbitals. According to our NBO analysis, the electron configuration of the iridium is 6s(0.48)5d( 8.02)6p( 0.32)7p( 0.19). The eight occupied MOs with 5d character according to the reference frame given in Fig. 6 are shown in Fig. 7. Our calculations indicate that the 5p and 5s-orbitals prevail their character as expected (Fig. 8).
The DFT calculations presented here show a positive \({V}_{zz}\) which is in contradiction with the reported presumably negative \({V}_{zz}\) assumed by Gal et al. [24]. Future experimental investigations with NFS experiments in high external fields can serve to determine the sign of the quadrupole splitting and may shine more light on the binding properties of catalytically active iridium complexes.
4 Conclusions
In this work it has been shown that the 193Ir NFS is an excellent alternative to conventional 193Ir Mössbauer spectroscopy. Moreover, we have shown that it is possible to calculate Mössbauer parameters like the isomer shift and the quadrupole splitting using state of the art DFT methods with satisfying accuracy. The combination of experimental 193Ir NFS and quantum chemical DFT methods may represent an important technique for future characterisation of the magnetic and electronic properties of iridium containing molecular systems.
Data availability
Any data is available on request to either the first or the communicating author.
References
Greenwood N. N., Gibb T. C.: Other Transition-metal Elements. In: N. N. Greenwood, T. C. Gibb) (eds.) Mössbauer Spectroscopy, pp 493–535. Dordrecht: Springer Netherlands (1971)
Wagner, F., Zahn, U.: Mssbauer isomer shifts, hyperfine interactions, and magnetic hyperfine anomalies in compounds of iridium. Z. Physik 233(1), 1–20 (1970). https://doi.org/10.1007/BF01396512
Perlow, G.J., et al.: Hyperfine Anomaly in Ir193 by Mössbauer Effect, and its application to determination of the Orbital Part of Hyperfine Fields. Phys. Rev. Lett. 23(13), 680–682 (1969). https://doi.org/10.1103/PhysRevLett.23.680
Alexeev, P., et al.: Nuclear resonant scattering from 193Ir as a probe of the electronic and magnetic properties of iridates. Sci. Rep. 9(1), 5097 (2019). https://doi.org/10.1038/s41598-019-41130-3
Yang, C.-H., et al.: Blue-emitting heteroleptic iridium(III) complexes suitable for high-efficiency phosphorescent OLEDs. Angewandte Chemie 46(14), 2418–2421 (2007). https://doi.org/10.1002/anie.200604733. (International ed. in English)
Reddy, M., Bejoymohandas, K.S.: Evolution of 2, 3′-bipyridine class of cyclometalating ligands as efficient phosphorescent iridium(III) emitters for applications in organic light emitting diodes. J. Photochem. Photobiol. C 29, 29–47 (2016). https://doi.org/10.1016/j.jphotochemrev.2016.10.001
Dragonetti, C., et al.: Simple novel cyclometallated iridium complexes for potential application in dye-sensitized solar cells. Inorg. Chim. Acta 388, 163 (2012). https://doi.org/10.1016/j.ica.2012.03.028
Day J. I. et al.: Advances in photocatalysis: a microreview of visible light mediated ruthenium and iridium catalyzed organic transformations. Org. Process Res. Dev. 20(7), 1156–1163 (2016). https://doi.org/10.1021/acs.oprd.6b00101
McDaniel, N.D., et al.: Cyclometalated iridium(III) Aquo complexes: Efficient and tunable catalysts for the homogeneous oxidation of water. J. Am. Chem. Soc. 130(1), 210–217 (2008). https://doi.org/10.1021/ja074478f
Hull, J.F., et al.: Highly active and robust Cp* iridium complexes for catalytic water oxidation. J. Am. Chem. Soc. 131(25), 8730–8731 (2009). https://doi.org/10.1021/ja901270f
Bismuto, A., et al.: CCDC 1913379: Experimental crystal structure determination. Camb. Crystallographic Data Centre (2019). https://doi.org/10.5517/ccdc.csd.cc2270x8
Taguchi, K., et al.: An efficient direct alpha-alkylation of ketones with primary alcohols catalyzed by ir(cod)Cl2/PPh3/KOH system without solvent. J. Am. Chem. Soc. 126(1), 72–73 (2004). https://doi.org/10.1021/ja037552c
Sakaguchi, S., et al.: Iridium-catalyzed transfer hydrogenation of alpha,beta-unsaturated and saturated carbonyl compounds with 2-propanol. J. Org. Chem. 66(13), 4710–4712 (2001). https://doi.org/10.1021/jo0156722
Okimoto, Y., et al.: Development of a highly efficient catalytic method for synthesis of vinyl ethers. J. Am. Chem. Soc. 124(8), 1590–1591 (2002). https://doi.org/10.1021/ja0173932
Morita, M., et al.: One-Pot synthesis of gamma,delta-unsaturated carbonyl compounds from allyl alcohols and vinyl or isopropenyl acetates catalyzed by IrCl(cod)2. J. Org. Chem. 71(16), 6285–6286 (2006). https://doi.org/10.1021/jo060860j
Ishii, Y., Sakaguchi, S.: A novel catalysis of [{IrCl(Cod)} 2 ] Complex in Organic syntheses. BCSJ 77(5), 909–920 (2004). https://doi.org/10.1246/bcsj.77.909
Crabtree, R.H., Morris, G.E.: Some diolefin complexes of iridium(I) and a trans-influence series for the complexes [IrCl(cod)L]. J. Organomet. Chem. 135(3), 395–403 (1977). https://doi.org/10.1016/S0022-328X(00)88091-2
Kiyooka, S., et al.: [IrCl(cod)]2-catalyzed direct oxidative esterification of aldehydes with alcohols. Tetrahedron 63(51), 12695–12701 (2007). https://doi.org/10.1016/j.tet.2007.10.003
Stęsik, K., et al.: Hydrosilylation of Carbonyl compounds Catalyzed by Iridium(I) complexes with (–)-Menthol-based phosphorus(III) ligands. Chem. Cat. Chem. 15(9), (2023). https://doi.org/10.1002/cctc.202201510
Rahaman, S.W., et al.: The cyclooctadiene ligand in [IrCl(COD)] 2 is hydrogenated under transfer hydrogenation conditions: A study in the presence of PPh 3 and a strong base in isopropanol. J. Organomet. Chem. 829, 14–21 (2017). https://doi.org/10.1016/j.jorganchem.2016.10.009
Srinivas, V., et al.: Iridium-Catalyzed Hydrosilylation of Sulfur-Containing olefins. Org. Lett. 20(1), 12–15 (2018). https://doi.org/10.1021/acs.orglett.7b02940
Hesp, K.D., et al.: Ir(COD)Cl2 as a catalyst precursor for the intramolecular hydroamination of unactivated alkenes with primary amines and secondary alkyl- or arylamines: A combined catalytic, mechanistic, and computational investigation. J. Am. Chem. Soc. 132(1), 413–426 (2010). https://doi.org/10.1021/ja908316n
Tuczek, F., et al.: 193 ir Mössbauer investigations of Iridium-C 60 adducts. Fullerene Sci. Technol. 5(2), 443–452 (1997). https://doi.org/10.1080/15363839708012003
Gál, M., et al.: Mössbauer magnetization and nuclear magnetic resonance measurements on some iridium(I) complexes with fullerene ligands. J. Radioanal Nucl. Chem. 260(1), 133–142 (2004). https://doi.org/10.1023/B:JRNC.0000027072.00903.fe
Chen, D., et al.: Luminescent Iridium(III) complexes supported by a tetradentate trianionic ligand Scaffold with mixed O, N, and C donor atoms: Synthesis, structures, Photophysical Properties, and material applications. Organometallics 36(7), 1331–1344 (2017). https://doi.org/10.1021/acs.organomet.7b00038
Sturhahn, W.: CONUSS and PHOENIX: Evaluation of nuclear resonant scattering data. Hyperfine Interact 125(1/4), 149–172 (2000). https://doi.org/10.1023/A:1012681503686
Reed, A.E., Curtiss, L.A., Weinhold, F.: Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint. Chem. Rev. 88(6), 899–926 (1988). https://doi.org/10.1021/cr00088a005
Frisch, M.J., Trucks, G.W., Schlegel, H.B., Scuseria, G.E., Robb, M.A., Cheeseman, J.R., Scalmani, G., Barone, V., Petersson, G.A., Nakatsuji, H., Li, X., Caricato, M., Marenich, A.V., Bloino, J., Janesko, B.G., Gomperts, R., Mennucci, B., Hratchian, H.P., Ortiz, J.V., Izmaylov, A.F., Sonnenberg, J.L., Williams-Young, D., Ding, F., Lipparini, F., Egidi, F., Goings, J., Peng, B., Petrone, A., Henderson, T., Ranasinghe, D., Zakrzewski, V.G., Gao, J., Rega, N., Zheng, G., Liang, W., Hada, M., Ehara, M., Toyota, K., Fukuda, R., Hasegawa, J., Ishida, M., Nakajima, T., Honda, Y., Kitao, O., Nakai, H., Vreven, T., Throssell, K., Montgomery Jr., J.A., Peralta, J.E., Ogliaro, F., Bearpark, M.J., Heyd, J.J., Brothers, E.N., Kudin, K.N., Staroverov, V.N., Keith, T.A., Kobayashi, R., Normand, J., Raghavachari, K., Rendell, A.P., Burant, J.C., Iyengar, S.S., Tomasi, J., Cossi, M., Millam, J.M., Klene, M., Adamo, C., Cammi, R., Ochterski, J.W., Martin, R.L., Morokuma, K., Farkas, O., Foresman, J.B., Fox, D.J.: Gaussian 16 Rev. C.01. Gaussian Inc. Wallingford CT (2016)
Grimme, S., et al.: A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132(15), 154104 (2010). https://doi.org/10.1063/1.3382344
Weigend, F., Ahlrichs, R.: Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to rn: Design and assessment of accuracy. Phys. Chem. Chem. Phys. PCCP 7(18), 3297–3305 (2005). https://doi.org/10.1039/b508541a
Weigend, F.: Accurate coulomb-fitting basis sets for H to Rn. Phys. Chem. Chem. Phys. 8(9), 1057–1065 (2006). https://doi.org/10.1039/b515623h
Neese, F., et al.: The ORCA quantum chemistry program package. J. Chem. Phys. 152(22), 224108 (2020). https://doi.org/10.1063/5.0004608
Pantazis, D.A., et al.: All-Electron Scalar relativistic basis sets for third-row transition metal atoms. J. Chem. Theory Comput. 4(6), 908–919 (2008). https://doi.org/10.1021/ct800047t
Kaneko, M., Nakashima, S.: Density functional theory study on the 193Ir Mössbauer Spectroscopic parameters of Vaska’s complexes and their oxidative adducts. Inorg. Chem. 60(17), 12740–12752 (2021). https://doi.org/10.1021/acs.inorgchem.1c00239
Nieuwpoort, W.C., et al.: Calibration constant for Fe57 Mössbauer isomer shifts derived from ab initio self-consistent-field calculations on octahedral Fe F6 and Fe (CN)6 clusters. Phys. Rev. B 17(1), 91–98 (1978). https://doi.org/10.1103/PhysRevB.17.91
Han, W.-G., et al.: DFT calculations of 57Fe Mössbauer isomer shifts and quadrupole splittings for iron complexes in polar dielectric media: Applications to methane monooxygenase and ribonucleotide reductase. J. Comput. Chem. 27(12), 1292–1306 (2006). https://doi.org/10.1002/jcc.20402
Sinnecker, S., et al.: Performance of nonrelativistic and quasi-relativistic hybrid DFT for the prediction of electric and magnetic hyperfine parameters in 57Fe Mössbauer Spectra. Inorg. Chem. 44(7), 2245–2254 (2005). https://doi.org/10.1021/ic048609e
Wagner, F.E., et al.: Mössbauer study of pentammine and pyridine complexes of iridium. Chem. Phys. 4(2), 284–288 (1974). https://doi.org/10.1016/0301-0104(74)80095-9
Gütlich, P., et al.: Mössbauer spectroscopy and transition metal chemistry: fundamentals and applications. Springer, Berlin, Heidelberg (2011)
Acknowledgements
V.S. and H.J.K acknowledge the support by the Deutsche Forschungsgemeinschaft (DFG) through CRC/TRR88, “Cooperative Effects in Homo-and Hetero-Metallic Complexes(3MET)” and through CRC/TRR173 “Spin + X” (V.S.). V.S. also acknowledges support by the CRC/ German Federal Ministry of Education and Research (BMBF) under 05K22UK1.
Funding
Open Access funding enabled and organized by Projekt DEAL. Deutsche Forschungsgemeinschaft (DFG): CRC/TRR88, “3MET”, Project A04; CRC/TRR173 “Spin + X”, Project A04.
German Federal Ministry of Education and Research (BMBF): Project 05K22UK1.
Author information
Authors and Affiliations
Contributions
M.H.H. and V.S. wrote the main manuscript text. O.L., R.S., and I.S. performed the NFS experiments. O.L. and M.H.H. performed data analysis. J.W. and M.H.H. performed DFT calculations. R.R. and V.S. wrote application for beamtime. A.O., A.H. and H.J.K. provided the sample. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Ethical approval
N.A., no human and/ or animal studies.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
ESM 1
(PDF 177 KB)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hoock, M.H., Leupold, O., Haag, A. et al. 193Ir nuclear forward scattering of an iridium(I) complex. Hyperfine Interact 244, 24 (2023). https://doi.org/10.1007/s10751-023-01836-3
Accepted:
Published:
DOI: https://doi.org/10.1007/s10751-023-01836-3