Abstract
Transitioning from perennial to non-perennial flow regimes causes ecological shifts in aquatic communities. Aquatic macroinvertebrates deploy resistance and resilience strategies to cope with flow intermittency, crucial in rivers with long-term seasonal dry episodes. Less is known, about how these strategies support community persistence in streams that only recently have experienced drying, and where local assemblages lack such adaptations. Our study conducted two four-season campaigns, separated by a one-year break, to assess macroinvertebrate responses in newly drying intermittent streams by comparing intermittent and perennial stream sections. We characterize communities from structural and functional perspectives, and then evaluate the response at the trait state level. We observed a decline in taxa richness and abundance, but not structural diversity, in response to flow intermittency. Resistance traits are more important than resilient traits in structuring macroinvertebrate communities in newly intermittent stream sections. Taxa in intermittent sections exhibit a smaller trait space, indicating lower functional redundancy. The macroinvertebrate response to intermittency lacks a predictable pattern, suggesting time-dependent and trait-state-specific colonization by adapted taxa and community assembly with resistance and resilience strategies. As river drought increases due to climate change, recognizing the temporal dimension becomes crucial for understanding ecological responses to intermittency.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Global change affects stream communities directly and indirectly through changes in flow regimes (Arnell & Gosling, 2013; Schneider et al., 2013). A global increase in the spatial and temporal extent of river drying, representing a fundamental shift in the hydrological regime of newly intermittent streams (Carey et al., 2021; Zipper et al., 2021; Lucas-Picher et al., 2022) is predicted to continue (Palmer et al., 2008). The interactive effects of global change and anthropogenic pressures, such as water abstraction and impoundment, lead to more frequent dry phases by influencing stream flow dynamics (Messager et al., 2021; Cunillera-Montcusí et al., 2023). Intermittent rivers constitute more than 50% of the total length of the current global river network (Datry et al., 2014; Messager et al., 2021). Low-order streams are more at risk of drying due to their small size and low discharge because they can form more than 70% of river networks, the global river network is also strongly affected by drying (Lowe & Likens, 2005; Döll & Schmied, 2012; Datry et al., 2014, 2018). The shift from perennial to non-perennial flow regimes causes substantial changes in community and ecosystem dynamics (Aspin et al., 2018; Crabot et al., 2021a, 2021b; Carey et al., 2023).
The taxonomic alpha diversity of macroinvertebrates is generally lower in intermittent compared to perennial streams across regions, seasons, network positions and stream types (Bonada et al., 2007; Soria et al., 2017; Carey et al., 2023). Environmental filtering acts on traits rather than species, and therefore, the functional aspects of communities are receiving increasing attention. Resistance and resilience are the most important strategies that enable aquatic organisms to survive drying events (Bogan et al., 2015, 2017; Chessman, 2015; Strachan et al., 2015; Aspin et al., 2019). Resistance reflects the ability of the species—and cumulatively the community—to survive the dry phase locally, while resilience describes the capacity to recolonize after water returns (Bogan et al., 2017; Carey et al., 2021).
Our knowledge of how macroinvertebrate assemblages respond to drought originates from the regions where seasonal drought is a natural phenomenon and has occurred historically or has been observed in the past decade (e.g. Australia: Rolls et al., 2016, Mediterranean: Bonada et al. 2007, United Kingdom: Hill et al., 2019; Sarremejane et al., 2020b; and several regions reviewed in Acuña et al., 2017). In contrast, shifts from perennial to intermittent flow are a relatively new phenomenon in the Continental biogeographical region (including the Pannonian ecoregion, Vanneuville et al. 2012) of Europe (Rubel & Kottek, 2010; Arnell & Gosling, 2013; Bartholy et al., 2014). Despite predicted increases in climatic extremity and associated river drying (IPCC, 2021), few studies have characterized ecological responses to the transition from perennial to intermittent flow, including the mechanisms and processes that drive macroinvertebrate community responses to unprecedented river drying (Řezníčková et al. 2007; Pařil et al., 2019; Crabot et al., 2021a, 2021b). Characterizing how macroinvertebrate communities respond to the first dry phase in newly intermittent rivers can help to develop strategies that support the resilience of ecosystems adapting to climate change.
This study aimed to report the response of macroinvertebrate assemblages in previously perennial streams to the first appearance of drying. We characterize communities from a functional perspective and evaluate the response at the trait state level. To achieve this, we surveyed macroinvertebrate assemblages during and after the first drying event at multiple sites in a river network, and compared the perennial and intermittent sections, to characterize how assemblages adapted to perennial flow response to the first drying event in newly intermittent streams. We hypothesized that traits promoting resistance and resilience strategies are more important in structuring macroinvertebrate communities in newly drying intermittent stream sections compared to perennial ones.
Material and methods
Study area
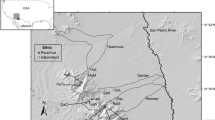
Field sampling was performed across a dendritic river network, the Bükkösdi-víz catchment, located in the Mecsek mountains in south-west Hungary (Fig. 1) within the Pannonian ecoregion (Vanneuville et al.2012). The recent climate is mainly temperate with a slight sub-Mediterranean influence, resulting in a moderately cool and dry climate. The annual temperature is 10.0–10.2 °C, the total annual sunshine duration is more than 2,000 h, and the average annual precipitation is approximately is around 760 mm, with slight differences among seasons (Ács et al., 2015; Mezősi, 2017). In the vicinity of the sampling site, as evidenced by data spanning the years 1991 to 2020, summer exhibits warm temperatures (mean: + 21.4 °C) accompanied by notable precipitation (mean: 72.3 mm), while winter is characterized by cold temperatures (mean: + 1.2 °C) and low precipitation levels (mean: 38.7 mm). Spring is marked by moderate temperatures (mean: + 11.6 °C) and precipitation (mean: 53 mm), whereas autumn experiences a gradual decline in temperatures (mean: + 11.6 °C) coupled with an increase in precipitation (mean: 60.3 mm) (OMSZ, 2024). Between 1981 and 2020, Hungary experienced a 1.7 °C increase in annual mean temperature, with the southwestern region, including the study site, showing a 1.5 °C rise. From 1901 to 2020, there was a 10% decrease in annual precipitation within the study area. Additionally, during this timeframe, the number of days with precipitation decreased by 17 days, while the longest dry period increased by 4 days (IEA, 2021). In line with global warming, regional temperatures are rising, and the study area is also becoming increasingly dry, rendering it one of the most vulnerable counties to drought and susceptible to the anticipated effects of drying (Buzási et al., 2021).
Sampling design
We studied 40 stream sections on first- to third-order streams including 18 perennial and 22 newly intermittent sections (Fig. 1). Sections alternating among flowing, non-flowing and dry phases were collectively called intermittent sections. Streams are characterized by a negligible amount of aquatic macrovegetation (less than 5% coverage), and rocky streambeds. In the upper part of the catchment, streams are in near-natural reference conditions; modest settlements are situated along the primary stream branch (along the third-order main stem), with minimal anthropogenic impact. All study stream sections belong to the same type in the European Union Water Framework Directive typology, i.e. high-altitude, calcareous streams with small or medium catchment areas, and they share the same regional species pool (Boda et al., 2023). During the Summer of 2012, a notable occurrence of drying phases was observed in certain sections, coinciding with the region's record-breaking heatwave duration, particularly in the southeastern part of Europe, encompassing Hungary (Ionita et al. 2021). However, apart from a single short-term (five weeks) drying event in 2012, the studied streams were characterized by stable, perennial flow until 2018, when drying was observed in September in several sections of streams. Macroinvertebrate sampling was initiated immediately after the onset of the first drying event.
Macroinvertebrate sampling
Macroinvertebrates were sampled seasonally in two four-season campaigns separated by a one-year break: in September 2018, February, April and July 2019; then in July and October 2020, and February and May 2021. Samples were collected if flowing water was present. Macroinvertebrate samplings were performed seasonally in eight sampling campaigns. Samples were collected using a standardized kick-and-sweep method with a 1-mm-mesh kick net with a 25 × 25 cm frame ensuring that all major habitat types were sampled in proportion to their occurrence (Boda et al., 2023). Each sample comprised 12 and 10 sample units in the first and second years, respectively. Each sample unit covers a 0.25 × 0.25 m area, thus the total sampling area was 0.625 m2 for 10 sampling units, and 0.75 m2 for 12 sample units, respectively. To account for the different numbers of sampling units in years 1 and 2, abundance data were converted to individuals m−2. Samples were pre-sorted in the field to identify protected species, which were registered and released. The rest of the sample was preserved in 70% ethanol. In the laboratory, all samples were sorted and all individuals were identified to species level, except for Diptera and Turbellaria, which were identified to family level.
In addition to the dates of the biological samplings, the entire sampling area, including all sampling sections, was checked on two additional occasions (in September and December 2020), resulting in a total of 10 instances when the wet or dry phases in the streambed were registered during the sampling campaigns (Online Resource Table S1).
Data analysis
To investigate the spatial autocorrelation, a Mantel test was conducted using Pearson's product-moment for correlation with 999 permutations comparing compositional distance with both direct geographical and (along-stream) hydrological distances. We first created dissimilarity matrices for the taxa data by calculating the pairwise community dissimilarities using the Bray–Curtis distance, then for the spatial distance, we calculated the dissimilarity matrices on pairwise Euclidean geographic and pairwise hydrological distances. The correlation between the geographical and hydrological distance was also tested. We calculated four structural metrics to compare macroinvertebrate assemblages in perennial and intermittent sections: mean taxa richness, abundance, Shannon diversity and evenness. The model matrix contains the calculated structural metrics, type (intermittent and perennial), sections and time. A linear mixed model was performed using the restricted maximum likelihood (REML) estimation method via the ‘lmer’ function in the lme4 R package (Bates et al., 2014), with section type (intermittent, perennial) as a fixed effect and sections and time (sampling date) as random effects.
A taxon can be referred to as a trait complex. As each taxon can possess several 'trait states' (i.e. trait categories or trait modalities), taxa can have a resistance-related trait state for one trait and a resilience-related trait state for another, while also possessing a non-related trait state. The functional trait data were gathered from the website freshwaterecology.info (Schmidt-Kloiber & Hering, 2015) and the DISPERSE database (Sarremejane et al., 2020a). To ensure the highest coverage, we used genus- and family-level data. If a trait was included in both databases, we used the database that had a higher data coverage for our dataset. We selected 62 trait states across 12 trait categories, including biological traits and ecological preferences associated with drying (Online Resource, Table S2).
Drought-relevant traits can be linked to either resilience or resistance, as they are the two main mechanisms for survival that act in different ways (Stubbington et al., 2017). Based on (Stubbington et al., 2017), See Table 4.3.1), for each trait, we identified the trait states that do/do not promote survival in case of flow intermittency (Online Resource, Table S2). For instance, a small body size trait state enhances resistance by promoting movement into the hyporheic zone during drying episodes, whereas large body sizes do not (Fig. 2). The same principle applies to resilience, where certain types of reproduction (such as ovoviviparity, terrestrial clutches, or asexual reproduction) increase resilience by promoting survival during dry episodes, but other states (e.g. isolated eggs and free clutches) do not (Fig. 2). Considering these distributions, all 62 trait states were assigned to one of four trait groups: Resilience (RL), non-Resilience (non-RL), Resistance (RT), and non-Resistance (non-RT) (Online Resource, Table S2).
We then compiled a biotic matrix (sampling occasion × genus) of relative abundance data (percentage of individuals / m2) and a trait matrix (genus × trait state) filled with the values of fuzzy codes from the trait databases. We calculated community-weighted means (CWM) to estimate community trait values weighted by the abundance of taxa in that community. Thus, combining these two matrices, a CWM matrix (sample × trait state) was calculated using Canoco (ter Braak & Smilauer, 1998). As a result, we got the CWM values for all sampling sections (both for intermittent and perennial sections) and the date for each state, respectively. The mean CWM values for each perennial and intermittent section, pertaining to each trait state, were calculated, respectively. T-test was used to calculate the difference between CMW values of intermittent and perennial sections. For each intermittent section, we subsequently computed the difference between the CWM values of trait states and the mean CWM values derived from all perennial sections. Utilizing a bar chart, we graphically illustrated the range of these differences, spanning from the highest to the lowest, with the median value prominently displayed within each bar. To visualize the differences for each trait state in each intermittent section, respectively, we created heatmaps in the R package ggplot2 (Garrett et al., 2018).
We plotted the trait states in each trait group associated with intermittent and perennial sections using the Indicator Taxa Analysis function in the biomonitoR package (Laini et al., 2022) with the Indicator Taxa Analysis function (IndVal; Dufrêne & Legendre, 1997). The function plot_indicator_taxa implements the group-equalised IndVal for both presence-absence and abundance data (De Cáceres & Legendre, 2009). We visualized significant relationships and trait states with indicator values higher than 60 with a Sankey diagram that links indicator trait states (De Cáceres & Legendre, 2009) to perennial and intermittent sections.
We quantified the amount of functional trait space occupied by the macroinvertebrate assemblages in intermittent and perennial sections. Two trait states (respiration: hydrostatic vesicle [aerial]; reproduction: asexual) that showed zero variance were omitted from the analyses. Following Schmera et al. (2022), the values of trait states belonging to the same trait were standardised to an interval [0,1], then a centred principal component analysis of incomplete data (Podani et al., 2021) was used to produce functional trait space. The multidimensional convex hull volume for the first five axes was calculated following the methodology outlined by Cornwell et al. (2006).
Results
We collected 598,573 individuals belonging to 280 taxa, from which 160 were identified species level and at 13 higher taxa (10 Gastropoda, 1 Bivalvia, 7 Hirudinea, 6 Crustacea, 19 Ephemeroptera, 14 Odonata, 3 Plecoptera, 23 Heteroptera, 35 Coleoptera, 4 Megaloptera, 30 Trichoptera, 7 Diptera and 1 Turbellaria).
The total number of taxa in perennial sections was higher (N = 161) than in intermittent sections (N = 142). Forty taxa were found exclusively in perennial sections, 21 taxa in intermittent sections and 121 taxa were observed in both sections. Linear mixed effect models showed that taxa richness was higher in perennials compared to intermittent sections (P = 0.041). Similarly, abundance was higher in perennial compared to intermittent sections (P = 0.005). In contrast, Shannon diversity and evenness were comparable in both section types (P = 0.550, P = 0.278 respectively; Fig. 3, Online Resource, Table S3).
There was no significant correlation between the taxonomic composition matrix and either the geographical (r = 0.139, P = 0.095) or hydrological (r = 0.695, P = 0.001) distance matrices, compositional similarity among sites was comparable regardless of distances between sections. There was a significant correlation between the geographical and hydrological distance (r = 0.694, P = 0.001).
The range of changes in the intermittent sections varied highly among intermittent sampling sections (Fig. 4b). The largest range of changes was observed at the following trait states the ≥ 1-year adult life span (als4), 2 similar-sized pairs of wings (wpt5), aerial active dispersal strategy (dis4), ovoviviparity reproduction (rep1) (RL); gill and tegument respiration (res2 and res1) [non-RT]; and No wings (Wpt1), burrower (lsr5) [non-RL]. RT states have generally a smaller range of changes (Fig. 4b). Individually, the smallest range of changes showed by clutches, free reproduction (Rep 5, non-RL), the housings against desiccation (ref3, RT), rheobiont in current preference (cup6, non-RT). Out of the 62 trait states, only three—the housings against desiccation (ref3-RT), rheobiont in current preference (cup6-non-RT) and preference for fast current velocity (cuv4-non-RT)—showed uniform pattern as they had negative deviations (red colour) in all intermittent sections compared to the mean of CWM of all perennial sections (Online Resource, Fig. S1). The section-specific changes varied highly among intermittent sampling sections (Online Resource, Fig. S1).
Functional trait state responses of macroinvertebrate communities to stream intermittency. a Significant (T-test, P < 0.05) negative (red) and positive (green) differences between the mean CWM values of trait states in intermittent compared to the mean CWM values in perennial streams for all sections, b The total range of the differences (bars) between the CWM values of trait states of intermittent sections and the mean CWM values derived from all perennial sections. The mean (●) and the total range (bars) of the intermittent sections are given. The gradient of colour represents the extent of differences from mean CWM values calculated from all perennial sections
In case of all-section changes in RT states, CWM values for 7 out of 8 states were higher in intermittent compared to perennial sections (Fig. 4a). For RL states, CWM values were higher in intermittent and perennial sections for six and nine states, respectively (Fig. 4a).
Most of the trait states were related to the perennial sections, and seven trait states were associated with intermittent sections (Fig. 5). Strong, significant relationships with section type were identified for 32 trait states: 16 with intermittent sections and 16 with perennial sections, whereas other states were not linked to either section (Online Resource, Table S3). No state group was detected where all the states are related only to perennial or intermittent sections. (Fig. 5). Macroinvertebrate assemblages from perennial and intermittent sections occupied 98% and 87% of the total functional space, respectively (Online Resource, Fig. S2).
Sankey diagram illustrating significant linkages between intermittent and perennial sections and trait states. Only states with indicator values higher than 60 are shown; others are listed in Online Resource, Table S3. All relationships are significant (P < 0.05); dark purple: Resistance trait states, light purple: non-Resistance trait states, dark green: Resilience trait states, light green: non-Resilience trait states
Discussion
The information we have on how macroinvertebrate assemblages respond to a drought mostly originates from regions where seasonal drought is a natural phenomenon, while our knowledge of how communities in perennial streams respond to drying is rather limited. We carried out two, four-season campaigns separated by a one-year break to examine the initial responses of macroinvertebrate assemblages to the drying of historically perennial streams by comparing macroinvertebrate assemblages from actual perennial and intermittent sections. We found that the richness and abundance of macroinvertebrate assemblages were lower in intermittent sections, but this is not reflected in diversity metrics. Resistance traits are more important than resilient traits in structuring macroinvertebrate communities in newly intermittent streams; however, trait states showed a section-specific and uneven response to drying. Our results also showed that taxa in intermittent sections occupied a lower amount of trait space than those of perennial ones, meaning that newly intermittent streams may be characterized by lower functional redundancy.
Drought-related disturbances generally reduce structural biological attributes such as taxa richness or abundance (Arscott et al., 2010; Bogan et al., 2013; Stubbington et al., 2017; White et al., 2018; Sarremejane et al., 2020b), which means that the less-resistant or less-resilient taxa vanish from the macroinvertebrate assemblages (Chase, 2007) when the drought intensity or severity increases. In line with previous findings, we found significantly lower taxa numbers and abundances in intermittent than in perennial stream sections (Boulton, 2003; Datry et al., 2014; Leigh & Datry, 2017). However, most of the taxa (66%, N = 121) can be found in both perennial and intermittent stream sections. Taxa that lack drying-resistance traits are able to tolerate short periods (from days to weeks, but not longer than a month) of drying, while taxa with resilient strategies can swiftly recover upon flow resumption, enabling rapid assemblage recovery (Pařil et al., 2019). A reduced number of drought-sensitive taxa consequently immediately decrease the diversity of a community (Arscott et al., 2010; Soria et al., 2017). Generally, taxa richness is significantly lower in intermittent than in perennial rivers (Soria et al., 2017). However, diversity changes can be affected by the timing, frequency and periodicity of drying (Crabot et al., 2020, 2021b). Greater decreases in alpha diversity occur more frequently at sites that are rarely dry compared to those that dry seasonally (Sarremejane et al., 2020b), and recent intermittency had a profound effect on species composition in streams affected by drying (Carey et al., 2023). In contrast, in our study, the decrease in taxa richness did not result in reduced diversity, as also found by Řezníčková et al. in Central European streams (2007). The modest reduction in taxa richness in our intermittent streams may indicate an initial response to the drying phenomenon, potentially escalating to a more pronounced and enduring decline during persistent drought (Hill et al., 2019). Alternatively, it could result from a rapid recovery process from nearby aquatic habitats, as connectivity considerably influences recolonization (Sarremejane et al., 2020b). Such decreases in taxa richness and abundance may represent a trade-off involving the non-selective elimination of the least abundant taxa (Pimm et al., 1988; Sarremejane et al., 2020b), the taxa eliminated by the drought and the abundance shift caused by taxa that exclusively occurs in intermittent sections. Even though the realignment of abundance features is a typical pattern in response to flow intermittency (Hille et al., 2014; Sarremejane et al., 2018, 2020b), no significant difference in evenness was observed (Fig. 4). Despite the reduced structural metrics, like richness and abundance in intermittent sections, diversity metrics such as Shannon diversity and Evenness did not differ between intermittent and perennial sections. This implies that the structural realignment in the intermittent sections was not reflected in the diversity metrics.
Generally, resistance and resilience strategies strongly respond to the flowing intermittency gradient. As flow intermittency increases, taxa with trait states that confer resistance and resilience to drying become more common in macroinvertebrate communities (Crabot et al., 2020, 2021a). Our findings reveal a prevalence of traits promoting resistance strategies in intermittent sections, driven by the following ecological factors: (i) shortened durations of water availability, which can limit larval and egg development of aquatic organisms; (ii) small size facilitating survival in interstitial water during dry episodes (Stubbington et al., 2019); (iii) the presence of resistance forms such as diapause or dormancy, eggs, or cocoons enabling organisms to withstand severe drying (Stubbington & Datry, 2013); (iv) limnophilic taxa showing a preference for slower currents (Hill et al., 2019); and (v) aerial respiration unaffected by drying due to independence from dissolved oxygen, which is less available in warmer shallow water (Stubbington, 2012; Crabot et al., 2021b). However, this intermittency-related increase cannot be observed for resilience trait states, as responses in these states are highly varied. The resistance trait states seem to be more sensitive to initial drying than the resilience ones, as more positive general changes occurred among resistance trait states than among resilience trait states (Fig. 4). Furthermore, with the increasing severity of drying, trait states that infer flow intermittency resistance or resilience may be selected at the expense of non-resistance and non-resilience states (Townsend & Hildrew, 1994; Aspin et al., 2018; Crabot et al., 2020, 2021b). Contrary to this, an unambiguous and mutually exclusive phenomenon in the relationship of intermittency-related and non-related states was not observed in our study. The section-specific and uneven response to the initial drought may be due to the transitional nature of hydrological regimes from perennial to intermittent across the whole catchment and the difference in their duration of flow cessation among the intermittent sections. Alternatively, it may indicate that taxa will inevitably continue to include some non-resistance and non-resilience trait states because any taxon only needs a few resistance or resilience traits to survive. Traits should be sorted along predictable lines for disturbance gradients (Southwood, 1977; Townsend & Hildrew, 1994). Drying as the primary hydrological determinant (Leigh & Datry, 2017) creates different habitats in perennial and intermittent sections that can accommodate taxa with different traits. This means that when the severity of drying increases, most (if not all) of the resistance- and resilience-related states are higher in the intermittent stream sections (Aspin et al., 2018; Crabot et al., 2021a). However, in our study of newly intermittent streams, we detected no such single trend for all states in a state group. The opposite phenomenon was observed since there is at least one state from each state group that can be linked more likely to perennial or intermittent sections (see Fig. 5). The potential endpoint of states or state groups at the conclusion of the transition process cannot be easily predicted, as flexible phenology or growth rates are not necessarily indicated by life-history traits (Carey et al., 2021). While phenology typically evolves in response to stable water regimes, populations can adapt to new conditions through phenological plasticity (Robinson & Buser, 2007).
Assemblages can fill different niches in the functional trait space between different types of habitats (Schmera et al., 2022). Thus, evaluating the functional traits space of macroinvertebrates helps identify functional differences among groups, enhancing our understanding of ecological communities in drying environments. We found that the functional trait space in intermittent sections is reduced compared to perennial sections, which suggests that intermittent sections have less complexity in their functional structure and that, consequently, communities in newly intermittent streams may have reduced capacity to cope with environmental change. Taxa replacement and an overall decrease in taxa numbers may reduce functioning, suggesting that functional redundancy should have a minor role in the assemblages in perennial streams. These streams may be characterized by lower functional redundancy than historically intermittent drying networks (Boersma et al., 2013; Sarremejane et al., 2020b; Crabot et al., 2021b). Changes in structural and functional aspects, however, are context-dependent, and they are strongly influenced by environmental elements such as connectivity (Sarremejane et al., 2020b; Crabot et al. 2021a), proximity of refuges (Gill et al., 2022), climatic characteristics (Vander Vorste et al., 2021), and temporal and spatial aspects of the drought (Aspin et al., 2019; Crabot et al., 2020, 2021a; Chanut et al., 2023). Therefore, these influencing factors of drought can have long-lasting effects on stream communities, potentially triggering substantial and irreversible changes (Cassidy et al. 2020).
Along with the intensification of climate change, the temporal aspects of the adaptation of aquatic organisms to drying are gaining more interest (Crabot et al., 2020; Carey et al., 2023). In this study, we showed that resistance is a more important strategy than resilience in intermittent streams; however, the response at the trait state level is section-specific and uneven to initial drying. The macroinvertebrate community does not have a well-defined response to a never-experienced environmental stress. More broadly, our results illustrate the need to consider the temporal context (in terms of time elapsed since the first appearance of drying), especially when evaluating ecological responses to drying.
Data availability
Data are available from the authors upon reasonable request.
References
Ács, F., H. Breuer & N. Skarbit, 2015. Climate of Hungary in the twentieth century according to Feddema. Theoretical and Applied Climatology 119: 161–169. https://doi.org/10.1007/s00704-014-1103-5.
Acuña, V., M. Hunter & A. Ruhi, 2017. Managing temporary streams and rivers as unique rather than second-class ecosystems. Biological Conservation. https://doi.org/10.1016/j.biocon.2016.12.025.
Arnell, N. & S. Gosling, 2013. The impacts of climate change on river flow regimes at the global scale. Journal of Hydrology 486: 351–364. https://doi.org/10.1016/j.jhydrol.2013.02.010.
Arscott, D., S. Larned, M. Scarsbrook & P. Lambert, 2010. Aquatic invertebrate community structure along an intermittence gradient: Selwyn River, New Zealand. Journal of the North American Benthological Society 29: 530–545. https://doi.org/10.1899/08-124.1.
Aspin, T., T. Matthews, K. Khamis, A. Milner, Z. Wang, M. O’Callaghan & M. Ledger, 2018. Drought intensification drives turnover of structure and function in stream invertebrate communities. Ecography. https://doi.org/10.1111/ecog.03711.
Aspin, T. W. H., K. Khamis, T. J. Matthews, A. M. Milner, M. J. O’Callaghan, M. Trimmer, G. Woodward & M. E. Ledger, 2019. Extreme drought pushes stream invertebrate communities over functional thresholds. Global Change Biology 25: 230–244. https://doi.org/10.1111/gcb.14495.
Bartholy, J., R. Pongrácz & I. Pieczka, 2014. How the climate will change in this century? Hungarian Geographical Bulletin 63: 55–67. https://doi.org/10.15201/hungeobull.63.1.5.
Bates, D., M. Mächler, B. Bolker & S. Walker, 2014. Fitting Linear Mixed-Effects Models Using lme4. ArXiv e-prints arXiv:1406: https://doi.org/10.18637/jss.v067.i01.
Boda, P., G. Várbíró & M. Ficsór, 2023. Módszertani Kézikönyv a Víz Keretirányelv feladataihoz kapcsolódóan a makroszkopikus vízi gerinctelenek mintavételéhez és ökológiai állapotértékeléséhez. Ökológiai Kutatóközpont, Budapest, http://real.mtak.hu/157944/.
Boersma, K., M. Bogan, B. Henrichs & D. Lytle, 2013. Invertebrate assemblages of pools in arid-land streams have high functional redundancy and are resistant to severe drying. Freshwater Biology. https://doi.org/10.1111/fwb.12280.
Bogan, M., K. Boersma & D. Lytle, 2013. Flow intermittency alters longitudinal patterns of invertebrate diversity and assemblage composition in an arid-land stream network. Freshwater Biology 57: 1016–1028. https://doi.org/10.1111/fwb.12105.
Bogan, M. T., K. S. Boersma & D. A. Lytle, 2015. Resistance and resilience of invertebrate communities to seasonal and supraseasonal drought in arid-land headwater streams. Freshwater Biology 60: 2547–2558. https://doi.org/10.1111/fwb.12522.
Bogan, M., E. Chester, T. Datry, A. Murphy, B. Robson, A. Ruhi, R. Stubbington & J. Whitney, 2017. Chapter 4.8: Resistance, resilience, and community recovery in intermittent rivers and ephemeral streams.
Bonada, N., M. Rieradevall & N. Prat, 2007. Macroinvertebrate community structure and biological traits related to flow permanence in a Mediterranean river network. Hydrobiologia 589: 91–106. https://doi.org/10.1007/s10750-007-0723-5.
Boulton, A. J., 2003. Parallels and contrasts in the effects of drought on stream macroinvertebrate assemblages. Freshwater Biology 48: 1173–1185. https://doi.org/10.1046/j.1365-2427.2003.01084.x.
ter Braak, C. & P. Smilauer, 1998. CANOCO Reference Manual and User’s Guide to Canoco for Windows: Software for Canonical Community Ordination (Version 4) Microcomputer Power, Ithaca, New York, USA, pp. 352
Buzási, A., T. Pálvölgyi & D. Esses, 2021. Drought-related vulnerability and its policy implications in Hungary. Mitigation and Adaptation Strategies for Global Change 26: 1–20. https://doi.org/10.1007/s11027-021-09943-8.
Carey, N., E. Chester & B. Robson, 2021. Life history traits are poor predictors of species responses to flow-regime change in headwater streams. Global Change Biology. https://doi.org/10.1111/gcb.15673.
Carey, N., E. Chester & B. Robson, 2023. Loss of functionally important and regionally endemic species from streams forced into intermittency by global warming. Global Change Biology. https://doi.org/10.1111/gcb.16650.
Cassidy, C., L. J. Grange, C. Garcia, S. G. Bolam & J. A. Godbold, 2020. Species interactions and environmental context affect intraspecific behavioural trait variation and ecosystem function. Proceedings of the Royal Society B 287(1919): 20192143. https://doi.org/10.1098/RSPB.2019.21.
Chanut, P., A. Drost, A. Siebers, A. Paillex & C. Robinson, 2023. Flow intermittency affects structural and functional properties of macroinvertebrate communities in alpine streams. Freshwater Biology. https://doi.org/10.1111/fwb.14018.
Chase, J., 2007. Drought mediates the importance of stochastic community assembly. Proceedings of the National Academy of Sciences of the United States of America 104: 17430–17434. https://doi.org/10.1073/pnas.0704350104.
Chessman, B., 2015. Relationships between lotic macroinvertebrate traits and responses to extreme drought. Freshwater Biology. https://doi.org/10.1111/fwb.12466.
Cornwell, W. K., D. W. Schwilk & D. D. Ackerly, 2006. A trait-based test for habitat filtering: convex hull volume. Ecology 87: 1465–1471. https://doi.org/10.1890/0012-9658(2006)87[1465:ATTFHF]2.0.CO;2.
Crabot, J., J. Heino, B. Launay & T. Datry, 2020. Drying determines the temporal dynamics of stream invertebrate structural and functional beta diversity. Ecography 43: 620–635. https://doi.org/10.1111/ecog.04835.
Crabot, J., M. Polášek, B. Launay, P. Pařil & T. Datry, 2021a. Drying in newly intermittent rivers leads to higher variability of invertebrate communities. Freshwater Biology 66(4): 730–744. https://doi.org/10.1111/fwb.13673.
Crabot, J., C. Mondy, P. Usseglio-Polatera, K. Fritz, P. Wood, M. Greenwood, M. Bogan, E. Meyer & T. Datry, 2021b. A global perspective on the functional responses of stream communities to flow intermittence. Ecography. https://doi.org/10.1111/ecog.05697.
Cunillera-Montcusí, D., J. M. Calero, S. Pölsterl, R. Argelich, P. Fortuño, N. Cid, N. Bonada & M. Cañedo-Argüelles, 2023. Navigating through space and time: A methodological approach to quantify spatiotemporal connectivity using stream flow data as a case study. Methods in Ecology and Evolution 14: 1–16. https://doi.org/10.1111/2041-210X.14105.
Datry, T., S. T. Larned, K. M. Fritz, M. T. Bogan, P. J. Wood, E. I. Meyer & A. N. Santos, 2014. Broad-scale patterns of invertebrate richness and community composition in temporary rivers: effects of flow intermittence. Ecography 37: 94–104. https://doi.org/10.1111/j.1600-0587.2013.00287.x.
Datry, T., A. Boulton, N. Bonada, K. Fritz, C. Leigh, S. Eric, K. Tockner, B. Hugueny & C. Dahm, 2018. Flow intermittence and ecosystem services in rivers of the Anthropocene. Journal of Applied Ecology. https://doi.org/10.1111/1365-2664.12941.
De Cáceres, M. & P. Legendre, 2009. Associations between species and groups of sites: Indices and statistical inference. Ecology 90: 3566–3574. https://doi.org/10.1890/08-1823.1.
Döll, P. & H. M. Schmied, 2012. How is the impact of climate change on river flow regimes related to the impact on mean annual runoff? A global-scale analysis. Environmental Research Letters 7: 014037. https://doi.org/10.1088/1748-9326/7/1/014037.
Dufrêne, M. & P. Legendre, 1997. Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecological Monographs 67: 345–366. https://doi.org/10.1890/0012-9615(1997)067[0345:SAAIST]2.0.CO;2.
Garrett, R., A. Nar, T. Fisher & K. Maurer, 2018. ggvoronoi: Voronoi diagrams and heatmaps with ggplot2. Journal of Open Source Software 3: 1096. https://doi.org/10.21105/joss.01096.
Gill, B., A. Sanabria, M. Gonzales, S. Carlson & M. Bogan, 2022. Effects of drying and orientation to perennial refuges on aquatic biodiversity across two basins differing in aridity. Ecosphere. https://doi.org/10.1002/ecs2.4071.
Hill, M., K. Mathers, S. Little, T. Worrall, J. Gunn & P. Wood, 2019. Ecological effects of a supra-seasonal drought on macroinvertebrate communities differ between near-perennial and ephemeral river reaches. Aquatic Sciences. https://doi.org/10.1007/s00027-019-0659-7.
Hille, S., E. Kristensen, D. Graeber, T. Riis, N. Jørgensen & A. Baattrup-Pedersen, 2014. Fast reaction of macroinvertebrate communities to stagnation and drought in streams with contrasting nutrient availability. Freshwater Science 33: 847–859. https://doi.org/10.1086/677554.
IEA 2021. Energy Technology RD&D Budgets: Analysis. https://www.iea.org/articles/hungary-climate-resilience-policy-indicator. Accessed 25 Feb 2024
IPCC - Intergovernmental Panel on Climate Change, 2021. Climate Change 2021 – The Physical Science Basis: Working Group I Contribution to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press; 2023. https://doi.org/10.1017/9781009157896
Ionita, M., D. E. Caldarescu & V. Nagavciuc, 2021. Compound hot and dry events in Europe: variability and large-scale drivers. Frontiers in Climate 3: 688991. https://doi.org/10.3389/fclim.2021.688991.
Laini, A., G. Burgazzi, R. Chadd, J. England, I. Tziortzis, M. Ventrucci, P. Vezza, P. Wood, P. Viaroli & S. Guareschi, 2022. Using invertebrate functional traits to improve flow variability assessment within European rivers. Science of the Total Environment 832: 155047. https://doi.org/10.1016/j.scitotenv.2022.155047.
Leigh, C. & T. Datry, 2017. Drying as a primary hydrological determinant of biodiversity in river systems: a broad-scale analysis. Ecography. https://doi.org/10.1111/ecog.02230.
Lowe, W. H. & G. E. Likens, 2005. Moving headwater streams to the head of the class. BioScience 55: 196–197. https://doi.org/10.1641/0006-3568(2005)055[0196:MHSTTH]2.0.CO;2.
Lucas-Picher, P., D. Argueso, E. Brisson, Y. Tramblay, P. Berg, A. Lemonsu, S. Kotlarski, C. Caillaud, N. Poncet, V. E. Cortes Hernandez & Y. Michau, 2022. Convection-permitting modeling with regional climate models: latest developments and next steps. In AGU Fall Meeting Abstracts (Vol. 2022, pp. A42M-01).
Messager, M., B. Lehner, C. Cockburn, N. Lamouroux, H. Pella, T. Snelder, K. Tockner, T. Trautmann, C. Watt & T. Datry, 2021. Global prevalence of non-perennial rivers and streams. Nature 594: 391–397. https://doi.org/10.1038/s41586-021-03565-5.
Mezősi, G., 2017. Physical geography of Hungary. Magyarország Természetföldrajza. https://doi.org/10.1007/978-3-319-45183-1.
OMSZ 2024. Országos Meteorológiai Szolgálat http://omsz.hu [National Meteorological Service]. http://omsz.hu
Palmer, M., C. Reidy Liermann, C. Nilsson, M. Flörke, J. Alcamo & N. Bond, 2008. Climate change and the world’s river basins: anticipating management options. Frontiers in Ecology and the Environment. https://doi.org/10.1890/060148.
Pařil, P., M. Polášek, B. Loskotová, M. Straka, J. Crabot & T. Datry, 2019. An unexpected source of invertebrate community recovery in intermittent streams from a humid continental climate. Freshwater Biology 64(11): 1971–1983. https://doi.org/10.1111/fwb.13386.
Pimm, S. L., H. L. Jones & J. Diamond, 1988. On the Risk of Extinction. The American Naturalist, Vol. 132. The University of Chicago Press, Chicago:, 757–785. https://doi.org/10.1086/284889.
Podani, J., T. Kalapos, B. Barta & D. Schmera, 2021. Principal component analysis of incomplete data – a simple solution to an old problem. Ecological Informatics 61: 101235. https://doi.org/10.1016/j.ecoinf.2021.101235.
Řezníčková, P., P. Pařil & S. Zahrádková, 2007. The ecological effect of drought on the macroinvertebrate fauna of a small intermittent stream–an example from the Czech Republic. International Review of Hydrobiology 92: 514–526. https://doi.org/10.1002/iroh.200610997.
Robinson, C. & T. Buser, 2007. Density-dependent life history differences in a stream mayfly (Deleatidium) inhabiting permanent and intermittent stream reaches. New Zealand Journal of Marine and Freshwater Research 41: 265–271. https://doi.org/10.1080/00288330709509914.
Rolls, R. J., J. Heino & B. Chessman, 2016. Unravelling the joint effects of flow regime, climatic variability and dispersal mode on beta diversity of riverine communities. Freshwater Biology 61: 1350–1364. https://doi.org/10.1111/fwb.12793.
Rubel, F. & M. Kottek, 2010. Observed and projected climate shifts 1901–2100 depicted by world maps of the Köppen-Geiger climate classification. Meteorologische Zeitschrift 19: 135–141. https://doi.org/10.1127/0941-2948/2010/0430.
Sarremejane, R., H. Mykrä, K.-L. Huttunen, K.-R. Mustonen, H. Marttila, R. Paavola, K. Sippel, N. Veijalainen & T. Muotka, 2018. Climate-driven hydrological variability determines inter-annual changes in stream invertebrate community assembly. Oikos 127: 1586–1595. https://doi.org/10.1111/oik.05329.
Sarremejane, R., N. Cid, R. Stubbington, T. Datry, M. Alp, M. Cañedo-Argüelles, A. Cordero-Rivera, Z. Csabai, C. Gutiérrez-Cánovas, J. Heino, M. Forcellini, A. Millán, A. Paillex, P. Pařil, M. Polášek, J. M. Tierno de Figueroa, P. Usseglio-Polatera, C. Zamora-Muñoz & N. Bonada, 2020a. DISPERSE, a trait database to assess the dispersal potential of European aquatic macroinvertebrates. Scientific Data Nature Publishing Group 7: 386. https://doi.org/10.1038/s41597-020-00732-7.
Sarremejane, R., J. England, C. E. M. Sefton, S. Parry, M. Eastman & R. Stubbington, 2020b. Local and regional drivers influence how aquatic community diversity, resistance and resilience vary in response to drying. Oikos 129: 1877–1890. https://doi.org/10.1111/oik.07645.
Schmera, D., J. Heino & J. Podani, 2022. Characterising functional strategies and trait space of freshwater macroinvertebrates. Scientific Reports Nature Publishing Group 12: 12283. https://doi.org/10.1038/s41598-022-16472-0.
Schmidt-Kloiber, A. & D. Hering, 2015. www.freshwaterecology.info – An online tool that unifies, standardises and codifies more than 20,000 European freshwater organisms and their ecological preferences. Ecological Indicators 53: 271–282. https://doi.org/10.1016/j.ecolind.2015.02.007.
Schneider, B., M. G. Ehrhart & W. H. Macey, 2013. Organizational climate and culture. Annual Review of Psychology 64: 361–388. https://doi.org/10.1146/annurev-psych-113011-143809.
Soria, M., C. Leigh, T. Datry, L. M. Bini & N. Bonada, 2017. Biodiversity in perennial and intermittent rivers: a meta-analysis. Oikos 126: 1078–1089. https://doi.org/10.1111/oik.04118.
Southwood, T. R. E., 1977. Habitat, the templet for ecological strategies? Journal of Animal Ecology 46: 337–365. https://doi.org/10.2307/3817.
Strachan, S., E. Chester & B. Robson, 2015. Freshwater invertebrate life history strategies for surviving desiccation. Springer Science Reviews 3: 57–75. https://doi.org/10.1007/s40362-015-0031-9.
Stubbington, R., 2012. The hyporheic zone as an invertebrate refuge: a review of variability in space, time, taxa and behaviour. Marine and Freshwater Research 63: 293–311. https://doi.org/10.1071/MF11196.
Stubbington, R. & T. Datry, 2013. The macroinvertebrate seedbank promotes community persistence in temporary rivers across climate zones. Freshwater Biology. https://doi.org/10.1111/fwb.12121.
Stubbington, R., M. T. Bogan, N. Bonada, A. J. Boulton, T. Datry, C. Leigh & R. VanderVorste, 2017. Chapter 4.3—the biota of intermittent rivers and ephemeral streams: aquatic invertebrates. In Datry, T., N. Bonada & A. Boulton (eds), Intermittent Rivers and Ephemeral Streams Academic Press, New York: 217–243. https://doi.org/10.1016/B978-0-12-803835-2.00007-3.
Stubbington, R., R. Sarremejane & T. Datry, 2019. Alpha and beta diversity of connected benthic–subsurface invertebrate communities respond to drying in dynamic river ecosystems. Ecography. https://doi.org/10.1111/ecog.04592.
Townsend, C. R. & A. G. Hildrew, 1994. Species traits in relation to a habitat templet for river systems. Freshwater Biology 31: 265–275. https://doi.org/10.1111/j.1365-2427.1994.tb01740.x.
Vander Vorste, R., R. Stubbington, V. Acuña, M. Bogan, N. Bonada, N. Cid, T. Datry, R. Storey, P. Wood & A. Ruhi, 2021. Climatic aridity increases temporal nestedness of invertebrate communities in naturally drying rivers. Ecography. https://doi.org/10.1111/ecog.05349.
Vanneuville, W., B. Werner, T. Kjeldsen, J. Miller, M. Kossida, A. Tekidou, A. Kakava & P. Crouzet, 2012. Water resources in Europe in the context of vulnerability: EEA 2012 state of water assessment. European Environment Agency. Copenhagen. http://www.eea.europa.eu/publications/water-resources-and-vulnerability/at_download/file.
White, J., A. House, N. Punchard, D. Hannah, N. Wilding & P. Wood, 2018. Macroinvertebrate community responses to hydrological controls and groundwater abstraction effects across intermittent and perennial headwater streams. Science of the Total Environment 610: 1514–1526. https://doi.org/10.1016/j.scitotenv.2017.06.081.
Zipper, S., J. Hammond, M. Shanafield, M. Zimmer, T. Datry, C. Jones, K. Kaiser, S. Godsey, R. Burrows, J. Blaszczak, M. Busch, A. Price, K. Boersma, A. Ward, K. Costigan, G. Allen, C. Krabbenhoft, W. Dodds, M. Mims & D. Allen, 2021. Pervasive changes in stream intermittency across the United States. Environmental Research Letters 16: 084033. https://doi.org/10.1088/1748-9326/ac14ec.
Acknowledgements
We extend our grateful thanks to Balázs J. Berta, Bernadett Boóz, Márk Ficsór, Arnold Móra, Bálint Pernecker, and Khouloud Sebteoui (University of Pécs), Csaba Békési, Gabriella Bodnár, Péter Borza, Judit Fekete, Szabolcs Mecsei, József Szekeres, and Júlia Szeles (Centre for Ecological Research) Csaba Deák and Kristóf Málnás for help in sample collection, taxa identification, and data curation. The authors thank Rachel Stubbington for her many insightful comments and suggestions on a previous version of the manuscript. The authors thank the anonymous reviewers for their constructive comments and suggestions.
Funding
Open access funding provided by HUN-REN Centre for Ecological Research. The project is exclusively supported by the National Research, Development and Innovation Office – NKFIH FK 135 136 grant (PB). PB is supported by the János Bolyai Research Scholarship of the Hungarian Academy of Sciences BO-00106-21 and by the ÚNKP-23-5 New National Excellence Program of the Ministry for Innovation and Technology from the source of the National Research, Development and Innovation Fund. DS was supported by the National Multidisciplinary Laboratory for Climate Change (RRF-2.3.1-21-2022-00014) and by the Sustainable Development and Technologies National Programme of the Hungarian Academy of Sciences (FFT NP FTA). BT is supported by ÚNKP-22-3-II New National Excellence Program of the Ministry for Innovation and Technology from the source of the National Research, Development and Innovation Fund.
Author information
Authors and Affiliations
Contributions
TB contributed towards conceptualization, investigation, data curation, formal analysis, methodology, visualization and writing—original draft. GV contributed towards data curation, formal analysis and writing—review and editing. ZC contributed towards conceptualization, investigation, methodology and writing—review and editing. DS contributed towards methodology, formal analysis and writing—review and editing. PB contributed towards conceptualization, investigation, methodology, writing—original draft, writing—review and editing, supervision and funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethical approval
Not applicable.
Additional information
Handling editor: Sally A. Entrekin
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bozóki, T., Várbíró, G., Csabai, Z. et al. Resistance not resilience traits structure macroinvertebrate communities in newly drying stream sections. Hydrobiologia (2024). https://doi.org/10.1007/s10750-024-05518-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10750-024-05518-1