Abstract
The high-elevation plateaus of the inter-Andean valleys are home to shallow lakes that have become eutrophic. These lakes share similarities with shallow lakes in temperate and subtropical areas. Because native species diversity is low, invasive species dominate the fish and macrophytes communities. The study aimed to investigate the behavioral response of the local Daphnia pulex from the Andean shallow Lake Yahuarcocha to the exotic submerged macrophyte Egeria densa and the exotic fish Poecilia reticulata. Laboratory habitat choice experiments revealed that D. pulex from Lake Yahuarcocha strongly avoid E. densa, irrespective of the presence of the fish P. reticulata or chemical cues indicating fish predation on D. pulex. This observation could be explained by the fact that P. reticulata displayed a strong attraction to E. densa during the daytime, probably to avoid bird predation. D. pulex from the nearby Lake San Pablo where P. reticulata is absent but where the fish community is dominated by Oncorhynchus mykiss displayed the same avoidance behavior to submerged macrophytes as D. pulex from Lake Yahuarcocha. These results indicate that macrophytes in these high-elevation shallow lakes may not facilitate top-down control of phytoplankton, since plants do not offer refuge to D. pulex from fish predation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Trophic interactions in shallow lakes have been described as one of the main forces driving the functioning of these ecosystems (Timms & Moss, 1984; Jeppesen et al., 1998; Burks et al., 2002). Cascading effects involving interactions between piscivorous and planktivorous fishes on large zooplankton can impact the development of algal blooms (Schriver et al., 1995; William & Moss, 2003; Meerhoff et al., 2007a). In this context, the presence of macrophytes can play a fundamental role in mediating interactions between fish and zooplankton, in addition to competing for nutrients with phytoplankton, favoring clear water states (Scheffer et al., 1993; Lauridsen et al., 1996; Søndergaard & Moss, 1998; Jeppesen et al., 2012).
In shallow lakes in temperate regions, experimental studies have demonstrated that submerged macrophytes provide refuge to zooplankton from fish predation (Schriver et al., 1995; Jacobsen & Perrow, 1998; Burks et al., 2002, 2006). Because Daphnia consume a wide size range of phytoplankton, high Daphnia densities can reduce phytoplankton blooms through grazing (Meijer et al., 1999; Perrow et al., 1999; Zhang et al., 2009). However, Daphnia is very sensitive to predation by visual predators such as planktivorous fish (Jeppesen et al., 2004; Lacerot et al., 2013). Therefore, top-down control of phytoplankton by Daphnia can only be achieved when water fleas can avoid predation by planktivorous fish (Jeppesen et al., 1997; Walsh et al., 2012). In nutrient-rich shallow lakes, submerged macrophyte beds offer Daphnia a refuge against fish predation (Lauridsen et al., 1996; Jeppesen et al., 1998; Meerhoff et al., 2003). This is the case in temperate shallow lakes, where planktivorous fish tend to avoid the submerged macrophyte vegetation, and, hence, the predation risk to Daphnia is higher outside the macrophyte than inside the vegetation (Burks et al., 2002). However, in shallow lakes at low land in tropical and subtropical areas, the fish community is often dominated by small fish that aggregate in the macrophytes beds rather than in the open water (Pelicice et al., 2005; Texeira de Mello et al., 2009; 2016). As a result, Daphnia tend to avoid macrophytes even when fish are present in such lakes (Meerhoff et al., 2006; Iglesias et al., 2007).
Shallow lakes located at high elevations in the Andean plateaus share similarities with tropical, subtropical, and temperate zones. They have a relatively low water temperature, comparable to temperate shallow lakes, and similar to shallow lakes in the tropics, they lack a pronounced seasonality (e.g., Gunkel & Casallas, 2002; Van Colen et al., 2017). It is unclear to what extent these shallow lakes in the tropical Andes are more similar to their counterparts in their functioning in tropical and subtropical or temperate areas. It has become clear that shallow lakes located in lowland tropical-subtropical areas function differently compared to temperate shallow lakes, and these differences have important implications for managing eutrophication (Jeppesen et al., 2005; Meerhoff et al., 2006; Kosten et al., 2009).
Whether Daphnia can use submerged macrophytes as a refuge from fish predation will depend on the fish communities in these lakes. Given that lakes at high elevations are isolated and have low biodiversity, fish communities are composed of a limited number of native species (e.g., Astroblepus spp., Eremophilus mutisii Humboldt, 1805) (Anderson & Maldonado-Ocampo, 2011; Jacobsen & Dangles, 2017). The low diversity of native species has made these lakes vulnerable to invasion by exotic fish species such as trout (Oncorhynchus mykiss Walbaum, 1792), guppy (Poecilia reticulata Peters, 1859), Nile tilapia (Oreochromis niloticus Linnaeus, 1758), and carp (Cyprinus carpio Linnaeus, 1758) (Anderson & Maldonado-Ocampo, 2011; Van Colen et al., 2017). Some of the most common effects of exotic fish species include declines in populations of native species (fish and invertebrates) through predation. Likewise, the introduction of rainbow trout (O. mykiss), sea trout (Salmo trutta Linnaeus, 1758), and the silverside (Basilichthys bonariensis Valenciennes, 1835) into Lake Titicaca in the Peru-Bolivia border caused the extinction of the endemic fish Orestias cuvieri (Valenciennes, 1846), and has caused drastic decline of at least three other species in the same genus (Villwock, 1994). Similarly, the Andean catfish population decreased drastically after exotic fish were introduced into San Pablo Lake (Vélez-Espino, 2004; Vimos et al., 2015). Introducing exotic species in freshwater habitats can profoundly change food web structure (Lodge et al., 1998; Deacon et al., 2011; Villéger et al., 2011). It is unknown, however, how these exotic species affect Daphnia behavior and the ecological functioning of high-elevation shallow lakes in the Andes.
Tropical lakes are vital freshwater reservoirs in the high plateaus of Andean regions. Despite their ecological and social relevance, we know little about these water bodies, especially the factors influencing their ecosystem functioning. This study focuses on a eutrophic shallow Lake Yahuarcocha, typical of high elevation with low species diversity in northern Ecuador. The lake suffers from a year-round cyanobacterial (e.g., Cylindrospermopsis, Planktothrix) bloom (Van Colen et al., 2017; Portilla et al., 2022). Guppy (P. reticulata) is an ornamental exotic fish that dominates the fish community in the lake. This species is known to exert intense predation pressure on Daphnia (Brewer et al., 1999; Lawal et al., 2012). No piscivorous fish species are known to occur in Lake Yahuarcocha; however, the generalist and opportunistic omnivorous fish O. niloticus and C. carpio are present. Submerged macrophytes (Egeria densa Planch.) were once common in the lake but are now restricted to a narrow area in the littoral zone because of the low water transparency.
As P. reticulata is a lowland subtropical species, we might expect local Daphnia pulex (Leydig, 1860) to behave similarly to Daphnia from shallow lakes located in tropical-subtropical areas, i.e., avoidance of macrophytes because fish forages in the macrophytes vegetation. On the other hand, P. reticulata behavior may differ from lakes with piscivorous fish due to the lack of predatory fish species in Yahuarcocha Lake. This study aimed to investigate the behavioral responses of D. pulex to the presence of P. reticulata and this interaction mediated by the exotic aquatic plants E. densa. Using laboratory habitat choice experiments, we determined the effect of exotic fish P. reticulata and the submerged macrophyte E. densa on the horizontal movement by D. pulex on the presence and absence of alarm signals (fish kairomone and crushed D. pulex). The behavior of D. pulex may strongly differ between populations due to different local evolutionary history (De Meester, 1996). Therefore, we also investigated a second D. pulex population from a nearby lake (San Pablo) where P. reticulata is absent and O. mykiss dominates the fish community to evaluate whether D. pulex from a nearby population would behave differently if it would be introduced in Lake Yahuarcocha. Based on previously published studies of shallow lakes in tropical and subtropical systems (Meerhoff et al., 2006; Iglesias et al., 2007; Meerhoff et al., 2007a, b) and shallow lakes in Mediterranean areas (Tavşanoğlu et al., 2012), we hypothesized that D. pulex from shallow lakes in the Andes would avoid submerged plants in the presence and absence of predators and predator cues.
Materials and methods
Study site
Lake Yahuarcocha is a large (2.6 km2) and relatively shallow lake (average depth 4.9 m) situated at an elevation of 2192 m in the inter-Andean valley in the north of Ecuador, near the city of Ibarra (0° 22′ N, 78° 06′ W). The lake is a popular destination for local tourists. Discharge of untreated sewage from restaurants and the local community has resulted in strong eutrophication of the lake. In 2017, chlorophyll a concentration was 150 µg L−1 and the Secchi depth was 0.30 m (Van Colen et al., 2017). The fish community consists of invasive species. The dominant species is the omnivorous guppy (P. reticulata). Other fish species are the larger Nile tilapia (O. niloticus) and carp (C. carpio). The submerged macrophytes community was dominated by Brazilian waterweed (E. densa), with other species such as Elodea canadensis Michx., Potamogeton pusillus L., and Myriophyllum aquaticum Vell. Submerged macrophytes covered about 4% of the lake area in earlier decades, but this was reduced to only 1.4% (Van Colen et al., 2017). Rotifers and cyclopoid copepods mainly represent the zooplankton community in the lake (rotifers:0.09 ± 0.01 mm; n = 30 Acanthocyclops sp.; 0.5 ± 0.16 mm; n = 30). D. pulex occur in the lake but its abundance was low (< 3 ind L−1), and individuals were small in size (1.2 ± 0.2 mm; n = 30).
We compared the behavioral response of D. pulex from Lake Yahuarcocha with D. pulex from nearby Lake San Pablo which has a different fish community. San Pablo is a large (17 km2) and deep lake (average depth 24.6 m) situated about 22 km from Yahuarcocha Lake, near the city of Otavalo (0° 12′ N, 78° 13′ W). This lake is situated at a slightly higher elevation (2660 m) and is slightly cooler than Lake Yahuarcocha (surface water temperature about 19 °C in 2018). Because of this lower temperature, the tropical fish P. reticulata does not occur in the lake, and the fish community is dominated by introduced trout species Micropterus salmoides (Lacepède, 1802) and O. mykiss. The lake was considered eutrophic with a chlorophyll a concentration of 5.5 µg L−1 and a Secchi depth of 3 m (Gunkel, 2000; Gunkel & Casallas, 2002). Submerged macrophytes occur in the littoral zone, with the dominant species being the invasive species Ceratophyllum demersum L., E. densa, Potamogeton striatus Ruiz & Pav., and Potamogeton illinoensis Morong. The zooplankton community is dominated by rotifers and cyclopoid copepods (Metacyclops sp.). The abundance of D. pulex at San Pablo Lake is around 5 individuals L−1, which is higher than D. pulex at Yahuarcocha Lake, and individuals were larger than in Lake Yahuarcocha (2.8 ± 0.1 mm; n = 30).
Pigmentation has been shown as a mechanism for protecting zooplankton from UV light (Hessen et al., 1999; Hansson et al., 2007). Although D. pulex live at relatively high altitudes in lakes Yahuarcocha and San Pablo, both are relatively turbid which significantly reduces the penetration of UV radiation in the water column (Laurion et al., 2000; Hayakawa & Sugiyama, 2008; Wang et al., 2020) and thus the melanin content in zooplankton (Côte et al., 2019). D. pulex individuals used in our experiments did not have any visible melanin content.
General experimental setup
We used habitat choice experiments to study the interactions between D. pulex and the submerged macrophyte E. densa in the presence and absence of cues for fish predation, and the response of P. reticulata to the macrophytes. We applied a modified methodology based on the experiments from Burks et al., (2001) and Meerhoff et al., (2003; 2006). All experiments were carried out between March and June 2018. Each experimental treatment was replicated five times. D. pulex were collected from lakes Yahuarcocha and San Pablo using a Schindler-Patalas trap (mesh size 64 µm). Live D. pulex were kept in aquaria filled with dechlorinated tap water (aerated for 5 days) and fed with dried yeast (50 µg L−1) for 2–3 days before the experiment. Only adult D. pulex were used in the experiments (> 1 mm for Lake Yahuarcocha, and > 2 mm for Lake San Pablo). Plants of E. densa were collected from the littoral zone of Lake San Pablo. The plants were thoroughly rinsed to remove macroinvertebrates. They were maintained in aquaria filled with dechlorinated tap water for up to 5 days before the experiment. The fish P. reticulata were collected from Lake Yahuarcocha using a kick-net (mesh size 2 mm). The fish were kept for up to 5 days and fed on commercial food for ornamental fish (containing fish and shrimp meal).
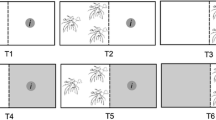
Laboratory habitat choice experiments with D. pulex were carried out in small aquaria of 15 L (50 × 10 cm and 30 cm high). The aquaria were filled with 12 L of dechlorinated tap water to avoid the presence of chemical cues derived from fish. The aquaria were positioned in a temperature-controlled room (20 °C) with uniform artificial lighting from above (12 µEinst m−2 s−1). A total of 8 treatments were carried out, 6 treatments were conducted for D. pulex behavior and 2 for fish behavior (Table1; Fig. 1, Supplementary Fig. 1): E. densa and artificial plants covering one side of the aquarium, 40 crushed D. pulex (alarm signal), and fish kairomone (10 adult fish for one full day in the aquarium); fish on the two feeding regimes (commercial fish food and D. pulex); for the fish behavior experiments, we used E. densa and artificial plants covering one side of the aquarium.
Schematic setup of the experiments evaluating the response of D. pulex to natural and artificial E. densa in different conditions (light and dark; crushed D. pulex and fish kairomone; fish fed on D. pulex and fish fed on commercial food), and the response of fish P. reticulate to natural and artificial plants in light and dark conditions
D. pulex response to submerged macrophytes natural and plastic exploratory experiments had shown that the distribution of D. pulex in the aquaria was stable after 15–30 min (see Supplementary Fig. 2). We also tested exploratory experimental behavior with another macrophyte M. aquaticum which was present in both lakes San Pablo and Yahuarcocha and we saw the same avoidance behavior by D. pulex (Supplementary Fig. 3).
Interaction between Daphnia pulex and submerged macrophytes (natural and artificial) in light and dark conditions
The first series of experiments explored the response of D. pulex from lakes Yahuarcocha and San Pablo to E. densa in the absence of fish cues and aimed to evaluate whether D. pulex detect submerged macrophytes using visual or chemical cues (Table.1, set a). The aquaria were divided in two; one side contained macrophytes (natural or artificial) and the side free of plants was considered as control with the treatments being randomly alternated between sides in the different replicates (Fig. 2). A total of 30 D. pulex individuals were added to the middle of the aquarium, and the position of D. pulex on both sides of the aquarium was recorded at 00:15, 00:30, 01:00, 01:30, 02:00,03:00,04:00, 05:00, and 06:00 h; nevertheless, we only use the information after 2 h. Treatments consisted of either real or artificial E. densa and experiments were carried out in light and in dark to test the first 2 hypotheses (Table.1, set a). Plastic macrophytes were morphologically highly similar to the natural macrophytes. The plastic macrophytes were rinsed and soaked in dechlorinated tap water for 5 days prior to the experiments to minimize the leaching of chemicals from the plastic. For both real and artificial macrophyte treatments, between 15 and 18 stems were bound together with parafilm with some enclosed stones to keep macrophytes from floating. This bunch of macrophytes was positioned on one side of the aquarium and occupied about one third of the total volume of the aquarium. Dark conditions were created by covering the aquarium with thick black cloth. The distribution of the D. pulex in the aquarium was recorded within less than 1 min after removing the dark cover. Furthermore, the distribution of D. pulex with either natural or plastic macrophytes on one side of the aquarium was compared to the distribution of Daphnia without macrophytes (Fig. 2).
Set up of the experiments evaluating the response of D. pulex to natural and artificial E. densa, both in light and dark conditions: three experimental setups were established and compared in light and dark conditions: 3 aquaria clear water (control treatment), 3 aquaria E. densa on one side, 3 aquaria artificial plants on one side, 2 (light and dark), 2 D. pulex populations (Yahuarcocha and San Pablo), replicated 5 times (180 experimental units in total)
Interaction between Daphnia pulex and submerged macrophytes (natural and artificial) under different chemical cues
D. pulex may sense the presence of fish predators by detecting chemicals derived from fish or chemicals derived from dead conspecifics (Stabell et al., 2003; Laforsch et al., 2006). The second series of experiments aimed to evaluate whether D. pulex altered its response to the submerged macrophytes E. densa in the presence of chemical cues that might signal fish predation (Fig. 3). These trials responded to the 3–4 hypothesis in Table 1 (set b). In these experiments, real E. densa and artificial plants were used as described in the set of experiments. For the first treatment, 10 fish were introduced into the aquarium for a full day before the experiments to ensure the presence of fish chemicals in the water. For the second treatment, crushed D. pulex was added to the water as chemicals derived from dead conspecifics are known as a signal for predation (Pijanowska, 1997).
Set up of the experiments evaluating the response of D. pulex to natural and artificial E. densa, both in crushed D. pulex and fish kairomone: three experimental units were established: 3 aquaria water (Ctrl), 3 aquaria E. densa, 3 aquaria artificial plants, 2 (crushed D. pulex and fish kairomone), 2 D. pulex populations (Yahuarcocha and San Pablo), replicated 5 times (180 experimental units)
Interaction between Daphnia pulex and submerged macrophytes in fish feeding conditions
In the third set experiment, D. pulex were offered the choice between E. densa on one side and live P. reticulata on the opposite side, responding to hypotheses 5–6 (Table 1, set c). Three adult live fish were suspended in the aquaria in a plastic enclosure full of small holes (Fig. 4). Preliminary experiments had shown that D. pulex did not respond to the presence of the plastic enclosure. To evaluate the influence of fish and E. densa on D. pulex horizontal movements, two conditions of fish feeding regimes were established, the first using fish that had been fed on D. pulex and the second using fish fed on commercial food for five days before the experiment (Fig. 4). Subsequently, the response of 30 D. pulex to the presence of fish was tested in the presence and absence of E. densa. The control treatment consisted of the aquarium without plants and fish (Fig. 4).
Set up of the experiments evaluating the response of D. pulex to E. densa and fish, both fish fed on D. pulex and commercial food conditions: 3 aquaria water, 3 aquaria Fish, 3 aquaria E. densa + fish, 2 (fish on D. pulex, fish fed on commercial food), 2 D. pulex populations (Yahuarcocha and San Pablo), replicated 5 times for Yahuarcocha and 3 times for San Pablo = 90 Replicates for Yahuarcocha and 54 replicates for San Pablo
Response of Poecilia reticulata to submerged macrophytes (natural and artificial) in light and dark conditions
Habitat choice experiments with the fish P. reticulata were carried out in aquaria of 24 L (40 × 20 cm and 30 cm high). The setup was the same as for the first series of experiments with D. pulex (Fig. 5) with the only difference being that a larger number of plant stems were needed to occupy about one third of the total volume of the aquarium. A total number of 10 adult fish were introduced to the middle of the aquarium and their distribution between the left and right side of the aquarium was determined after 6 h in light and in the dark. The experiments aimed to study the response of the fish P. reticulata to the submerged macrophyte E. densa, and to assess whether the fish rely on visual or chemical cues to detect the macrophytes, responding to hypothesis 7–8 (Table 1, set 8). Treatments consisted of either natural or artificial E. densa and experiments were carried out in light and dark conditions (Fig. 5). The distribution of P. reticulata in aquaria with either the real or artificial macrophytes on one side of the aquarium was compared to their distribution in aquaria free of macrophytes.
Statistical analysis
For D. pulex habitat choice experiments, two-way analysis of variance (two-way ANOVA) was used to test for the independent and interacting effects of D. pulex population origin and the treatments on the distribution of D. pulex in the aquarium. The results of the D. pulex behavior series were analyzed in two-way ANOVAs. For the P. reticulata habitat choice experiment, one-way ANOVA was used to test the effect of the treatments on the distribution of the fish in the aquarium. Tukey’s post hoc test was used for multiple pairwise comparisons between treatments. To meet ANOVA premises, data were transformed (sqrt (x + 1). In each experiment, we compared the distribution of D. pulex or fish in the experimental treatments with the distribution in an aquarium lacking any objects. All statistical analyses were performed using R version 3.5.2 (The R Foundation for Statistical Computing, Austria) with a significance level of 5% (P < 0.05) (RCore Team 2019).
Results
Interaction between Daphnia pulex and submerged macrophytes (natural and artificial plants) in light and dark conditions
The first series of experiments explored the response of D. pulex from lakes Yahuarcocha and San Pablo to the submerged macrophyte E. densa in the absence of cues for fish predation (Fig. 6; Table 2). D. pulex avoided submerged macrophytes in light conditions and not in dark ones, supporting our first hypothesis. In light, natural and plastic plants had a significant effect on D. pulex (Two-way ANOVA F = 97.3, P < 0.001). Water fleas from Yahuarcocha Lake strongly repelled either natural macrophytes (94 ± 6%) or plastic plants (94 ± 6%). As well as, D. pulex from San Pablo Lake avoided plastic plants (83 ± 11%), and E. densa (94 ± 8%). The avoidance response of D. pulex to submerged plants in both situations (E. densa and plastic plants) was stable after 15 min until the end of the experiment (2 h). Nearly all water fleas were observed on the opposite side of the aquarium when E. densa or plastic plants were added to one side of the aquarium. When no macrophytes were present, D. pulex were relatively equally distributed between both sides of the aquarium irrespective of light and dark conditions. In dark conditions, D. pulex did not respond to E. densa, and artificial plants, D. pulex were randomly distributed over both sides of the aquarium. In light and dark, no significant differences were observed between D. pulex from both lakes, indicating that D. pulex from Yahuarcocha Lake responded similarly to the presence of macrophytes than D. pulex from San Pablo Lake (Table 2).
Response of D. pulex from lakes Yahuarcocha and San Pablo to natural and plastic submerged macrophytes in light and dark conditions. Data show the horizontal movement behavior of water fleas, the graph itself shows the percentage of the D. pulex on one side of the aquarium (mean ± SD). The response of D. pulex to the treatments was compared to the reference situation with no macrophytes in the aquarium (‘Ctrl’ “E. densa” and “artificial plants”), the small letters over the bars indicate significant differences among the treatments according to Tukey post hoc test
Interaction between Daphnia pulex and submerged macrophytes (natural and artificial plants) under different chemical cues
The second series of experiments tested whether the response of D. pulex to the submerged macrophyte was modified in the presence of cues that might signal fish predation (Fig. 7; Table 3). Contrary to our third hypothesis, when the water in the aquaria contained chemicals excreted by the fish P. reticulata, 97% of D. pulex from Yahuarcocha and more than 90% of D. pulex from San Pablo avoided aquatic plants whether they were real or plastic (Two-way ANOVA F = 234.4, P < 0.001) (Fig. 7). In contrast to our fourth hypothesis, D. pulex also avoided the submerged macrophytes when the water contained chemicals released by dead conspecifics (crushed D. pulex) (Two-way ANOVA F = 77.02, P < 0.001). The percentage of avoidance was 95% for D. pulex from Yahuarcocha Lake and 84% for D. pulex from San Pablo Lake. In this experiment, no significant interactions were observed between the treatment (natural and plastic plants) and population effects, indicating no significant differences in the response of the two Daphnia populations (San Pablo Lake and Yahuarcocha Lake) to the submerged macrophytes. Similarly, to the previous experiment, there were no significant effects between E. densa and artificial plants, while D. pulex showed the same aversion to submerged plants.
Response of D. pulex from lakes Yahuarcocha and San Pablo to alarm signal and fish kairomone. Data show the horizontal movement behavior of water fleas from two lakes (Yahuarcocha Lake and San Pablo) avoiding submerged plants (mean ± SD). The graph itself shows the percentage of the D. pulex on one side of the aquarium. The treatments were explored through a two-way ANOVA. The response of D. pulex to the treatments was compared to the reference situation with not macrophytes in the aquarium (‘Ctrl’ “E. densa” and “artificial plants”), the small letters over the bars indicate significant differences among the treatments according to Tukey post hoc test
Interaction between Daphnia pulex and submerged macrophytes in fish feeding conditions
The fifth series of experiments aimed to investigate the response of D. pulex facing E. densa and fish from two food regimes (fish fed on D. pulex and fed on commercial food). In contrast to our fifth and sixth hypothesis, when offered the choice between E. densa and live fish suspended in the aquarium in an enclosure, D. pulex avoided macrophytes and aggregated on the side of the aquarium with the live fish (Fig. 8). Both feeding regimes had significant differences between treatments (macrophytes and fish); in the experiment with fish fed on D. pulex (Two-way ANOVA F = 192.31, P < 0.001), and in the experiment with fish fed on commercial food (Two-way ANOVA F = 106, P < 0.001). When live fish were present on one side of the aquarium in the absence of submerged macrophytes, the D. pulex were randomly distributed over the aquarium and did not seem to detect the presence of the fish in the enclosure. The presence of living fish did not significantly affect the behavior of D. pulex (Fig. 8). D. pulex from San Pablo and Yahuarcocha Lake showed the same behavioral response when facing fish, they avoided macrophytes (Table 4).
Response of D. pulex from Yahuarcocha and San Pablo Lake to different fish food regimes. Data show the horizontal movement behavior of water fleas from two lakes (Yahuarcocha Lake and San Pablo) avoiding submerged plants (mean ± SD). The graph itself shows the percentage of the D. pulex on one side of the aquarium. The treatments were explored through a two-way ANOVA. The response of D. pulex to the treatments was compared to the reference situation with not macrophytes in the aquarium (‘Ctrl’ “E. densa” and “artificial plants”), the small letters over the bars indicate significant differences among the treatments according to Tukey post hoc test
Fish response to submerged macrophytes (dark and light conditions)
The four series of experiments aimed to investigate the response of the fish P. reticulata to the submerged macrophyte E. densa (Fig. 9, Table 5). In line with our seventh and eighth hypotheses, when submerged plants were present, fish were significantly attracted to the macrophytes, whether the macrophytes were real or plastic (more than 71%) (One-way ANOVA F = 16.16, P < 0.001) in light. Moreover, in the absence of submerged macrophytes, fish were randomly distributed over the aquarium. In dark conditions, however, an opposite response was observed, and P. reticulata significantly avoided the macrophytes (less than 22% attraction) (One-way ANOVA F = 34, P < 0.001) (Fig. 9).
Effect of submerged macrophytes on the behavior of P. reticulata. Data show the horizontal movement behavior of fish from lakes Yahuarcocha attracted to submerged plants (mean ± SD). The graph itself shows the percentage of the fish on one side of the aquarium. The treatments were explored through a one-way ANOVA. The response of fish to the treatments was compared to the reference situation with no macrophytes in the aquarium (‘Ctrl’ “E. densa” and “artificial plants”), the small letters over the bars indicate significant differences among the treatments according to Tukey post hoc test
Discussion
In light conditions, our results showed that D. pulex from Lake Yahuarcocha and Lake San Pablo avoided the submerged macrophytes E. densa irrespective of the presence of cues and signal a risk of fish predation. D. pulex avoided E. densa on the basis of mechanical cues, as they displayed the same response to plastic or natural plants. Our results coincide with those from shallow lakes in subtropical systems (Meerhoff et al., 2006; Iglesias et al., 2007; Meerhoff et al., 2007a, b), shallow lakes at tropical lowlands (Dos Santos et al., 2020), and shallow lakes in Mediterranean systems (Tavşanoğlu et al., 2012), where Daphnia avoidance of macrophytes has been shown as the most common antipredator behavior during daytime. Furthermore, our findings differ from those presented by Burks et al., (2001) in shallow lakes intemperate systems, where aquatic vegetation provides refuge to zooplankton against fish predation; and from those presented by Montiel-Martínez et al., (2015) in a tropical lake Xochimilco, where cladocerans use macrophytes to reduce predation risk, this shallow system is located at a similar elevation to our lakes.
Small fish-sized composition dominates the fish population in subtropical and tropical eutrophic shallow lakes (Meerhoff et al., 2007a, b; Teixeira de Mello et al., 2009, 2016). When predators are present, prey seeks out protected habitats to avoid them, and small-sized fish tend to forage among the macrophytes as shelter from predation (Pelice et al., 2005; Lopes et al., 2015; Figueiredo et al., 2015). The avoidance of macrophytes by D. pulex may be linked to the observation that the dominant fish in Lake Yahuarcocha is P. reticulata, which is strongly attracted to the submerged macrophyte during the day and avoids plants during the night. This behavior of P. reticulata is similar to that of other small tropical and subtropical fish species that aggregate within submerged macrophytes beds (Meerhoff et al., 2003; Pelicice et al., 2005; Teixeira de Mello et al., 2009, 2016); however, these earlier studies did not account for differences in plant occupancy between light and dark conditions. Our findings indicate that D. pulex behavior in dark conditions was homogenous on both sides of the aquariums, with no attraction or repellence to submerged macrophytes.
In natural lakes or streams, P. reticulata is commonly observed hiding among macrophytes in the presence of predatory fish (Farr, 1975; Magurran, 2005). The observation that P. reticulata is attracted by macrophytes in Lake Yahuarcocha is somewhat surprising because no predatory fish species are present in the lake. The only larger fish species present in Lake Yahuarcocha are O. niloticus and C. carpio, but based on stomach content analysis and survival of P. reticulata in aquaria containing these two species, it is unlikely that these fish species feed on P. reticulata. Nevertheless, the vegetation also protects from aerial predators (Araújo et al., 2009). A sizeable egret colony is present in Lake Yahuarcocha and these birds most likely feed on P. reticulata. The fact that the fish hide between the macrophytes in the light but not in the dark is in line with predator avoidance behavior in response to visual predators (Magurran, 2005; Figueiredo et al., 2015).
Even though P. reticulata is absent in Lake San Pablo, D. pulex from this lake also avoided the submerged macrophyte E. densa in the absence and presence of cues for fish predation. The dominant fish species in this lake is O.mykiss, which was introduced for sport fishing (Gunkel, 2000; Velez-Espino, 2004). Both adults and fry of this species are known to feed on D. pulex in Andean Lakes (Aguilera et al., 2006). The lack of seasonality in this lake may allow O. mykiss to reproduce throughout the year, resulting in a year-round presence of fish fry that may forage in the submerged macrophytes vegetation. The aversion to submerged macrophytes by the D. pulex from Lake San Pablo in the presence of fish predation cues was less strong than D. pulex from Lake Yahuarcocha, probably because predation pressure between macrophyte vegetation is weaker in Lake San Pablo than in Lake Yahuarcocha. Nevertheless, D. pulex from Lake San Pablo did not use submerged plants as a predator refuge, most probably due to the occupation of macroinvertebrate predators such as the active predator of zooplankton Gammarus (Wilhelm & Schindler, 1999; Wilhelm et al., 2000), which is as abundant between submerged plants in San Pablo Lake. The deep water in the lake may provide a more secure refuge for fish predation than the littoral macrophytes vegetation, and D. pulex uses diel vertical migration to avoid predation rather than diel horizontal migration (Tavşanoğlu et al., 2012). Observations in Lake San Pablo show that a maximum abundance of D. pulex is observed just below the euphotic zone (unpublished observations).
It is well known that D. Pulex can detect and avoid submerged macrophytes (Pennak, 1973; Burks et al., 2002; Meerhoof et al., 2006). There is evidence that D. pulex may rely on visual, physical, and chemical cues to detect macrophytes (Burks et al., 2001; Meerhoof et al., 2006; Tavşanoğlu et al., 2012; Choi & Kim, 2020). D. pulex responded similarly to natural and plastic macrophytes suggesting that chemical cues from E. densa were not important. D. pulex repelled macrophytes in the light but not in the dark suggesting that D. pulex relied on visual rather than physical cues to avoid the macrophytes. Daphnia is also known to detect the presence of fish, even Daphnia that live in a fishless environment (e.g., De Meester, 1993). Daphnia relies on chemicals released by the fish (DeMeester, 1993; Laforsch et al., 2006; Detmer & Wahl, 2021) or on chemicals released by dead conspecifics consumed by fish (Stabell et al., 2003). Contrary to the findings of Detmer & Wahl, (2021) where D. pulex avoided fish fed on D. pulex water fleas, D. pulex from our experiments did not avoid fish fed on water fleas nor fish fed on commercial food, our results did not show any change in behavior avoidance of D. pulex with respect to submerged macrophytes. Therefore, this is a permanent and non-plastic characteristic of the two D. pulex populations. These findings are consistent with earlier research conducted by Meerhoff et al., (2006) in subtropical ecosystems and by Tavşanolu et al., (2007) in Mediterranean shallow lakes.
The results of our experiments indicate that D. pulex from two lakes with a different community of invasive fish species display a strong avoidance of submerged macrophytes, even in the presence of cues for fish predation. As a result, submerged macrophytes are unlikely to facilitate top-down control of phytoplankton by D. pulex, as is the case in temperate shallow lakes (Jeppesen et al., 1997; Stansfield et al., 1997; Burks et al., 2002). The behavior of D. pulex in these high-elevation lakes appears to be influenced by the community of invasive species that have been introduced into such ecosystems. Although the temperature is at the lower end of the range for tropical fish species in these lakes, the lack of a cold season likely helps these species to thrive (e.g., P. reticulata in Lake Yahuarcocha). Even when the water temperature is too low for this species to survive, as in Lake San Pablo, cold-water species such as O. mykiss possibly result in continuously high predation pressure on D. pulex because these fish can reproduce throughout the year. It is unclear to what extent these invasive fish species have modified the behavior of D. pulex as little is known about the native fish species that occurred in this lake. Early reports by von Humboldt described the occurrence of “small black fish” in these lakes, which probably correspond to the native Andean catfishes or ‘preñadilla’ (Astroblepus spp., Steinitz-Kannan et al., 1983). Unfortunately, these fish have now disappeared from these lakes and relict populations survive only in pristine streams in the region (Velez-Espino, 2004; Anderson & Maldonado-Ocampo, 2011). Because these native fish species have very different feeding ecology, being most benthic invertebrate feeders (Velez-Espino, 2004), their influences on D. pulex were probably different from the invasive species that have replaced them.
Conclusion
Shallow lakes at high altitudes in the Andes like Yahuarcocha Lake are dominated by small invasive fish species such as P. reticulata. In this respect, these lakes are similar to shallow lakes in lowland tropical and subtropical systems that are dominated by native small species such as Cnesterodon decemmaculatus (Jenyns, 1842) (Jeppesen et al., 2005; Teixeira de Mello et al., 2009). D. pulex at high altitudes in Andean shallow lakes did not find refuge in submerged macrophytes despite the presence and absence of fish and warning signs from fish during the day. This is because small fish are attracted to submerged plants during the day, probably to avoid bird predation. These results indicate that submerged macrophytes in these high-elevation shallow lakes may not facilitate top-down control of phytoplankton, given that these plants do not offer refuge to D. pulex from fish predation. It is therefore questionable whether techniques of biomanipulation applied in temperate lakes can be employed at high altitude shallow lakes in the Andes.
Data availability
All data and material generated or analyzed during this study are available from the corresponding author upon reasonable request.
References
Aguilera, X., S. Declerck, L. De Meester, M. Maldonado & F. Ollevier, 2006. Tropical high Andes lakes: a limnological survey and an assessment of exotic rainbow trout (Oncorhynchus mykiss). Limnologica 36(4): 258–268. https://doi.org/10.1016/j.limno.2006.08.001.
Anderson, E. & J. Maldonado-Ocampo, 2011. A regional perspective on the diversity and conservation of tropical Andean fishes. Conservation Biology. https://doi.org/10.1111/j.1523-1739.2010.01568.x.
Araújo, F., M. Peixoto, B. Pinto & T. Teixeira, 2009. Distribution of guppies Poecilia reticulata (Peters, 1860) and Phalloceros caudimaculatus (Hensel, 1868) along a polluted stretch of the Paraíba do Sul River, Brazil. Brazilian Journal of Biology 69: 41–48. https://doi.org/10.1590/s1519-69842009000100005.
Brewer, M., P. Dawidowicz & S. Dodson, 1999. Interactive effects of fish kairomone and light on Daphnia escape behavior. Journal of Plankton Research 2: 1317–1335. https://doi.org/10.1093/plankt/21.7.1317.
Burks, R., E. Jeppesen & D. Lodge, 2001. Littoral zone structures as Daphnia refugia against fish predators. Limnology and Oceanography 46: 230–237. https://doi.org/10.4319/lo.2001.46.2.0230.
Burks, R., D. Lodge, E. Jeppesen & T. Lauridsen, 2002. Diel horizontal migration of zooplankton: costs and benefits of inhabiting the littoral. Freshwater Biology 47: 343–365. https://doi.org/10.1046/j.1365-2427.2002.00824.x.
Burks, R., G. Mulderij, E. Gross, I. Jones, L. Jacobsen, E. Jeppesen & E. Donk, 2006. Center stage: the crucial role of macrophytes in regulating trophic interactions in shallow lake wetlands. In Wetlands: Functioning, Biodiversity Conservation, and Restoration. Springer, Berlin: 37–59. https://doi.org/10.1007/978-3-540-33189-6_3
Choi, J. & S. Kim, 2020. A study of the distribution of Daphnia obtusa and Simocephalus vetulus in response to varying environmental conditions using field and microcosm approaches. Water 12(3): 815. https://doi.org/10.3390/w12030815.
Côte, J., C. Pilisi, O. Morisseau, C. Veyssière, A. Perrault, S. Jean & L. Jacquin, 2019. Water turbidity affects melanin-based coloration in the gudgeon: a reciprocal transplant experiment. Biological Journal of the Linnean Society 128(2): 451–459. https://doi.org/10.1093/biolinnean/blz102.
De Meester, L., 1993. Genotype, fish-mediated chemical, and phototactic behavior in Daphnia Magna. Ecology 74: 1467–1474. https://doi.org/10.2307/1940075.
De Meester, L., 1996. Local genetic differentiation and adaptation in freshwater zooplankton populations: patterns and processes. Ecoscience 3: 385–399. https://doi.org/10.1080/11956860.1996.11682356.
Deacon, A., I. Ramnarine & A. Magurran, 2011. How reproductive ecology contributes to the spread of a globally invasive fish. PLoS ONE 6: e24416. https://doi.org/10.1371/journal.pone.0024416.
Detmer, T. & D. Wahl, 2021. Effects of habitat and fish type and diet on the behavior of Daphnia. Inland Waters 11(1): 57–66. https://doi.org/10.1080/20442041.2020.1766919.
Dos Santos, N., L. Stephan, A. Otero, C. Iglesias & M. Castilho-Noll, 2020. How free-floating macrophytes influence interactions between planktivorous fish and zooplankton in tropical environments? An in-lake mesocosm approach. Hydrobiologia 847: 1357–1370. https://doi.org/10.1007/s10750-020-04194-1.
Farr, J., 1975. The role of predation in the evolution of social behavior of natural populations of the guppy, Poecilia reticulata (Pisces: Poeciliidae). Evolution 29: 151–158. https://doi.org/10.1111/j.1558-5646.1975.tb00822.x.
Figueiredo, B., R. Mormul & S. Thomaz, 2015. Swimming and hiding regardless of the habitat: prey fish do not choose between a native and a non-native macrophyte species as a refuge. Hydrobiologia 746: 285–290. https://doi.org/10.1007/s10750-014-2096-x.
Gunkel, G., 2000. Limnology of an equatorial high mountain lake in Ecuador, Lago San Pablo. Limnologica 30: 113–120. https://doi.org/10.1016/s0075-9511(00)80005-5.
Gunkel, G. & J. Casallas, 2002. Limnology of an equatorial high mountain lake—Lago San Pablo, Ecuador: the significance of deep diurnal mixing for lake productivity. Limnologica 32: 33–43. https://doi.org/10.1016/s0075-9511(02)80015-9.
Hansson, L., S. Hylander & R. Sommaruga, 2007. Escape from UV threats in zooplankton: a cocktail of behavior and protective pigmentation. Ecology 88(8): 1932–1939. https://doi.org/10.1890/06-2038.1.
Hayakawa, K. & Y. Sugiyama, 2008. Spatial and seasonal variations in attenuation of solar ultraviolet radiation in Lake Biwa, Japan. Journal of Photochemistry and Photobiology B: Biology 90(2): 121–133. https://doi.org/10.1016/j.jphotobiol.2007.11.008.
Hessen, D., J. Borgeraas, K. Kessler & U. Refseth, 1999. UV-B susceptibility and photoprotection of Arctic Daphnia morphotypes. Polar Research 18(2): 345–352. https://doi.org/10.3402/polar.v18i2.6594.
Iglesias, C., G. Goyenola, N. Mazzeo, M. Meerhoff, E. Rodo, & E. Jeppesen, 2007. Horizontal dynamics of Zooplankton in subtropical Lake Bianca (Uruguay) hosting multiple Zooplankton predators and aquatic plant refuges. In Shallow Lakes in a Changing World: Proceedings of the 5th International Symposium on Shallow Lakes, Held at Dalfsen, The Netherlands, 5–9 June 2005. Springer Netherlands: 179–189. https://doi.org/10.1007/s10750-007-0599-4
Jacobsen, D. & O. Dangles, 2017. Organism and diversity patterns at high altutudes in Ecology of high altitude waters. Oxford University Press. https://doi.org/10.1093/oso/9780198736868.001.0001.
Jacobsen, L. & M. Perrow, 1998. Predation risk from piscivorous fish influencing the diel use of macrophytes by planktivorous fish in experimental ponds. Ecology of Freshwater Fish 7(2): 78–86. https://doi.org/10.1111/j.1600-0633.1998.tb00174.x.
Jeppesen, E., J. Jensen, M. Søndergaard, T. Lauridsen, L. Pedersen & L. Jensen, 1997. Top-down control in freshwater lakes: the role of nutrient state, submerged macrophytes and water depth. Hydrobiologia 342(343): 151–164. https://doi.org/10.1007/978-94-011-5648-6_17.
Jeppesen, E., M. Søndergaard, J. Jensen, E. Mortensen, A. Hansen & T. Jørgensen, 1998. Cascading trophic interactions from fish to bacteria and nutrients after reduced sewage loading: an 18-year study of a shallow hypertrophic lake. Ecosystems 1(3): 250–267. https://doi.org/10.1007/s100219900020.
Jeppesen, E., J. Jensen, M. Søndergaard, M. Fenger-Grøn, M. Bramm, K. Sandby & H. Rasmussen, 2004. Impact of fish predation on cladoceran body weight distribution and zooplankton grazing in lakes during winter. Freshwater Biology 49(4): 432–447. https://doi.org/10.1111/j.1365-2427.2004.01199.x.
Jeppesen, E., M. Søndergaard, J. Jensen, K. Havens, O. Anneville, L. Carvalho & D. Gerdeaux, 2005. Lake responses to reduced nutrient loading an analysis of contemporary long-term data from 35 case studies. Freshwater Biology 50: 1747–1771. https://doi.org/10.1111/j.1365-2427.2005.01415.x.
Jeppesen, E., M. Søndergaard, & K. Christoffersen (Eds), 2012. The Structuring Role of Submerged Macrophytes in Lakes (Vol. 131). Springer, New York. https://doi.org/10.1007/978-1-4612-0695-8
Kosten, S., G. Lacerot, E. Jeppesen, Marques D. da Motta, E. H. van Nes, N. Mazzeo & M. Scheffer, 2009. Effects of submerged vegetation on water clarity across climates. Ecosystems 12: 1117–1129. https://doi.org/10.1007/s10021-009-9277-x.
Lacerot, G., C. Kruk, M. Luerling & M. Scheffer, 2013. The role of subtropical zooplankton as grazers of phytoplankton under different predation levels. Freshwater Biology 58(3): 494–503. https://doi.org/10.1111/fwb.12075.
Laforsch, C., L. Beccara & R. Tollrian, 2006. Inducible defenses: the relevance of chemical alarm cues in Daphnia. Limnology and Oceanography 51: 1466–1472. https://doi.org/10.4319/lo.2006.51.3.1466.
Lauridsen, T., L. Pedersen, E. Jeppesen & M. Sønergaard, 1996. The importance of macrophyte bed size for cladoceran composition and horizontal migration in a shallow lake. Journal of Plankton Research 18: 2283–2294. https://doi.org/10.1093/plankt/18.12.2283.
Laurion, I., M. Ventura, J. Catalan, R. Psenner & R. Sommaruga, 2000. Attenuation of ultraviolet radiation in mountain lakes: factors controlling the among-and within-lake variability. Limnology and Oceanography 45(6): 1274–1288. https://doi.org/10.4319/lo.2000.45.6.1274.
Lawal, M., C. Edokpayi & A. Osibona, 2012. Food and feeding habits of the guppy, Poecilia reticulata, from drainage canal systems in Lagos, Southwestern Nigeria. West African Journal of Applied Ecology 20: 1–9.
Lodge, D., R. Stein, K. Brown, A. Covich, C. Brönmark, J. Garvey & S. Klosiewskt, 1998. Predicting impact of freshwater exotic species on native biodiversity: challenges in spatial scaling. Australian Journal of Ecology 23: 53–67. https://doi.org/10.1111/j.1442-9993.1998.tb00705.x.
Magurran, A., 2005. Evolutionary ecology: the Trinidadian guppy. Oxford University Press on Demand. https://doi.org/10.1093/acprof:oso/9780198527855.003.0002.
Meerhoff, M., N. Mazzeo, B. Moss & L. Rodríguez-Gallego, 2003. The structuring role of free-floating versus submerged plants in a subtropical shallow lake. Aquatic Ecology 37: 377–391. https://doi.org/10.1023/b:aeco.0000007041.57843.0b.
Meerhoff, M., C. Fosalba, C. Bruzzone, N. Mazzeo, W. Noordoven & E. Jeppesen, 2006. An experimental study of habitat choice by Daphnia: plants signal danger more than refuge in subtropical lakes. Freshwater Biology 51: 1320–1330. https://doi.org/10.1111/j.1365-2427.2006.01574.x.
Meerhoff, M., C. Iglesias, F. De Mello, J. Clemente, E. Jensen, T. Lauridsen & E. Jeppesen, 2007a. Effects of habitat complexity on community structure and predator avoidance behaviour of littoral zooplankton in temperate versus subtropical shallow lakes. Freshwater Biology 52(6): 1009–1021. https://doi.org/10.1111/j.1365-2427.2007.01748.x.
Meerhoff, M., J. Clemente, F. de Mello, C. Iglesias, A. Pedersen & E. Jeppesen, 2007b. Can warm climate-related structure of littoral predator assemblies weaken the clear water state in shallow lakes? Global Change Biology 13(9): 1888–1897. https://doi.org/10.1111/j.1365-2486.2007.01408.x.
Meijer, M., I. de Boois, M. Scheffer, R. Portielje & H. Hosper, 1999. Biomanipulation in shallow lakes in The Netherlands: an evaluation of 18 case studies. Hydrobiologia 408: 13–30. https://doi.org/10.1007/978-94-017-2986-4_2.
Montiel-Martínez, A., J. Ciros-Pérez & G. Corkidi, 2015. Littoral zooplankton–water hyacinth interactions: habitat or refuge? Hydrobiologia 755: 173–182. https://doi.org/10.1007/s10750-015-2231-3.
Pelicice, M., A. Agostinho & M. Thomaz, 2005. Fish assemblages associated with Egeria in a tropical reservoir: investigating the effects of plant biomass and diel period. Acta Oecologica 27(1): 9–16. https://doi.org/10.1016/j.actao.2004.08.004.
Pennak, R., 1973. Some evidence for aquatic macrophytes as repellents for a limnetic species of Daphnia. Internationale Revue Der Gesamten Hydrobiologie Und Hydrographie 58: 569–576.
Perrow, M., A. Jowitt, J. Stansfield & G. Phillips, 1999. The practical importance of the interactions between fish, zooplankton and macrophytes in shallow lake restoration. Hydrobiologia 395: 199–210. https://doi.org/10.1007/978-94-017-3282-6_19.
Pijanowska, J., 1997. Alarm signals in Daphnia? Oecologia 112: 12–16. https://doi.org/10.1007/s004420050277.
Portilla, K., E. Velarde, E. Decaestecker, F. Teixeira de Mello & K. Muylaert, 2022. Potential submerged macrophytes to mitigate eutrophication in a high-elevation tropical shallow lake—a Mesocosm experiment in the Andes. Water 15(1): 75. https://doi.org/10.3390/w15010075.
Scheffer, M., S. Hosper, M. Meijer, B. Moss & E. Jeppesen, 1993. Alternative equilibria in shallow lakes. Trends in Ecology & Evolution 8(8): 275–279. https://doi.org/10.1016/0169-5347(93)90254-m.
Schrive, P., J. Bøgestrand, E. Jeppesen & M. Søndergaard, 1995. Impact of submerged macrophytes on fish-zooplanl phytoplankton interactions: large-scale enclosure experiments in a shallow eutrophic lake. Freshwater Biology 33(2): 255–270. https://doi.org/10.1111/j.1365-2427.1995.tb01166.x.
Søndergaard, M., & B. Moss, 1998. Impact of submerged macrophytes on phytoplankton in shallow freshwater lakes. In The Structuring Role of Submerged Macrophytes in Lakes. Springer, New York: 115–132. https://doi.org/10.1007/978-1-4612-0695-8_6
Stabell, O., F. Ogbebo & R. Primicerio, 2003. Inducible defences in Daphnia depend on latent alarm signals from conspecific prey activated in predators. Chemical Senses 28: 141–153. https://doi.org/10.1093/chemse/28.2.141.
Stansfield, J., M. Perrow, L. Tench, A. Jowitt, & A. Taylor, 1997. Submerged macrophytes as refuges for grazing Cladocera against fish predation: observations on seasonal changes in relation to macrophyte cover and predation pressure. In Shallow Lakes’ 95. Springer, Dordrecht: 229–240. https://doi.org/10.1007/978-94-011-5648-6_25
Steinitz-Kannan, M., P. Colinvaux & R. Kannan, 1983. Limnological studies in Ecuador: 1. A survey of chemical and physical properties of Ecuadorian lakes. Arch. Hydrobiol. Suppl 65: 61–105.
Tavşanoğlu, Ü., Ç. Idil, Ş Erdoğan, M. Meerhoff, E. Jeppesen & M. Beklioglu, 2012. Sediments, not plants, offer the preferred refuge for Daphnia against fish predation in Mediterranean shallow lakes: an experimental demonstration. Freshwater Biology 57(4): 795–802. https://doi.org/10.1111/j.1365-2427.2012.02745.x.
Teixeira de Mello, F., M. Meerhoff, Z. Pekcan-Hekim & E. Jeppesen, 2009. Substantial differences in littoral fish community structure and dynamics in subtropical and temperate shallow lakes. Freshwater Biology 54: 1202–1215. https://doi.org/10.1111/j.1365-2427.2009.02167.x.
Teixeira de Mello, F., V. de Oliveira, S. Loverde-Oliveira, V. Huszar, J. Barquín, C. Iglesias & N. Mazzeo, 2016. The structuring role of free-floating plants on the fish community in a tropical shallow lake: an experimental approach with natural and artificial plants. Hydrobiologia 778(1): 167–178. https://doi.org/10.1007/s10750-015-2447-2.
Timms, R. & B. Moss, 1984. Prevention of growth of potentially dense phytoplankton populations by zooplankton grazing, in the presence of zooplanktivorous fish, in a shallow wetland ecosystem. Limnology and Oceanography 29(3): 472–486. https://doi.org/10.4319/lo.1984.29.3.0472.
Van Colen, W., K. Portilla, T. Oña, G. Wyseure, P. Goethals, E. Velarde & K. Muylaert, 2017. Limnology of the neotropical high elevation shallow lake Yahuarcocha (Ecuador) and challenges for managing eutrophication using biomanipulation. Limnologica 67: 37–44. https://doi.org/10.1016/j.limno.2017.07.008.
Velez-Espino, L., 2004. Threatened fishes of the world: Astroblepus ubidiai (Pellegrin, 1931) (Astroblepidae). Environmental Biology of Fishes 71: 296–296. https://doi.org/10.1007/s10641-004-1034-6.
Villéger, S., S. Blanchet, O. Beauchard, T. Oberdorff & S. Brosse, 2011. Homogenization patterns of the world’s freshwater fish faunas. Proceedings of the National Academy of Sciences 108: 18003–18008. https://doi.org/10.1073/pnas.1107614108.
Villwock, W., 1994. Consecuencias de la introducción de peces exóticos sobre las especies nativas del lago Titicaca. Ecología En Bolivia 23: 49–56. https://doi.org/10.17151/luaz.2013.37.15.
Vimos, D., A. Encalada, B. Ríos-Touma, E. Suárez & N. Prat, 2015. Effects of exotic trout on benthic communities in high-Andean tropical streams. Freshwater Science 34(2): 770–783. https://doi.org/10.1086/681540.
Walsh, M., J. DeLong, T. Hanley & D. Post, 2012. A cascade of evolutionary change alters consumer-resource dynamics and ecosystem function. Proceedings of the Royal Society B: Biological Sciences 279(1741): 3184–3192. https://doi.org/10.1098/rspb.2012.0496.
Wang, Q., Y. Zhang, X. Wangjin, Y. Wang, G. Meng & Y. Chen, 2020. The adsorption behavior of metals in aqueous solution by microplastics effected by UV radiation. Journal of Environmental Sciences 87: 272–280. https://doi.org/10.1016/j.jes.2019.07.006.
Wilhelm, F. & D. Schindler, 1999. Effects of Gammarus lacustris (Crustacea: Amphipoda) on plankton community structure in an alpine lake. Canadian Journal of Fisheries and Aquatic Sciences 56(8): 1401–1408. https://doi.org/10.1139/f99-067.
Wilhelm, F., D. Schindler & A. McNaught, 2000. The influence of experimental scale on estimating the predation rate of Gammarus lacustris (Crustacea: Amphipoda) on Daphnia in an alpine lake. Journal of Plankton Research 22(9): 1719–1734. https://doi.org/10.1093/plankt/22.9.1719.
Williams, A. & B. Moss, 2003. Effects of different fish species and biomass on plankton interactions in a shallow lake. Hydrobiologia 491: 331–346. https://doi.org/10.1023/a:1024456803994.
Zhang, X., T. Warming, H. Hu & K. Christoffersen, 2009. Life history responses of Daphnia magna feeding on toxic Microcystis aeruginosa alone and mixed with a mixotrophic Poterioochromonas species. Water Research 43(20): 5053–5062. https://doi.org/10.1016/j.watres.2009.08.022.
Acknowledgements
This study was funded by the VLIRUOS TEAM Project ZEIN2015PR404. We thank the students from the UTN LABINAM team for their help with the experiments: Jorge Revelo, Andrés Ipiales, Alexis Galarza, Kevin Patiño, Fernanda Benavidez, Erika Pujota, Diego Olivo, Elizbeth Astudillo, and Maria Elena Ochoa. FTM was supported by the Sistema Nacional de Investigadores (SNI), and the Programa de Desarrollo de las Ciencias Básicas (PEDECIBA Geociencias and Biología, Uruguay).
Funding
This study was funded by the VLIRUOS TEAM Project ZEIN2015PR404.
Author information
Authors and Affiliations
Contributions
KP and KM were involved in experiment design, and running experiments and they wrote the initial drafts of the manuscript and conceptualization. EV, TO, and KM contributed to resources, project administration, and funding acquisition; KP, KM, ED, and FTdeM were involved in the critical review and revision of the manuscript. KM was involved in the supervision of the whole work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
Not applicable.
Informed consent
I can confirm that the manuscript has been read and approved by all named authors and that there are no other persons who satisfied the criteria for authorship but are not listed. I further confirm that all have approved the order of authors listed in the manuscript.
Additional information
Handling Editor: Julie Coetzee
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Portilla, K., Velarde, E., Oña, T. et al. Effects of the invasive fish Poecilia reticulata on the behavioral response of Daphnia pulex to the exotic submerged macrophyte Egeria densa. Hydrobiologia 851, 1183–1201 (2024). https://doi.org/10.1007/s10750-023-05377-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-023-05377-2