Abstract
The functional group (FG) concept suggests that species having different phylogenetic origins but possessing similar functional characteristics can be considered as functional groups and these co-occur in the phytoplankton. Here, we study how functional redundancy of phytoplankton taxa (within group richness) contribute to the species diversity of assemblages in an oxbow lake in the Carpathian Basin. We found that although the observed functional redundancy was similar among several FGs, the shape of the species accumulation curves of these groups was considerably different, implying that the observed species numbers alone do not represent the real functional redundancy of the groups. We demonstrated that FGs that showed asymptotes in species richness estimates in small spatial scale, exhibited steady increase in large spatial, and temporal scales. The contribution of FGs to species richness depended strongly on the relative biomass of each FG. Species accumulation curves of those groups of which elements dominated in the phytoplankton, appeared to be approaching asymptotes. Since the shapes of species accumulation curves refer to the strengths of within-group competition among constituent species, our results imply that functional redundancy of phytoplankton is influenced by the role that the elements play within the assemblages.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The high diversity of phytoplankton is one of the most remarkable features of aquatic systems and became the focus of many studies, especially after Hutchinson (1961) in his seminal paper asked: “How is it possible for a number of species to coexist in a relatively isotropic or unstructured environment, all competing for the same sorts of materials?“. Several theories have been proposed to explain this “paradox of the plankton”, and now we know that the environment in which phytoplankters live is neither isotropic, nor unstructured. Planktic algae compete for a range of various limited resources, and are exposed to many different biotic (competition, grazing, parasitism, allelopathic substances) and abiotic constraints (light or nutrient limitation, sinking loss etc.). Most authors argue that Hardin’s competitive exclusion principle refers to equilibrium systems, but phytoplankton may never reach complete equilibrium due to external forces, which may set back succession repeatedly (Richerson et al., 1970; Sommer et al., 1993; Scheffer et al., 2003). Model simulations showed that competition for limiting resources results in chaotic oscillations among competing species, even in the lack of external spatial, or temporal physical forces (Huisman & Weissing, 1999). Furthermore, besides the various external forces and resources traditionally considered to be limiting (i.e. nutrients and light), several additional limiting factors (behavioural effects, predator–prey interactions, release of allelochemicals) might have further pronounced influence on the coexistence of planktic algae (Roy & Chattopadhyay, 2007). Diversity, however, can be studied at various organization levels ranging from genes to ecosystems (Yoccoz et al., 2001), and the level selected depends on the question raised. It has been demonstrated that functional properties of macroscopic plant communities could be better explained by functional rather than by species diversity (Tilman et al., 1997). Studies on microscopic communities also demonstrated that the use of functional diversity could be helpful in the understanding of functioning and key processes in algal (Steneck et al., 1994), or in bacterial assemblages (Zak et al., 1994).
The recognition that phytoplankton assemblages consist of phylogenetically different but functionally similar groups of algae has led to the development of functional group concept in phytoplankton ecology. Many approaches are now available that provide simple (Margalef, 1978; Kruk et al., 2010), or more detailed classification systems (Reynolds et al., 2002; Salmaso & Padisák, 2007). The functional group concept sensu Reynolds (FGs) has been successfully applied in both theoretical and applied studies. It has been demonstrated that functional composition of phytoplankton responds to changes in mixing regime (Becker et al., 2009; Wang et al., 2011), to anthropogenic pressures (Abonyi et al., 2012), and to hydrological changes in rivers (Stanković et al., 2012; Abonyi et al., 2014). The seasonal dynamics of the plankton (Padisák et al., 2003; Salmaso & Padisák, 2007; Wang et al., 2018) can also be described and interpreted by the functional group approach. The FG approach has also been used to better understand the productivity-diversity relationship for algae (Borics et al., 2012, 2014, Skácelová & Lepš, 2014; Török et al., 2016), or the response of algal assemblages to climate change (Domis et al., 2007).
Phytoplankton FGs have well-defined habitat preferences (Reynolds et al., 2002; Padisák et al., 2009), and the elements of the groups can flourish if their preferences can be successfully matched with characteristics of the habitat they live in (Reynolds, 2012). The algal groups occurring in a given habitat might show marked differences in their functional redundancy; i.e. in the number of species performing similar functions, which means that contribution of the groups to the overall species richness of phytoplankton is not equal (Borics et al., 2012).
However for species rich microscopic assemblages, the number of observed species is a negatively biased estimator of the total species richness (Walther & Moore, 2005). This problem can be surmounted by the application of species accumulation curves (Gotelli & Colwell, 2001) that makes possible the comparison of observed and predicted richness.
Here we studied the phytoplankton of a eutrophic, small, temperate oxbow lake in the Tisza river valley (Hungary). We hypothesized that contribution of the functional groups of algae to the overall species richness of the phytoplankton in a given lake is different, and these differences can be studied by analysing the species accumulation curves created for each functional group. The aim was to highlight differences in the functional redundancy of algae in natural eutrophic phytoplankton communities. We especially focused on how the various functional groups contribute to the overall richness of the lake, and how their contribution is influenced by their relative abundance, and by the different spatial and temporal scales.
We hypothesized that
-
(i)
similar level of functional redundancy (based on the observed number of species in FGs) does not necessarily represent equal roles in contributing to the overall richness of phytoplankton assemblages;
-
(ii)
relative biomass abundance of a functional group determines its contribution to the species richness.
Methods
Study area
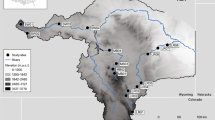
Phytoplankton diversity was studied in a small eutrophic water body, the Malom-Tisza oxbow (48 01′14″N; 21 11′27″E, Hungary), located in the middle of the Tisza River Valley, Carpathian Basin. The 11-km-long oxbow is separated into five sections. The middle section is the Malom-Tisza oxbow with a surface area of about 0.46 km2, a length of 4.2 km, a mean depth of 3 m, and a maximum depth of 12 m. Like many small oxbow lakes in the Tisza Valley, it shows apparent horizontal differences in macrophyte coverage (Borics et al., 2011), and it is characterized by strong summer thermal stratification (Borics et al., 2015). This spatial heterogeneity allows the development of various microhabitats in the oxbows, providing opportunities for the occurrence of highly diverse phytoplankton assemblages (Krasznai et al., 2010).
Sampling design
Spatial samplings
Samples were collected on 23 July 2007 during the characteristic summer thermal stratification period (Borics et al., 2015). To cover all characteristic habitats along the longitudinal section of the Malom-Tisza oxbow, a spatially intensive sampling scheme was applied (Fig. 1a). A total of 33 samples were collected from 11 sample locations along the lake. At each sampling location, three independent samples were taken at 1 m distance from each other from the euphotic layer using a tube sampler (length 2.5 m, diameter 0.06 m).
At smaller spatial scale, a vertical grid sampling scheme was implemented in the cross section of the oxbow (Fig. 1b). In the middle cross section of the oxbow, a total of 69 samples were collected with a hard plastic tube-sampler (length: 5 m, diameter: 0.03 m). In this case, the samples were collected from 5 water columns (left bank, left centre, middle, right centre, right bank at equal distances (15 m) from each other), in 25 cm vertical resolution.
Temporal samplings
The temporal samplings were performed between 2004 and 2010 in the vegetation periods. Three to five samples were taken in the May–October periods in every year. Altogether 25 samples were collected. In each of the sampling events, single samples were collected by tube sampler from the euphotic layer of the water column in the middle of the Northern curve of the oxbow (Fig. 1). Euphotic depth (Deu) was estimated from the Secchi disc depth (SD; Deu = 2.5 × SD). Samples were preserved in situ with Lugol’s solution.
Sample preparation
Phytoplankton cell numbers were enumerated by a Leica DMIL inverted bright-field microscope in 5 cm3 counting chambers (Lund et al., 1958; Utermöhl, 1958). During the counting of algal units, we applied three complementary approaches. To estimate the number of large-celled taxa, or large colonial forms (> 50 µm) the entire surface of the chamber was counted at × 100 magnification. In the case of smaller individuals (between 5 and 50 µm), a minimum of 400 specimens per sample were counted in transects at × 630 magnification. The smallest algae (< 5 µm, for example Romeria spp. or Chlorella spp.), which occasionally appeared in high abundance in the samples, were counted separately in every field of transects at × 630 magnification. Specific phytoplankton biovolume was estimated according to Hillebrand et al. (1999).
Assignment to functional groups
Each species was assigned to one of the functional groups proposed and encoded with letters by Reynolds et al. (2002) and reviewed by Padisák et al. (2009). This classification is based on the adaptive properties of planktic algae and their sensitivity to a range of environmental factors. Each group is named by letter codes. Since some of the functional groups contained only a few species, we used a simplified classification; that is, groups having similar ecological characteristics were merged into larger groups. Thus, eight functional groups were formed: (1) unicellular Chlorococcales; (2) algae without shape resistance, including planktic diatoms and desmids; (3) bloom-forming cyanobacteria; (4) small and middle sized flagellates; (5) large metaphytic flagellates, mostly euglenoids; (6) metaphytic desmids; (7) coenobial Chlorococcales; (8) large-celled flagellated planktic algae (dinoflagellates and Gonyostomum spp.). Coda of the functional groups that were assigned to larger categories and relevant species of these groups are shown in Table 1.
We considered the within-group species richness as a measure of functional redundancy (Wohl et al., 2004).
Statistical analyses
To facilitate comparisons among the community structures, species rank-frequency plots were constructed for the three sampling schemes. Relative frequency of species was expressed as percentage of samples containing the species.
Testing the first hypothesis Chao’s sample-based extrapolation curves (Chao et al., 2014) were used to estimate species richness for the three datasets (longitudinal section, cross section and time series). This non-asymptotic approach aims to compare diversity estimates for equally large or equally complete samples; it is based on the seamless rarefaction and extrapolation (R/E) sampling curves of three diversity metrics (richness, Shannon and Simpson indices). Since these kinds of estimators provide reliable extrapolation only for short-ranges, in each case we calculated the species richness for an extrapolated sample size of twice the number of observed samples (Colwell et al., 2012).
As we hypothesized, similarities in the observed functional redundancy does not necessarily represent an equal role in contributing to the overall richness of phytoplankton assemblages, therefore, we compared the shape of species saturation curves in the case of each FG. The shape of the curves, i.e. the presence or absence of asymptotes, clearly shows the differences between the diversity of the groups despite having similar observed richness values. Chao’s curves were constructed for each algal group and shapes of the curves were compared. Although the curves might have considerable dissimilarities, which could be visible by eye, characterization of the curves by numerical value (or values) is necessary. Species accumulation curves are frequently approximated by power functions (Arrhenius, 1921; Dengler, 2009) in which the exponent indicates the rate of increase. In our case, we found apparent differences in the curves, and the very low r2 values indicated that the shape of the curves could not been approximated well by power functions. Therefore, similarly to the half-saturation constant applied in enzyme kinetics, we calculated an Nh value corresponding to the number of samples in which half of the observed number of species occurred (Fig. 2). To make the comparisons among the three sampling layouts possible, we standardized Nh by dividing their values by the total number of samples (Nt):
where Ψ is the standardized value used in comparisons; Nh is the number of samples in which half of the observed species number occurs; and Nt is the total number of samples in each sampling layout. The standardized value (Ψ) was then used to characterize the species accumulation curves in each functional group. Theoretically, in case of linear increase the value of Ψ is 0.5, while low values (Ψ < 0.1) characterize the curves that show rapid increase and level off even at low sample numbers. Higher values of Ψ indicate that the real functional redundancy is considerably higher than the observed within group richness.
Although Nt was not identical across the three sampling layouts (short, long and temporal scale), this can potentially influence the values of Ψ in those cases, when species accumulation curves do not show asymptotes. However, the doubled sample numbers (that we used during extrapolation) were large enough for each FGs to approximate their asymptotes. Thus, Ψ is a reliable metric for evaluating the differences both within and across the sampling layouts.
To study the second hypothesis, we plotted Ψ of each FG against the mean relative biomass of FGs in samples of the three sampling layouts. Ordinary least squares method was used to fit the regression models. All statistical analyses were performed in R. Rarefaction and extrapolation curves were fitted using the iNEXT R package (Hsieh et al., 2016).
Results
Species richness and Ψ values
Investigation of the spatial samples revealed the presence of 190 euplanktic species. From the 33 points of the longitudinal Section 180 species were detected, while from the 69 points of the cross-Section 64 species were detected. In contrast, in the time series samples 291 planktic algae were observed. Although there were 169 species that occurred exclusively in the time series samples and 64 species in spatial samples, there was a considerable overlap between the species pool of the spatial and time series samples.
The extrapolation curves identified differences in richness values among the 3 sampling layouts (Fig. 3). The estimated species richness was greatest in the time series samples (370), followed by that of the longitudinal section (221). The lowest estimated value was obtained in the cross-Section (66) where the increase of species richness showed an asymptote.
Both the observed and estimated richness values and Ψ values of the bootstrapped species extrapolation curves constructed for the 8 functional groups showed remarkable differences (Table 2 and 3; Fig. 4a–c).
A three-time difference between the lowest and highest Ψ values was observed in the case of the time series samples (Table 3). The lowest value of Ψ (0.17) belonged to the group of planktic diatoms of which curves showed only very slight increase at the highest range of sample numbers. In contrast, in the case of the group of metaphytic desmids the Ψ value was 0.42. This value is remarkably high considering that the value of Ψ = 0.5 means linear increase in richness values along the sample number. The euglenoids, which similarly to the most of the desmids are considered metaphytic taxa, have been also characterized by high Ψ value (0.29).
Considerably higher scatter in the Ψ values was found in both sets of the spatially replicated samples (Table 3). The values ranged between 0.01 and 0.36 in the case of the samples of longitudinal section, and between 0.01 and 0.31 regarding the cross section’s samples. In samples taken in the cross section very low Ψ values (Ψ = 0.01) were found for three groups (planktic diatoms and desmids, bloom forming cyanobacteria, and large sized flagellates). The curves of these groups showed characteristic asymptotes. Small and middle celled flagellates exhibited the highest Ψ value (0.31) showing a steady increase even at the highest sample number.
As to the samples taken from the longitudinal section Ψ values varied considerably, with the highest values (0.36) obtained again for the metaphytic desmids. This value means steep increase in species richness even at the highest range of the sample number. Values of the other groups indicated a moderate increase in species numbers with the exception of bloom forming cyanobacteria (Ψ = 0.01), which group, similarly to the vertical section samples, also produced a richness ceiling.
When checking the sensitivity of Ψ to the functional redundancy, we found that the Ψ values did not show any statistical relationship with the richness values. High and low Ψ values belonged to both small and large species numbers (Fig. 5). This explicitly means that Ψ values can serve as reliable metrics for characterization of species accumulation curves.
Strong negative relationships were found between the Ψ values and the mean relative biomass of the functional groups for data of all three sampling layouts (Fig. 6). These results imply that species richness of the successful competitors which dominate the phytoplankton tend to level off even at small sample numbers. In contrast, high Ψ values and steady increase in the species richness can be expected in those groups in which the elements usually constitute only a small percentage of the assemblages.
Characteristics of frequency distributions
Species rank–frequency plots constructed for the data of the three sampling campaigns revealed two types of patterns (Fig. 7). Characteristic L-shaped distribution could be observed in the case of time series- and longitudinal section samples. In the longitudinal section, due to the large habitat diversity, we did not find species that would have been present in each sample. In case of time series samples, considerably different assemblages were sampled; therefore, the most frequently occurring species were found only in 70% of the samples. However, rank abundance plot of cross section samples showed a typical log-normal distribution with several frequently occurring species. In the cross section, due to the proximity of the samples, there were several species which occurred in almost all of the samples. However, the lower plateaus of all three curves showed particularly strong resemblance in the frequency distributions of species. Approximately 40% of the observed species belonged to the lower range of the curves, where frequency of the species was less than 5%.
Discussion
Comparing the taxonomic richness of the samples taken in the longitudinal and in the cross section of the oxbow, and the shapes of species accumulation curves we found remarkable differences. The higher number of taxa in the longitudinal section can be accounted for by larger diversity of the habitats. In a previous study Borics et al. (2011) have shown that the pelagic and littoral plant zones alternate along the longitudinal axis of the Malom-Tisza oxbow, providing various mezo- and microhabitats for the algae. This finding supports the view that spatial structuring of the habitats and the high level of environmental complexity maintain high phytoplankton diversity (Ptacnik et al., 2010).
Although the studied oxbow was stably stratified at the time of samplings, our results suggest that the long scale longitudinal habitat differences have a much larger impact on the species composition and richness than the effect of the stratification, which occur in a smaller spatial scale and shapes diversity in the cross section samples of the oxbow (Borics et al., 2015).
The number of taxa observed in time series samples highly exceeded those found in the spatial replicates. However, it can be explained by the fact that the spatially replicated samples show only the actual pattern of phytoplankton. In contrast, when a longer time series is studied, seasonal changes of the background variables i.e. temperature, light, stratification (Reynolds, 1989), grazing (Sterner, 1989) and the strength of interspecific competition (Sommer, 1989) basically determine the pattern of phytoplankton succession. These altogether highly contribute to profound seasonal compositional changes of the phytoplankton even in very small lakes (Grigorszky et al., 2000).
Species richness estimated by the time series samples is comparable to those reported for considerably larger lakes (Table 4). Although because of the differences in sampling effort across the various studies, a firm conclusion should not be drawn from this comparison, it clearly illustrates the unique richness of the studied oxbow. Phytoplankton diversity is influenced by various characteristics of the systems, such as productivity (Skacelova & Leps, 2014; Török et al., 2016), lake size (Smith et al., 2005), habitat diversity (Borics et al., 2003), or regional processes (Soininen et al., 2007). Investigating the productivity/diversity relationship for phytoplankton, Borics et al. (2014) demonstrated that species richness of lake phytoplankton peaks in the Chl-a range ~ 60–80 µg l−1, and about 20.5 mg l−1 biomass value (Török et al., 2016) in the eutrophic range. Although the size of the water bodies can also be an important predictor of species richness if a large spatial scale is considered (Smith et al., 2005; Stomp et al., 2011), as our results suggest, its role should not be overestimated. Recent studies revealed that in spatially heterogeneous aquatic environments, the occurrence of various microhabitats is highly responsible for the maintenance of diverse planktic microflora (Várbíró et al., 2017). In light of these findings, it is reasonable to suppose that the high species richness of the studied oxbow is due partly to its eutrophic character, to the large habitat diversity created by the pronounced horizontal differences along the lake basin (Borics et al., 2011), and—although to a less pronounced extent—to the vertical stratification of the water column (Borics et al., 2015). These physical characteristics contribute to the establishment of populations including various groups of flagellated algae, which can move to find their optimal position in the water column with respect to light, temperature and nutrients. These flagellated phytoplankters being characteristic for eutrophic, stratifying systems (Reynolds, 2006) constituted a significant portion of the species pool of the studied oxbow. However, the exceptionally high taxonomic richness of the oxbow and the fact that species richness has not levelled off, neither in the case of the spatially (longitudinal section), nor in the temporally replicated samples, can be explained by the large proportion of metaphytic species in the plankton samples. This ultimately can be traced back to the morphology of the lake basin, which enables the development of a macrophyte belt along the entire length of the oxbow, covering approximately 20% of the lake basin.
The unexpectedly high species richness revealed in the longitudinal sampling scheme has important consequences for understanding assembly rules in phytoplankton. Contrary to plants and sessile animal communities where competition for space determines the structure of communities (Sebens, 1982), in the case of phytoplankton, space competition might not be a crucial issue. It means that although the relative abundance of species that live in suboptimal conditions for growth and reproduction decreases below the detection limit of traditional sample processing techniques, these species do not necessarily disappear from the system. Both the results of rank-frequency analysis of the phytoplankton and the non-asymptotic character of the species accumulation curves suggest that a considerable portion of the lake’s species pool remains undetected for the observer. However, some of the rare species can occasionally occur in time series samples, which potentially implies that despite their low abundance, they can build stable populations in standing waters at a longer temporal scale.
Functional redundancy is a key feature of ecosystems, because redundant species are considered necessary to ensure ecosystem resilience to disturbances (Walker, 1995). The differences of species accumulation curves of the various FGs supported our hypothesis that a similar level of functional redundancy does not necessarily represent equal roles in contributing to the overall richness of phytoplankton assemblages. This finding has an important theoretical implication, because differences in species accumulation curves of FGs suggest differences in community assembly rules by which communities are organized (Cornell & Lawton, 1992). Based on the presence or absence of biotic interactions among the resident species living in a local habitat, Cornell and Lawton (1992) classified communities as interactive and non-interactive. In non-interactive communities, species respond to abiotic factors identically, biotic interactions are absent, and thus, population dynamics of species is independent of each other (Caswell, 1976). This model predicts that local species richness in these communities is not saturated if an infinitely large regional species pool is present. Species accumulation curves of most functional groups in the oxbow had no asymptotes. In our case, this was especially true for those groups (desmids and large celled flagellates), which preferably have metaphytic origin (Naselli-Flores & Barone, 2012). In contrast, bloom-forming cyanobacteria displayed an upper limit to local richness. These differences corroborated our hypothesis that relative biomass abundances of FGs determine their contribution to the species richness. Bloom-forming cyanobacteria have a special role in late summer phytoplankton assemblages. Elements of this group are good light competitors and constitute dense, low diversity populations in late summer assemblages of eutrophic lakes (Dokulil & Teubner, 2000; Padisák et al., 2003). Besides their regular dominance, these taxa can also be characterized by wide regional distribution, and thus can be considered as core species (Hanski, 1982) of the phytoplankton. The richness ceiling that was observed in the longitudinal section for bloom forming cyanobacteria cannot be explicitly explained by pool exhaustion (Cornell, 1993), because results on the temporally replicated samples revealed that both the observed and estimated number of taxa within this group is considerably higher than the value at which the saturation had been attained. As it was highlighted by Cornell (1993), within-group competition among constituent core species supports species saturation. Dominance of the good light competitor K strategist blue greens is a typical example of this phenomenon. They occur in large abundance, can be characterized with low Ψ values, show asymptotes in species richness, and thus, constitute a strongly interactive part of the phytoplankton. However, in contrast to the spatially replicated samples, in the case of time series samples, bloom forming cyanobacteria did not show asymptotes. This finding suggests that competition which is an important assembly rule for phytoplankton does not lead to species exclusion in longer time scale, or if it happens, these taxa can easily be re-established from the adjacent water bodies.
In the light of our findings, an interesting parallelism can also be drawn between microscopic aquatic and macroscopic terrestrial systems. The best light competitors of the temperate forests are the tall trees that provide canopy cover and consequent shade for the herbaceous plants living on the floor of the forest. These latter plants cannot compete for light, but can tolerate its low availability. Due to their good light-competitive abilities, trees enjoy plenty of sunshine, can attain high biomass, and can control the herb-layer diversity (Wulf & Naaf, 2009), but their richness in temperate forests is limited and is highly exceeded by the richness of the herb-layer (Mölder et al., 2008). This phenomenon seems to be analogous to our findings, where despite the dominance of blue-greens, richness of outcompeted taxa can still remain reasonably high.
Conclusions
Our results revealed surprisingly high actual species richness for a small stratified oxbow in the Tisza River Valley (Hungary), confirming that habitat diversity is elementary to maintain high species diversity in phytoplankton.
We demonstrated that the sample-based species accumulation curves of phytoplankton functional groups (FGs) show considerable differences (having asymptotes or showing steady increase even at large sample numbers), and thus, their contribution to the overall species richness is different.
The presence or lack of asymptotes of species accumulation curves seems to imply different intensities of within-group interactions. Based on this finding, we consider phytoplankton as assemblages that contain interactive and non-interactive elements. The functional approach applied in this study helps to characterize the phytoplankton diversity, and provides us with new perspectives to understand the assembly rules of natural phytoplankton assemblages.
References
Abonyi, A., M. Leitão, A. M. Lançon & J. Padisák, 2012. Phytoplankton functional groups as indicators of human impacts along the River Loire (France). Hydrobiologia 698(1): 233–249.
Abonyi, A., M. Leitão, I. Stanković, G. Borics, G. Várbíró & J. Padisák, 2014. A large river (River Loire, France) survey to compare phytoplankton functional approaches: do they display river zones in similar ways? Ecological Indicators 46: 11–22.
Arrhenius, O., 1921. Species and area. Journal of Ecology 9: 95–99.
Becker, V., V. L. M. Huszar & L. O. Crossetti, 2009. Responses of phytoplankton functional groups to the mixing regime in a deep subtropical reservoir. Hydrobiologia 628(1): 137–151.
Bondarenko, N.A., 1995 List of planktonic algae in Lake Baikal. In: Guide and key to pelagic animals of Baikal Timoshkin. O. A. (ed.), Nauka, Novosibirsk, 621–630.
Borics, G., B. Tóthmérész, I. Grigorszky, J. Padisák, G. Várbíró & S. Szabó, 2003. Algal assemblage types of bog-lakes in Hungary and their relation to chemistry, hydrological conditions and habitat diversity. Hydrobiologia 502(1–3): 145–155.
Borics, G., A. Abonyi, E. Krasznai, G. Várbíró, I. Grigorszky, S. Szabó, C. S. Deák & B. Tóthmérész, 2011. Small-scale patchiness of the phytoplankton in a lentic oxbow. Journal of Plankton Research 33(6): 973–981.
Borics, G., B. Tóthmérész, B. A. Lukács & G. Várbíró, 2012. Functional groups of phytoplankton shaping diversity of shallow lake ecosystems. Hydrobiologia 698(1): 251–262.
Borics, G., J. Görgényi, I. Grigorszky, Z. S. László-Nagy, B. Tóthmérész, E. Krasznai & G. Várbíró, 2014. The role of phytoplankton diversity metrics in shallow lake and river quality assessment. Ecological Indicators 45: 28–36.
Borics, G., A. Abonyi, G. Várbíró, J. Padisák & E. T-Krasznai, 2015. Lake stratification in the Carpathian basin and its interesting biological consequences. Inland Waters 5(2): 173–186.
Caswell, H., 1976. Community structure: a neutral model analysis. Ecological Monographs 46(3): 327–354.
Chao, A., N. J. Gotelli, T. C. Hsieh, E. L. Sander, K. H. Ma, R. K. Colwell & A. M. Ellison, 2014. Rarefaction and extrapolation with Hill numbers: a framework for sampling and estimation in species diversity studies. Ecological Monographs 84(1): 45–67.
Colwell, R. K., A. Chao, N. J. Gotelli, S. Y. Lin, C. X. Mao, R. L. Chazdon & J. T. Longino, 2012. Models and estimators linking individual-based and sample-based rarefaction, extrapolation and comparison of assemblages. Journal of Plant Ecology 5: 3–21.
Cornell, H. V., 1993. Unsaturated patterns in species assemblages: the role of regional processes in setting local species richness. Species diversity in ecological communities. University of Chicago Press, Chicago, Illinois, USA: 243–252.
Cornell, H. V. & J. H. Lawton, 1992. Species interactions, local and regional processes, and limits to the richness of ecological communities: a theoretical perspective. Journal of Animal Ecology 61: 1–12.
Dengler, J., 2009. Which function describes the species–area relationship best? A review and empirical evaluation. Journal of Biogeography 36(4): 728–744.
Dokulil, M. T. & K. Teubner, 2000. Cyanobacterial dominance in lakes. Hydrobiologia 438(1–3): 1–12.
Domis, L. N. D. S., W. M. Mooij & J. Huisman, 2007. Climate-induced shifts in an experimental phytoplankton community: a mechanistic approach. Hydrobiologia 584(1): 403–413.
Gotelli, N. J. & R. K. Colwell, 2001. Quantifying biodiversity: procedures and pitfalls in the measurement and comparison of species richness. Ecology Letters 4(4): 379–391.
Grigorszky, I., S. Nagy, L. Krienitz, K. T. Kiss, M. M. Hamvas, A. Tóth, G. Borics, C. S. Máthé, B. Kiss, G. Y. Borbély, G. Y. Dévai & J. Padisák, 2000. Seasonal succession of phytoplankton in a small oligotrophic oxbow and some consideration to the PEG model. Internationale Vereinigung für theoretische und angewandte Limnologie: Verhandlungen 27(1): 152–156.
Hanski, I., 1982. Dynamics of regional distribution: the core and satellite species hypothesis. Oikos 38: 210–221.
Hillebrand, H., C. D. Dürselen, D. Kirschtel, U. Pollingher & T. Zohary, 1999. Biovolume calculation for pelagic and benthic microalgae. Journal of Phycology 35(2): 403–424.
Hsieh, T.C., K.H. Ma & A. Chao, 2016. iNEXT: An R package for interpolation and extrapolation of species diversity (Hill numbers). Methods in Ecology and Evolution. R package version 2.0.8, URL: http://chao.stat.nthu.edu.tw/blog/software-download
Huisman, J. & F. J. Weissing, 1999. Biodiversity of plankton by species oscillations and chaos. Nature 402(6760): 407.
Hutchinson, G. E., 1961. The paradox of the plankton. The American Naturalist 95(882): 137–145.
Krasznai, E., G. Borics, G. Várbíró, A. Abonyi, J. Padisák, C. S. Deák & B. Tóthmérész, 2010. Characteristics of the pelagic phytoplankton in shallow oxbows. Hydrobiologia 639: 261–269.
Kruk, C., V. L. Huszar, E. T. Peeters, S. Bonilla, L. Costa, M. Lürling, C. S. Reynolds & M. Scheffer, 2010. A morphological classification capturing functional variation in phytoplankton. Freshwater Biology 55(3): 614–627.
Laugaste, R., V. V. Jastremskij & I. Ott, 1996. Phytoplankton of lake Peipsi-Pihkva: species composition, biomass and seasonal dynamics. Hydrobiologia 338: 49–62.
Lund, J. W. G., C. Kipling & E. D. Cren, 1958. The inverted microscope method of estimating algal numbers and the statistical basis of estimations by counting. Hydrobiologia 11(2): 143–170.
Margalef, R., 1978. Life-forms of phytoplankton as survival alternatives in an unstable environment. Oceanologica Acta 1(4): 493–509.
McIntire, C. D., G. L. Larson, R. E. Truitt & M. K. Debacon, 1996. Taxonomic structure and productivity of phytoplankton assemblages in Crater Lake, Oregon. Lake and Reservoir Management 12(2): 259–280.
Mölder, A., M. Bernhardt-Römermann & W. Schmidt, 2008. Herb-layer diversity in deciduous forests: raised by tree richness or beaten by beech? Forest Ecology and Management 256(3): 272–281.
Munawar, M. & I. F. Munawar, 1978. Phytoplankton of Lake Superior 1973. Journal of Great Lakes Research 4(3): 415–442.
Naselli-Flores, L. & R. Barone, 2012. Phytoplankton dynamics in permanent and temporary Mediterranean waters: is the game hard to play because of hydrological disturbance? Hydrobiologia 698(1): 147–159.
Padisák, J., 1992. Seasonal succession of phytoplankton in a large Shallow Lake (Balaton, Hungary)–a dynamic approach to ecological memory, its possible role and mechanisms. Journal of Ecology 80(2): 217–230.
Padisák, J., G. Borics, G. Fehér, I. Grigorszky, I. Oldal, A. Schmidt & Z. S. Zámbóné-Doma, 2003. Dominant species, functional assemblages and frequency of equilibrium phases in late summer phytoplankton assemblages in Hungarian small shallow lakes. Hydrobiologia 502(1–3): 157–168.
Padisák, J., L. O. Crossetti & L. Naselli-Flores, 2009. Use and misuse in the application of the phytoplankton functional classification: a critical review with updates. Hydrobiologia 621(1): 1–19.
Ptacnik, R., S. D. Moorthi & H. Hillebrand, 2010. Hutchinson reversed, or why there need to be so many species. In Woodward, G. (ed.), Integrative Ecology: From Molecules to Ecosystems, Vol. 43. Academic Press, Elsevier Ltd., Cambridge: 1–33.
Reynolds, C. S., 1989. Physical determinants of phytoplankton succession. Plankton Ecology. Springer, Berlin Heidelberg: 9–56.
Reynolds, C. S., 2006. The ecology of phytoplankton. Cambridge University Press, Cambridge.
Reynolds, C. S., 2012. Environmental requirements and habitat preferences of phytoplankton: chance and certainty in species selection. Botanica Marina 55(1): 1–17.
Reynolds, C. S., V. Huszar, C. Kruk, L. Naselli-Flores & S. Melo, 2002. Towards a functional classification of the freshwater phytoplankton. Journal of Plankton Research 24(5): 417–428.
Richerson, P., R. Armstrong & C. R. Goldman, 1970. Contemporaneous Disequilibrium, a New Hypothesis to Explain the „Paradox of the Plankton”. Proceedings of the National Academy of Sciences 67(4): 1710–1714.
Roy, S. & J. Chattopadhyay, 2007. Towards a resolution of ‘the paradox of the plankton’: A brief overview of the proposed mechanisms. Ecological Complexity 4(1): 26–33.
Salmaso, N. & J. Padisák, 2007. Morpho-functional groups and phytoplankton development in two deep lakes (Lake Garda, Italy and Lake Stechlin, Germany). Hydrobiologia 578(1): 97–112.
Scheffer, M., S. Rinaldi, J. Huisman & F. Weissing, 2003. Why plankton communities have no equilibrium: solutions to the paradox. Hydrobiologia 491(1–3): 9–18.
Sebens, K. P., 1982. Competition for space: growth rate, reproductive output, and escape in size. American Naturalist 120: 189–197.
Skácelová, O. & J. Lepš, 2014. The relationship of diversity and biomass in phytoplankton communities weakens when accounting for species proportions. Hydrobiologia 724(1): 67–77.
Smith, V. H., B. L. Foster, J. P. Grover, R. D. Holt, M. A. Leibold & F. de NoyellesJr, 2005. Phytoplankton species richness scales consistently from laboratory microcosms to the world’s oceans. Proceedings of the National Academy of SciencesUSA 102: 4393–4396.
Soininen, J., M. Kokocinski, S. Estlander, J. Kotanen & J. Heino, 2007. Neutrality, niches, and determinants of plankton metacommunity structure across boreal wetland ponds. Ecoscience 14(2): 146–154.
Sommer, U., 1989. The role of competition for resources in phytoplankton succession. Plankton Ecology. Springer, Berlin Heidelberg: 57–106.
Sommer, U., J. Padisák, C. S. Reynolds & P. Juhász-Nagy, 1993. Hutchinson’s heritage: the diversity-disturbance relationship in phytoplankton. Hydrobiologia 249(1–3): 1–7.
Stanković, I., T. Vlahović, M. G. Udovič, G. Várbíró & G. Borics, 2012. Phytoplankton functional and morpho-functional approach in large floodplain rivers. Hydrobiologia 1(698): 217–231.
Steneck, R. S. & M. N. Dethier, 1994. A functional group approach to the structure of algal-dominated communities. Oikos 69: 476–498.
Sterner, R. W., 1989. The role of grazers in phytoplankton succession. Plankton Ecology. Springer, Berlin Heidelberg: 107–170.
Stomp, M., J. Huisman, G. Mittelbach, E. Litchman & C. Klausmeier, 2011. Large-scale biodiversity patterns in freshwater phytoplankton. Ecology 92: 2096–2107.
Tilman, D., J. Knops, D. Wedin, P. Reich, M. Ritchie & E. Siemann, 1997. The influence of functional diversity and composition on ecosystem processes. Science 277(5330): 1300–1302.
Török, P., E. T-Krasznai, V. B-Béres, I. Bácsi, G. Borics & B. Tóthmérész, 2016. Functional diversity supports the biomass-diversity humped-back relationship in phytoplankton assemblages. Functional Ecology 30: 1593–1602.
Utermöhl, H., 1958. Zur Vervollkommnung der quantitativen Phytoplankton-Methodik. Mitteilunger der internationale Vereinigung für Theoretische und Angewandte Limnologie 9: 1–38.
Várbíró, G., J. Görgényi, B. Tóthmérész, J. Padisák, É. Hajnal & G. Borics, 2017. Functional redundancy modifies species–area relationship for freshwater phytoplankton. Ecology and Evolution 7(23): 9905–9913.
Walker, B., 1995. Conserving biological diversity through ecosystem resilience. Conservation Biology. 9: 747–752.
Walther, B. A. & J. L. Moore, 2005. The concepts of bias, precision and accuracy, and their use in testing the performance of species richness estimators, with a literature review of estimator performance. Ecography 28(6): 815–829.
Wang, L., Q. Cai, Y. Xu, L. Kong, L. Tan & M. Zhang, 2011. Weekly dynamics of phytoplankton functional groups under high water level fluctuations in a subtropical reservoir-bay. Aquatic Ecology 45(2): 197–212.
Wang, C., B. Viktória, C. Stenger-Kovács, X. Li & A. Abonyi, 2018. Enhanced ecological indication based on combined planktic and benthic functional approaches in large river phytoplankton ecology. Hydrobiologia 818(1): 163–175.
Wohl, D. L., S. Arora & J. R. Gladstone, 2004. Functional redundancy supports biodiversity and ecosystem function in a closed and constant environment. Ecology 85(6): 1534–1540.
Wulf, M. & T. Naaf, 2009. Herb layer response to broadleaf tree species with different leaf litter quality and canopy structure in temperate forests. Journal of Vegetation Science 20(3): 517–526.
Yoccoz, N. G., J. D. Nichols & T. Boulinier, 2001. Monitoring of biological diversity in space and time. Trends in Ecology & Evolution 16(8): 446–453.
Zak, J. C., M. R. Willig, D. L. Moorhead & H. G. Wildman, 1994. Functional diversity of microbial communities: a quantitative approach. Soil Biology and Biochemistry 26(9): 1101–1108.
Acknowledgements
Open access funding provided by MTA Centre for Ecological Research (MTA ÖK). This work was funded by the GINOP-2.3.2-15-2016-00019, OTKA K104279, OTKA K116639 grants, and by the National Research, Development and Innovation Office (NKFIH, Ref. No.: PD 124681).
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Judit Padisák
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Görgényi, J., Tóthmérész, B., Várbíró, G. et al. Contribution of phytoplankton functional groups to the diversity of a eutrophic oxbow lake. Hydrobiologia 830, 287–301 (2019). https://doi.org/10.1007/s10750-018-3878-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-018-3878-3