Abstract
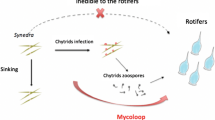
The Phytomyxea (“plasmodiophorids”) including both Plasmodiophorida and Phagomyxida is a monophyletic group of Eukaryotes composed of obligate biotrophic parasites of green plants, brown algae, diatoms and stramenopiles commonly found in many freshwater, soil and marine environments. However, most research on Phytomyxea has been restricted to plant pathogenic species with agricultural importance, thereby missing the huge ecological potential of this enigmatic group of parasites. Members of the Phytomyxea can induce changes in biomass in their hosts (e.g. hypertrophies of the host tissue) under suitable environmental conditions. Upon infection they alter the metabolism of their hosts, consequently changing the metabolic status of their host. This results in an altered chemical composition of the host tissue, which impacts the diversity of species which feed on the tissues of the infected host and on the zoospores produced by the parasites. Furthermore, significant amounts of nutrients derived from the hosts, both primary producers (plants and algae) and primary consumers (litter decomposers and plant parasites [Oomycetes]), can enter the food web at different trophic levels in form of zoospores and resting spores. Large numbers of zoospores and resting spores are produced which can be eaten by secondary and tertiary consumers, such as grazing zooplankton and metazoan filter-feeders. Therefore, these microbes can act as energy-rich nutrient resources which may significantly alter the trophic relationships in fresh water, soil and marine habitats. Based on the presented data, Phytomyxea can significantly contribute to the complexity and energy transfer within food webs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
During the past decade an unexpectedly high diversity of microbial eukaryotes has been revealed in many aquatic habitats (Moreira & López-García, 2002; Gleason et al., 2008; Lefèvre et al., 2008; Lopez-Garcia & Moreira, 2008; Sime-Ngando et al., 2010). Amongst these eukaryotes the heterotrophic flagellates are thought to play key roles in aquatic ecosystems as parasites and saprobes. Heterotrophic flagellates (HF) are defined as microbes with zoospores approximately less than or equal to 5 μm in diameter and without chlorophyll (Sime-Ngando et al., 2010). Their roles in food webs include recycling inorganic and organic nutrients, contributing to the energy flow in food webs, trophic upgrading and regulation of population size of their hosts (Sime-Ngando et al., 2010).
Based on the morphology of both the primary and secondary zoosporic stages, Phytomyxea can be included in the ecological concept of HF as proposed by Sime-Ngando et al. (2010). However, phytomyxean zoospores lack distinctive morphological features which would allow them to be easily identified within a pool of HF. In the studies by Lefèvre et al. (2007, 2008) the analysis of HF populations revealed a considerable number of zoospores belonging to saprobic or parasitic zoosporic true fungi (Chytridiomycota, Blastocladiomycota, etc.) and groups of parasitic protists. Although no Phytomyxea were detected by Lefèvre et al. (2008), we propose that these microbes are frequently present in freshwater, soil and marine ecosystems. Recently evidence has suggested that the Phytomyxea have been under-sampled in DNA-based environmental screening studies in many ecosystems because of negative primer bias (Neuhauser et al., 2010). However, DNA sequences of some previously unknown taxa of Phytomyxea have been identified in clone libraries from a saline meromictic lake (Takishita et al., 2007a), and in sediment of a deep sea methane cold steep (Takishita et al., 2007b). Furthermore, a high diversity of known and unsampled taxa has been detected in randomly collected soil and fresh water samples using primers specific for Plasmodiophorida (Neuhauser et al., unpublished). Collectively these studies support the view that this group of microbes has been previously under-sampled in many ecosystems. Furthermore it can be expected that Phytomyxea are widely distributed in many aquatic ecosystems and play more important roles as biotrophs than previously thought.
The purposes of this article are (i) to describe the distinctive features of Phytomyxea, (ii) to provide the basis for considering them as members of the HF group and (iii) to discuss some of their potential ecological roles in freshwater, soil and marine ecosystems. The ecological potential will be illustrated using data available from the sparse literature on aquatic hosts and from the more detailed studies on soil-borne plant pathogens. We expect that the underlying mechanisms of parasitism are similar in all aquatic, soil and marine habitats.
Classification
The Phytomyxea are a monophyletic group of Eukaryotes. During the past few years the common name “plasmodiophorids” as introduced by Braselton has been used for this group (Braselton, 1995). The true taxonomic position of this group has been debated frequently since the publication of the original description of Plasmodiophora brassicae Woronin (reviewed by Neuhauser et al., 2010). Woronin (1878) classified Pl. brassicae originally as a protist (sensu Haeckel). Later the Phytomyxea were thought to belong to the Mastigomycota together with other zoosporic fungi and stramenopiles. Sparrow (1960) included them in the aquatic phycomycetes. Currently they are considered to be members of the protist supergroup Rhizaria, because the affiliation of Phytomyxea to the Rhizaria has been indicated repeatedly by molecular studies (Cavalier-Smith, 1993; Bulman et al., 2001; Archibald & Keeling, 2004; Nikolaev et al., 2004; Bass et al., 2009). Throughout this article we will use zoological nomenclature for the higher taxonomic levels. Tentatively the Class Phytomyxea is placed within the Phylum Cercozoa and the Subphylum Endomyxa (Bass et al., 2009). Based on 18S rDNA data the Class Phytomyxea comprises two orders: Plasmodiophorida, which are mainly parasites of green plants and the Phagomyxida which are parasites of diatoms and brown algae. Currently there are 41 known species belonging to twelve genera (Table 1).
Life cycles
All known species of Phytomyxea are obligate biotrophs. These microbes have complex life cycles with two free swimming zoosporic stages, two plasmodial stages that are linked to the host, a thin-walled zoosporangial stage and thick-walled resting spores. In some species the resting spores aggregate into cytosori which are the most prominent and important morphological characteristic. The form, size and pigmentation of the resting spores and the mode of cytosoral aggregation are used for species delimitation in Plasmodiophorida. These thick-walled resting spores have not been identified in the Phagomyxida yet. But recently evidence indicates that two morphologically different types of zoospores are produced by the algal endoparasite Maullinia ectocarpii I. Maier, E. R. Parodi, Westermeier & D. G. Müller and the presence of sporangia that slightly differ in thickness of cell walls was reported (Parodi et al., 2010). As detailed knowledge on the life cycles of the phytomyxean parasites of true aquatic organisms is very sparse, the life cycle illustrated below summarises the current knowledge of the universal stages of the life cycle observed in most Phytomyxea.
The life cycle starts when a single primary zoospore emerges out of one resting spore (thick-walled sporangium). These primary zoospores are (2) 2.5–4.5 (5) μm in size and pyriform to spindle shaped, with two flagellae [dimensions are (min) median width − median length (max) of all species where the zoospore size is given by Karling (1968)]. The shorter flagellum is blunt ended whereas the longer flagellum has a whiplash tailpiece. In contact with a suitable host the zoospore encysts and infects the host plant with a specialised extrusome: the Stachel and Rohr (Aist & Williams, 1971). In the host plant the parasite enlarges and forms a multinucleate plasmodium (primary plasmodium). During this stage the cruciform mitotic division typical for Phytomyxea can be observed. The nucleolus is enlarged and elongated into two directions resulting in the cross-like structure visible in TEM sections (Braselton et al., 1975; Braselton, 2001). The primary plasmodium undergoes a series of concerted nuclear divisions before cleaving into zoosporangia. In these zoosporangia one to numerous secondary zoospores develop. Morphologically these secondary zoospores cannot be differentiated from the primary zoospores, but in species where both forms are known, the secondary zoospores seem to be slightly larger than the primary zoospores with an average size of (1.5) 3.4–4.6 (7) μm (Karling, 1968). The secondary zoospores infect a suitable host in which secondary plasmodia are formed. These secondary plasmodia develop into resting spores and the life cycle is completed. But the life cycle is complicated. Primary zoospores can develop into resting spores, secondary zoospores can develop into zoosporangia and zoospores can shed their flagellae and can be propelled by amoeboid movement. When and how meiotic recombination in Phytomyxea takes place is still presumptive and debated (Ingram & Tommerup, 1972; Mithen & Magrath, 1992; Braselton, 2001; Manzanares-Dauleux et al., 2001; Fähling et al., 2004; Siemens et al., 2009b).
There is a considerable lack of knowledge of the life cycles of many species of Phytomyxea, but the life cycle of Pl. brassicae has been thoroughly studied (reviewed by Dixon, 2009b; Kageyama & Asano, 2009). Consequently we base some of our conclusions on data derived from parasites of “terrestrial” plants, and we expect the underlying processes to be very similar for parasites of aquatic plants. This viewpoint is supported by the descriptions of the response of host plants to an infection. These are quite similar for “terrestrial” and true aquatic host plants (Karling, 1968). Although most Phytomyxea are thought to be host-specific, some species can develop zoosporangia in alternative species of host plants (Karling, 1968; Legrève et al., 2005). These zoosporangia lack morphological traits which allow discrimination between species. Nothing is known about either the interaction between the parasites and the alternative host plants or the frequency of infection. Therefore, the discussion of the possible roles of Phytomyxea in the environment will be limited to the life cycle in the primary host species.
Ecological roles
In general Plasmodiophorida have received the attention of plant pathologists because some species cause notorious plant diseases and/or transmit plant viruses of economic importance. However, of the 41 known species of Phytomyxea only four species have been studied intensively during the past few decades. Species parasitic on aquatic and marine hosts such as oomycetes, brown algae and diatoms have hardly been studied at all during this period. Nevertheless, their hosts have been found more and more to play important roles in aquatic and marine habitats, such as brown algae, which are considered to be the “forests of the sea” because of their important role as primary producers. Other phytomyxids are parasitic on seagrasses, which are marine flowering plants that grow completely submersed in pure sea or estuarine water, and also which are important primary producers (Orth et al., 2006). The ecological importance of seagrasses for stabilising sediments, as organic carbon producers, and for nutrient cycling has lead to huge restoration and conservation efforts recently (Orth et al., 2006).
Because of their obligate biotrophic life cycle phytomyxids spend most of their lives endobiotically inside their hosts. However, they usually propagate with free swimming zoospores, the only stage of their life cycle that is not directly dependent upon a living host. These highly motile zoospores require water (water bodies, soil water and water films) to successfully complete their life cycles. 23 of the described species depend on hosts that are true aquatic organisms, namely oomycetes, diatoms, brown algae, freshwater aquatic plants and seagrass (Table 1), and many of the terrestrial host plants live in (periodically) wet soils with a high availability of water (e.g. flood plains, wet meadows). Although Plasmodiophorida infecting agricultural plants are classified as soil-borne pathogens, one should note that at agricultural sites the soil water trends to be at high and at constant levels because of irrigation during the growing season. Therefore, we consider the zoospores to be an aquatic stage of the phytomyxid life cycle, and that these are very likely to be included into aquatic food webs.
An anecdotal support that phytomyxids are prey in aquatic food webs was given by observations of Dylewski & Miller (1983) who reported that the survival of Woronina pythii Goldie-Sm. in hemp seed dual cultures with its host (Pythium spp.) is limited to 10 days when the culture is contaminated with bacteria or other micro-organisms. Besides being prey themselves, phytomyxid parasites can influence food webs by altering their hosts’ metabolism and by reducing the energy available to their hosts. Therefore, they can impact biotic (e.g. grazers) and abiotic factors (e.g. energy value of host tissue) in their environment. Phytomyxea have a huge ecological potential to contribute to energy transfer within ecosystems. Nothing is known about trophic strategies of phytomyxean zoospores. The energy needed for the subsequent development of the obligate biotrophic phytomyxids is thought to be exclusively derived from their hosts. Thus, we propose two roles for Phytomyxea. They can (i) facilitate the transfer of energy derived from primary producers (i.e., parasites of green plants, green and brown algae, diatoms), or from saprobes and other parasites to the pool of HF (i.e., parasites of heterotrophic stramenopiles) and (ii) indirectly affect the carbon flows in aquatic ecosystems by altering growth and chemical composition of their host (especially species causing hypertrophies of their host).
Phytomyxea as food source
Owing to their multiphasic life cycle, phytomyxean parasites can be integrated into the food web at different stages with differing nutritional values. The zoosporic stages are actively swimming in water, whereas the resting spores are introduced into the ecosystem passively upon disintegration of host tissue. Resting spores and zoospores can be dispersed by water currents and by animal vectors and, if the resting spores are resistant to drying, they can be carried by the wind as well from one habitat to another (Faggian et al., 1999; Dixon, 2009b; Faggian & Strelkov, 2009). This can result in their introduction as energy-rich compounds into completely new ecosystems. Although the resting spores are not solely aquatic (i.e. they can be part of terrestrial and rhizosphere ecosystems as well) we will discuss the implications of resting spores on food webs, as some information about their chemical composition and persistence is available. We only consider resting spores that are free in the environment (soil, water, or sediment) and we will focus on the implications of resting spores floating in the water column or sedimented for aquatic environments.
Resting spores can serve as long-term storage of nutritional energy in the ecosystem thus resulting in a sequential transfer of nutritional energy into the environment. It has been repeatedly reported that not all zoospores are released from the resting spores from one plant at the same time (Pendergrass, 1950; Karling, 1968; Dixon, 2009b). Moreover, the resting spores provide the parasites with the capacity for long term survival in the environment. In field studies on Pl. brassicae a half-life of resting spore viability of 3–6 years and longevity of up to 18 years has been reported (Wallenhammar, 1998). These values are considered to be true for most phytomyxids forming thick-walled resting spores. The long survival times in soil imply that the cell wall is very resistant to degradation. Experiments on the ingestion of resting spores of Spongospora subterranea (Wallr.) Lagerh. and Pl. brassicae showed that the resting spores can survive the passage through the digestive systems of livestock (Karling, 1968; Merz, 2008) and earthworms (Friberg et al., 2008). The potential to digest the components of the resting spores varies with species eating them. Therefore, it is possible that resting spores might be consumed sequentially by more than one individual before being completely digested. The consumption by animals or large protists which are not able to break down the resting spores completely can results in a spatial transfer of nutritional energy in the ecosystem. Resting spores presumably have large energy reserves allowing them quickly to react to favourable environmental conditions, germinate and infect a suitable host. Quantitative data on the chemical composition of the resting spores of phytomyxid species besides Pl. brassicae are missing, but similar values can be assumed for other phytomyxids forming resting spores. The walls of the resting spores of Pl. brassicae contain high amounts of high energy substances such as chitin (25% of the cell wall), proteins (33%), lipids (17.5%) and carbohydrates (2.5%; Buczacki & Moxham, 1983). Recently the fatty acid composition of the resting spores of Pl. brassicae was investigated and arachidonic acid (20ω6.9.12.15) was found to be the most abundant fatty acid (36% of total lipid content, Sundelin et al., 2010). Also during the late stages of pathogen development trehalose is produced in high amounts in the plant, which is thought to be responsible for the stability of the resting spores (Brodmann et al., 2002).
The zoosporic stages of Phytomyxea are more important for a “quick” energy transfer from primary producers and decomposers to other trophic levels. Although the primary and secondary zoospores cannot be distinguished morphologically, their function in phytomyxean dispersal seems to differ. Primary zoospores emerge out of resting spores which are—when outside the plant tissue—“randomly” distributed in the environment and are, therefore, thought to be responsible for the colonisation of a new host (long range dispersal, Dixon, 2009b). The secondary zoospores are quickly produced in high numbers upon the primary infection of a new host and were found to circle around and re-infect the host (Pendergrass, 1950; Goldie-Smith, 1954; Karling, 1968; Dixon, 2009b). Therefore, the primary zoospores are more likely found outside of the area of influence of their host whereas the secondary zoospores are found mainly within the area influenced by the host (e.g. rhizosphere, phyllosphere of aquatic plants). The host excretes substances into the environment, which creates a rhizosphere microcosm that is known to be colonised by a different community of bacteria, protists and animals (Bonkowski, 2004) than found in free soil or water bodies. This consequently implies that the energy transferred in form of primary and secondary zoospores to the next trophic level might impact different food webs.
Energy transfer mediated by Phytomyxea
When present in a host population Phytomyxea are responsible for a considerable amount of additional biomass production as typically many plants are infected. In his monograph “The Plasmodiophorales” Karling (1968) considers all species of Phytomyxea to be rare except for Pl. brassicae, Sp. subterranea and Woronina pythii. Recent results provide evidence that other species such as Polymyxa betae Keskin, Px. graminis Ledingham, Sorosphaera veronicae J. Schröt., So. viticola Kirchm., Neuh. & L. Huber and Ligniera junci (Schwartz) Maire & A. Tisson can be found abundantly in habitats in or in close proximity to fresh water (Neuhauser et al., 2005; Huber et al., 2007; Neuhauser & Kirchmair, 2009). Some of the less known species are probably widely distributed, but often overlooked because of a very rapid life cycle. Karling (1944) noted in his studies on the brown algal parasite Phagomyxa algarum Karling that it has a life cycle of 1–2 days. After this time the host cells together with all remnants of the parasite are destroyed. Nevertheless, this time is very short compared to the times required for development of other Phytomyxea that were reported to be 16–40 days (M. ectocarpii, Maier et al., 2000), 20–30 days (Pl. brassicae, Kageyama & Asano, 2009) or 20–25 days (W. polycystis, Goldie-Smith, 1954).
Based on records in the literature the marine species Tetramyxa parasitica K. I. Goebel, Pl. diplantherea (Ferd. & Winge) Ivimey Cook and Pl. bicaudata Feldmann seem to be abundant where suitable hosts grow (Karling, 1968; den Hartog, 1989; Walker & Campbell, 2009). Karling (1968) lists places in Sweden, Finland, Norway, Denmark, Germany, the UK, France, Morocco, Italy, Poland, Holland, USA and New Zealand where T. parasitica was found, thus indicating a global distribution of this species. Studies on herbarium specimens of Zostera spp. revealed that plants infected with Pl. bicaudata have been deposited worldwide without recognition of the parasite (den Hartog, 1989). Moreover, within this study evidence was found that Phytomyxea are able to follow their host over long distances. In populations of the invasive species Zostera japonica Aschers. & Graebn. near Vancouver, the presence of Pl. bicaudata was reported 10 years after plant invasion (den Hartog, 1989). Plasmodiophora diplantherea appears to be common in the Gulf of Mexico (Louisiana, Mississippi) and along the coast of Florida (Braselton & Short, 1985; Walker & Campbell, 2009). All three parasites cause marked hypertrophies of their host together with reduced shoot and root growth. However, the size of the galls is not directly proportional to the number of spores produced, indicating that additionally a considerable amount of biomass is produced in the form of host tissue (Wernham, 1935; Dixon, 2009a).
Species not causing marked hypertrophies of their host cells can convert a considerable amount of energy from their host during their life cycle as well. Pendergrass (1950) reported that within days up to 65% of the hyphae of the oomycete Brevilegnia spp. were colonised by Octomyxa brevilegniea Pend. Similar infection rates can be assumed for Woronina polycystis (Cornu) A. Fisch. a species which is parasitic on host species in the Saprolegniales. Goldie-Smith (1954) reported that “Encysted spores have been seen in such large numbers that they formed a ‘fur over the hyphae’ of oomycetes”. In plants of Juncus spp. infected with Ligniera junci approximately 20–50% of the root hairs are filled with the parasite (Neuhauser et al. unpublished).
Indirect changes in food webs by altering growth and chemical composition of the host plant
A change in the chemical composition of the host alters its edibility and subsequently the organisms which feed on them (Niquil et al., 2010). Various micro- and macro-organisms feeding on the roots or grazing on microorganisms are attracted to usable substrates (Adl & Gupta, 2006; Friberg et al., 2008; Gleason et al., 2008). Clubbed roots—like other galls induced by Phytomyxea—are a rich source for nutrients and are quickly colonised by bacteria and fungi after the resting spores were formed (Woronin, 1878; Chupp, 1917; Karling, 1968; Dixon, 2009b). The ecological potential of Phytomyxea to alter the energy transfer to the food web will be exemplified using mainly data from Pl. brassicae supplemented with data on other Phytomyxea. Given the similar life cycle of phytomyxean species and the similar reaction of their hosts to infection, most of the metabolic processes discussed below may be similar in other species, especially to the ones causing hypertrophies of their host.
The processes triggered in the host leading to hypertrophies are similar for all phytomyxean species. When a compatible host is infected, the colonisation of the host tissue is accompanied by increased cell divisions of the host cells (Karling, 1968). Eighteen phytomyxean species causing hypertrophies have been described, most of them parasitic on true aquatic plants (Table 1). The processes leading to gall formation were studied in detail for Pl. brassicae (reviewed by Ludwig-Muller et al., 2009), but the possible effect of these alterations on the energy flux into the ecosystem has, as far as we are aware, not been considered previously.
Plasmodiophorabrassica takes over the hosts’ metabolism resulting in an altered chemical composition of clubroots compared to healthy roots, making them attractive as food source. The plasmodia of Pl. brassicae produce small amounts of glucosinolates (plant growth hormones) in the roots which consequently create a sink for sugars, auxins and other plant hormones (Ludwig-Muller et al., 2009; Siemens et al., 2009a, b). As the disease progresses, photosynthetic assimilate are increasingly transported from the leaves to the root system (Mitchell & Rice, 1979). This is reflected by up-regulated transport processes of sugars, lipids, ions, N, S and P (Devos et al. 2006; Siemens et al., 2006). Consequently hexoses, sucrose and starch are accumulated in the roots (Mithen & Magrath, 1992; Evans & Scholes, 1996; Brodmann et al., 2002) together with a higher content of glucose, which is formed instead of fructose biphosphate (Devos et al., 2006). The metabolism of polyamines—important components of the plant cell wall—was up-regulated as well (Jubault et al., 2008). High concentrations of phytoalexines and phytoanticipines—low molecular weight phenolic compounds linked to plant defence—were found in the roots (Soledade et al., 2008) together with increased amounts of flavanoids (Ludwig-Muller et al., 2009). Upon infection the composition of fatty acids also changes (Sundelin et al., 2010).
All the changes in the chemical composition of the host, which were previously discussed, result in an improvement of its food quality. The amount of easily metabolised nutrients (glucose, sucrose and starch) in the root is increased together with an increased input of amino acids, sterols and fatty acids making the clubbed roots an attractive food source. The processes triggered in the plant at molecular level and consequently the substances enriched in the galls vary slightly between cultivars and species of Brassica spp. (Ludwig-Muller et al., 2009). Although the changes discussed above have been documented for soil ecosystems only, we hypothesise that some of these changes also impact the energy fluxes triggered in the host plant when galls of other phytomyxean species are formed (i.e. creating a sink for photosynthetic assimilates), and possibly to a lesser extent in non-hypertrophic species.
This view is supported by reports on increased aggregation of starch in host cells infected by Tetramyxa parasitica (host Ruppia spp.), Sorodiscus karlingii Ivimey Cook (host Chara spp.), Woronina glomerata (host Vaucheria spp.) and Phagomyxa algarum (host Bachelotia antillarum (Grunow) Gerloff and Hincksia mitchelliae (Harvey) Silva; Karling, 1968). Karling (1944) observed a change from brown to green in the colour of the host brown alga H. mitchelliae upon infection with Ph. algarum, possibly reflecting a change of the photosynthetic status of the host. Pendergrass (1950) noted an increased transport of nutrition globules to cells of the oomycete Brevilegnia spp. infected with Octomyxa brevilegniea. The pronounced modification of the metabolism of the host seems to be undoubtedly a universal process during infection by Phytomyxea. This conclusion is further supported by the observation that when the plasmodia surround the host nucleus it becomes enlarged and stays visible until the resting spores are formed (Karling, 1928; Karling, 1944; Miller, 1959; Dylewski & Miller, 1983; Kageyama & Asano, 2009).
There are two modes by which non-hypotrophic species can indirectly influence the energy input into the environment by: (i) increasing the root biomass and (ii) reducing the root biomass. An increase in root biomass was recently observed in roots of Juncus spp. growing in englacial streams. Roots infested with Ligniera junci showed an increased growth of root hair (Neuhauser & Kirchmair, 2009; Figs. 1–3). Ligniera junci causes no other visible symptoms of disease on its host plant, but infected roots of Juncus spp. could easily be identified by this increased formation of root hair.
A decrease in size and number of roots was reported especially for Phytomyxea parasitising marine plants. The two marine species Pl. diplantherae, a parasite of Halodule wrightii Asch., and Pl. bicaudata, parasitic on small Zostera spp. cause galls at the internodes and rhizomes of their host plants. In both species a reduced growth of the roots of host plants was positively correlated with an increased rate of uprooting (den Hartog, 1989; Walker & Campbell, 2009). In addition, den Hartog (1989) noted that thoroughly searching a Zostera-bed resulted in finding only three plants infected with Pl. bicaudata, whilst back at the beach he quickly found additional infected plants which had been washed there. Besides the possible relevance of this parasites for sea grass restoration projects, this uprooting was considered to be a strategy of the parasite for long distance dispersal and the colonisation of new host populations. Also, the floating plants can provide a considerable input into food webs outside the sea grass beds considering infection densities of 53–100% of H. wrightii with Pl. diplantherae (Walker & Campbell, 2009).
Concluding remarks and future perspectives
Based on the data we have presented, an enormous potential for Phytomyxea to influence the energy fluxes in aquatic ecosystems becomes evident. In aquatic ecosystems the zoospores of Phytomyxea facilitate a significant transfer of energy from primary producers (photosynthetic organisms) and heterotrophic stramenopiles (primary consumers) to secondary and tertiary consumers such as grazing zooplankton or filter feeding metazoa. The energy-rich resting spores of phytomyxean parasites have the potential to transfer energy from primary producers to consumers in nutrient depleted habitats by being carried over long distances by water flow, wind or animal vectors. Resting spores can also be considered as excellent energy resources on a temporal scale, as they can remain viable for many years. Moreover Phytomyxea change the metabolic status of their hosts, resulting in an altered chemical composition and altered amount of host biomass.
Considering the fact that only species causing marked hypertrophies to host plants have been described from freshwater or marine habitats, there might be a hidden biodiversity of Phytomyxea causing no hypertrophies. According to Hudson et al. (2006) parasites play essential roles in ecosystem function and stability. Parasites influence not only the fitness of their hosts, but can serve as link between trophic levels (Lafferty et al., 2008). Phytomyxea are very commonly found in many freshwater, soil and marine ecosystems. Therefore, we expect these microbes to significantly contribute to food webs by facilitating the transfer of matter and energy between trophic levels and to increase the complexity of food webs.
Besides a basic understanding of the life cycle and taxonomy of many species, very little is known about the ecological importance of the Phytomyxea. Many of the parasites of aquatic plants, algae and diatoms have been reported only once, and research on the Phytomyxea has been limited to a few species causing plant diseases of economic importance. The very sparse data available on diversity, abundance and distribution in fresh water and marine habitats allow only a rough estimation of the ecological importance of this group. To reliably evaluate their possible regulatory or selective roles in these ecosystems, qualitative and quantitative data on their distribution and their interaction with primary and alternative host plants are needed. Recent 18S rDNA surveys have revealed a complex composition of small flagellate eukaryotes in many different aquatic habitats (Moreira & López-García, 2002; Gleason et al., 2008; Lefèvre et al., 2008; Lopez-Garcia & Moreira, 2008; Sime-Ngando et al., 2010). In all studies an unexpectedly high diversity of parasitic organisms was found, but the Phytomyxea still remain under-sampled (Neuhauser et al., 2010). Also the few Phytomyxea rDNA sequences identified in these studies could not be assigned to previously described species (Bass et al., 2009; Neuhauser et al., 2010). Studies with primers specific for the Phytomyxea could provide information on biodiversity, distribution and quantity of Phytomyxea in a wide range of aquatic habitats. Without this basic knowledge the ecological significance of this group of organisms can hardly be evaluated.
References
Adl, M. S. & V. Gupta, 2006. Protists in soil ecology and forest nutrient cycling. Canadian Journal of Forest Research-Revue Canadienne De Recherche Forestiere 36: 1805–1817.
Aist, J. R. & P. H. Williams, 1971. Cytology and kinetics of cabbage root hair penetration by Plasmodiophora brassicae. Canadian Journal of Botany 49: 2023–2034.
Archibald, J. M. & P. J. Keeling, 2004. Actin and ubiquitin protein sequences support a cercozoan/foraminiferan ancestry for the plasmodiophorid plant pathogens. The Journal of Eukaryotic Microbiology 51: 113–118.
Bass, D., E. E. Y. Chao, S. Nikolaev, A. Yabuki, K. I. Ishida, C. Berney, U. Pakzad, C. Wylezich & T. Cavalier-Smith, 2009. Phylogeny of novel naked filose and reticulose Cercozoa: Granofilosea cl. n. and Proteomyxidea. Protist 160: 75–109.
Bonkowski, M., 2004. Protozoa and plant growth: the microbial loop in soil revisited. New Phytologist 162: 617–631.
Braselton, J. P., 1995. Current status of the plasmodiophorids. Critical Reviews in Microbiology 21: 263–275.
Braselton, J. P., 2001. Plasmodiophoromycota. In McLaughlin, D. J., E. G. McLaughlin & P. A. Lemke (eds), The Mycota VII Part A. Systematics and Evolution. Springer-Verlag, Berlin: 81–91.
Braselton, J. P. & F. T. Short, 1985. Karyotypic analysis of Plasmodiophora diplantherae. Mycologia 77: 940–945.
Braselton, J. P., C. E. Miller & D. G. Pechak, 1975. The ultrastructure of cruciform nuclear division in Sorosphaera veronicae (Plasmodiophoromycete). American Journal of Botany 62: 349–358.
Brodmann, D., A. Schuller, J. Ludwig-Müller, R. A. Aeschbacher, A. Wiemken, T. Boller & A. Wingler, 2002. Induction of trehalase in Arabidopsis plants infected with the trehalose-producing pathogen Plasmodiophora brassicae. Molecular Plant–Microbe Interactions 15: 693–700.
Buczacki, S. T. & S. E. Moxham, 1983. Structure of the resting spore wall of Plasmodiophora brassicae revealed by electron microscopy and chemical digestion. Transactions of the British Mycological Society 81: 221–231.
Bulman, S. R., S. F. Kuhn, J. W. Marshall & E. Schnepf, 2001. A phylogenetic analysis of the SSU rRNA from members of the Plasmodiophorida and Phagomyxida. Protist 152: 43–51.
Cavalier-Smith, T., 1993. Kingdom Protozoa and its 18 phyla. Microbiological Reviews 57: 953–994.
Chupp, C., 1917. Studies on clubroot of cruciferous plants. Cornell Agriculture Experimental Station Bulletin 387: 421–452.
den Hartog, C., 1989. Distribution of Plasmodiophora bicaudata a parasitic fungus on small Zostera species. Diseases of Aquatic Organisms 6: 227–230.
Devos, S., K. Laukens, P. Deckers, D. Van der Straeten, T. Beeckman, D. Inze, H. Van Onckelen, E. Witters & E. Prinsen, 2006. A hormone and proteome approach to picturing the initial metabolic events during Plasmodiophora brassicae infection on Arabidopsis. Molecular Plant–Microbe Interactions 19: 1431–1443.
Dixon, G. R., 2009a. The occurrence & economic impact of Plasmodiophora brassicae & clubroot disease. Journal of Plant Growth Regulation 28: 194–202.
Dixon, G. R., 2009b. Plasmodiophora brassicae in its environment. Journal of Plant Growth Regulation 28: 212–228.
Dylewski, D. P. & C. E. Miller, 1983. Cruciform nuclear division in Woronina pythii (Plasmodiophoromycetes). American Journal of Botany 70: 1325–1339.
Evans, J. L. & J. D. Scholes, 1996. The effect of clubroot upon the carbon metabolism of its host? Journal of Experimental Botany 47: 72.
Faggian, R. & S. E. Strelkov, 2009. Detection & measurement of Plasmodiophora brassicae. Journal of Plant Growth Regulation 28: 282–288.
Faggian, R., A. C. Lawrie, S. R. Bulman & I. J. Porter, 1999. Specific polymerase chain reaction primers for the detection of Plasmodiophora brassicae in soil & water. Phytopathology 89: 392–397.
Fähling, M., H. Graf & J. Siemens, 2004. Characterization of a single-spore isolate population of Plasmodiophora brassicae resulting from a single club. Journal of Phytopathology 152: 438–444.
Friberg, H., J. Lagerlöf, K. Hedlund & B. Rämert, 2008. Effect of earthworms & incorporation of grass on Plasmodiophora brassicae. Pedobiologia 52: 29–39.
Gleason, F. H., M. Kagami, E. Lefevre & T. Sime-Ngando, 2008. The ecology of chytrids in aquatic ecosystems: roles in food web dynamics. Fungal Biology Reviews 22: 17–25.
Goldie-Smith, E. K., 1954. The position of Woronina polycystis in the Plasmodiophoraceae. American Journal of Botany 41: 441–448.
Huber, L., G. Eisenbeis, E. H. Ruehl, V. Pagay & M. Kirchmair, 2007. Distribution and host range of the grapevine plasmodiophorid Sorosphaera viticola. Vitis 46: 23–25.
Hudson, P. J., A. P. Dobson & K. D. Lafferty, 2006. Is a healthy ecosystem one that is rich in parasites? Trends in Ecology and Evolution 21: 381–385.
Ingram, D. S. & I. C. Tommerup, 1972. The life history of Plasmodiophora brassicae Woron. Proceedings of the Royal Society London B 180: 103–112.
Jubault, M., C. Hamon, A. Gravot, C. Lariagon, R. Delourme, A. Bouchereau & M. J. Manzanares-Dauleux, 2008. Differential regulation of root arginine catabolism and polyamine metabolism in clubroot-susceptible and partially resistant Arabidopsis genotypes. Plant Physiology 146: 2008–2019.
Kageyama, K. & T. Asano, 2009. Life cycle of Plasmodiophora brassicae. Journal of Plant Growth Regulation 28: 203–211.
Karling, J. S., 1928. Studies in the chytridiales III. A parasitic chytrid causing cell hypertrophy in Chara. American Journal of Botany 15: 485–487.
Karling, J. S., 1944. Phygomyxa algarum n. gen., n. sp., an unusual parasite with Plasmodiophoralean and Protomyxean characteristics. American Journal of Botany 31: 38–52.
Karling, J. S., 1968. The Plasmodiophorales, 2nd Completely Revised Edition. Hafner Publishing Company, New York.
Lafferty, K. D., S. Allesina, M. Arim, C. J. Briggs, G. DeLeo, A. P. Dobson, J. A. Dunne, P. T. Johnson, A. M. Kuris, D. J. Marcogliese, N. D. Martinez, J. Memmott, P. A. Marquet, J. P. McLaughlin, E. A. Mordecai, M. Pascual, R. Poulin & D. W. Thieltges, 2008. Parasites in food webs: the ultimate missing links. Ecology Letters 11: 533–546.
Lefèvre, E., C. Bardot, C. Noel, J. F. Carrias, E. Viscogliosi, C. Amblard & T. Sime-Ngando, 2007. Unveiling fungal zooflagellates as members of freshwater picoeukaryotes: evidence from a molecular diversity study in a deep meromictic lake. Environmental Microbiology 9: 61–71.
Lefèvre, E., B. Roussel, C. Amblard & T. Sime-Ngando, 2008. The molecular diversity of freshwater picoeukaryotes reveals high occurrence of putative parasitoids in the plankton. PloS One 3: e2324.
Legrève, A., J. Schmit, C. Bragard & H. Maraite, 2005. The role of climate and alternative hosts in the epidemiology of rhizomanina. Proceedings of the Sixth Symposium of the International Working Group on Plant Viruses with Fungal Vectors.
Lopez-Garcia, P. & D. Moreira, 2008. Tracking microbial biodiversity through molecular and genomic ecology. Research in Microbiology 159: 67–73.
Ludwig-Muller, J., E. Prinsen, S. A. Rolfe & J. D. Scholes, 2009. Metabolism and plant hormone action during clubroot disease. Journal of Plant Growth Regulation 28: 229–244.
Maier, I., E. Parodi, R. Westermeier & D. G. Muller, 2000. Maullinia ectocarpii gen. et sp. nov. (Plasmodiophorea), an intracellular parasite in Ectocarpus siliculosus (Ectocarpales, Phaeophyceae) and other filamentous brown algae. Protist 151: 225–238.
Manzanares-Dauleux, M. J., I. Divaret, F. Baron & G. Thomas, 2001. Assessment of biological and molecular variability between & within field isolates of Plasmodiophora brassicae. Plant Pathology 50: 165–173.
Merz, U., 2008. Powdery scab of potato – occurrence, life cycle and epidemiology. American Journal of Potato Research 85: 241–246.
Miller, C. E., 1959. Studies on the life cycle and taxonomy of Ligniera verrucosa. American Journal of Botany 46: 725–729.
Mitchell, D. T. & K. A. Rice, 1979. Translocation of C-14-labeled assimilates in cabbage during club root development. Annals of Applied Biology 92: 143–152.
Mithen, R. & R. Magrath, 1992. A contribution to the life history of Plasmodiophora brassicae – secondary plasmodia development in root galls of Arabidopsis thaliana. Mycological Research 96: 877–885.
Moreira, D. & P. López-García, 2002. The molecular ecology of microbial eukaryotes unveils a hidden world. Trends in Microbiology 10: 31–38.
Neuhauser, S. & M. Kirchmair, 2009. Ligniera junci, a plasmodiophorid re-discovered in roots of Juncus in Austria. Österreichische Zeitschrift für Pilzkunde 18: 151–157.
Neuhauser, S., L. Huber & M. Kirchmair, 2005. Sorosphaera veronicae; neu für Österreich. Österreichische Zeitschrift für Pilzkunde 14: 303–308.
Neuhauser, S., S. Bulman & M. Kirchmair, 2010. Plasmodiophorids: The challenge to understand soil-borne, obligate biotrophs with a multiphasic life cycle. In Gherbawy, Y. & K. Voigt (eds), Current Advances in Molecular Identification of Fungi. Springer, Heidelberg: 51–78.
Nikolaev, S. I., C. Berney, J. F. Fahrni, I. Bolivar, S. Polet, A. P. Mylnikov, V. V. Aleshin, N. B. Petrov & J. Pawlowski, 2004. The twilight of Heliozoa and rise of Rhizaria, an emerging supergroup of amoeboid eukaryotes. Proceedings of the National Academy of Sciences of the United States of America 101: 8066–8071.
Niquil, N., M. Kagami, J. Urabe, U. Christaki, E. Viscogliosi & T. Sime-Ngando, 2010. Potential role of fungi in plankton food web functioning and stability: a simulation analysis based on Lake Biwa inverse model. Hydrobiologia (in press). doi:10.1007/s10750-010-0308-6.
Orth, R. J., T. J. B. Carruthers, W. C. Dennison, C. M. Duarte, J. W. Fourqurean, K. L. Heck, A. R. Hughes, G. A. Kendrick, W. J. Kenworthy, S. Olyarnik, F. T. Short, M. Waycott & S. L. Williams, 2006. A global crisis for seagrass ecosystems. Bioscience 56: 987–996.
Parodi, E. R., E. J. Caceres, R. Westermeier & D. G. Müller, 2010. Secondary zoospores in the algal endoparasite Maullinia ectocarpii (Plasmodiophoromycota). Biocell 34: 45–52.
Pendergrass, W. R., 1950. Studies on a plasmodiophoraceaous parasite, Octomyxa brevilegniae. Mycologia 42: 279–289.
Siemens, J., I. Keller, J. Sarx, S. Kunz, A. Schuller, W. Nagel, T. Schmulling, M. Parniske & J. Ludwig-Muller, 2006. Transcriptome analysis of Arabidopsis clubroots indicate a key role for cytokinins in disease development. Molecular Plant–Microbe Interactions 19: 480–494.
Siemens, J., H. Graf, S. Bulman, O. In & J. Ludwig-Muller, 2009a. Monitoring expression of selected Plasmodiophora brassicae genes during clubroot development in Arabidopsis thaliana. Plant Pathology 58: 130–136.
Siemens, J., S. Bulman, F. Rehn & T. Sundelin, 2009b. Molecular biology of Plasmodiophora brassicae. Journal of Plant Growth Regulation 28: 245–251.
Sime-Ngando, T., E. Lefevre & F. H. Gleason, 2010. Hidden diversity among aquatic heterotrophic flagellates: ecological potentials of zoosporic fungi. Hydrobiologia. doi:10.1007/s10750-010-0230-y.
Soledade, M., C. Pedras, Q.-A. Zheng & S. Strelkov, 2008. Metabolic changes in roots of the oilseed canola infected with the biotroph Plasmodiophora brassicae: phytoalexins and phytoanticipins. Journal of Agricultural and Food Chemistry 56: 9949–9961.
Sparrow Jr., F. K., 1960. Aquatic Phycomycetes. University of Michigan Press, Ann Arbor.
Sundelin, T., C. B. Christensen, J. Larsen, K. Moller, M. Lubeck, L. Bodker & B. Jensen, 2010. In planta quantification of Plasmodiophora brassicae using signature fatty acids & real-time PCR. Plant Disease 94: 432–438.
Takishita, K., M. Tsuchiya, M. Kawato, K. Oguri, H. Kitazato & T. Maruyama, 2007a. Genetic diversity of microbial eukaryotes in anoxic sediment of the saline meromictic lake Namako-ike (Japan): on the detection of anaerobic or anoxic-tolerant lineages of eukaryotes. Protist 158: 51–64.
Takishita, K., N. Yubuki, N. Kakizoe, Y. Inagaki & T. Maruyama, 2007b. Diversity of microbial eukaryotes in sediment at a deep-sea methane cold seep: surveys of ribosomal DNA libraries from raw sediment samples & two enrichment cultures. Extremophiles 11: 563–576.
Walker, A. K. & J. Campbell, 2009. First records of the seagrass parasite Plasmodiophora diplantherae from the northcentral Gulf of Mexico. Gulf and Caribbean Research 21: 63–65.
Wallenhammar, A. C., 1998. Observations on yield loss from Plasmodiophora brassicae infections in spring oilseed rape. Zeitschrift für Pflanzenkrankheiten und Pflanzenschutz-Journal of Plant Diseases and Protection 105: 1–7.
Wernham, C. C., 1935. A species of Sorodiscus on Heteranthera. Mycologia 27: 262–273.
Woronin, M., 1878. Plasmodiophora brassicae. Urheber der Kohlpflanzen Hernie. Jahrbuch für wissenschaftliche Botanik 11: 548–574.
Acknowledgments
SN and MK wish to thank R. Pöder, University of Innsbruck, Austria for passing on his knowledge and expertise. SN was supported by a Hertha-Firnberg research grant (Austrian Science Fund (FWF) grant T379-B16).
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest editors: T. Sime-Ngando & N. Niquil / Disregarded Microbial Diversity and Ecological Potentials in Aquatic Systems
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Neuhauser, S., Kirchmair, M. & Gleason, F.H. The ecological potentials of Phytomyxea (“plasmodiophorids”) in aquatic food webs. Hydrobiologia 659, 23–35 (2011). https://doi.org/10.1007/s10750-010-0508-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-010-0508-0