Abstract
Background
In patients affected by heart failure (HF) with reduced ejection fraction (HFrEF), pharmacological treatments have been proven to alleviate symptoms and improve prognosis, while no treatment other than sodium-glucose co-transporter-2 inhibitors have demonstrated significant effects in HF with preserved ejection fraction (HFpEF). Left atrium decompression devices (LADd) have been recently investigated as a new interventional approach in patients with HFpEF.
Objectives
To assess the efficacy of LADd on soft endpoints in HF patients across the spectrum of ejection fraction.
Methods
PubMed and Web of Science were searched without restrictions from inception to 28 May 2022 to identify valuable articles. The studies that met the inclusion criteria were analyzed. The prespecified main outcomes were the change from baseline in 6-min walking distance (6MWD), NYHA class and health-related quality of life (HRQoL). Secondary outcomes were reduction in HF hospitalizations, echocardiographic, and hemodynamic parameters.
Results
Eleven studies, with a total of 547 patients, were included. LADd significantly improved 6MWD by 43.95 m (95% CI 29.64–58.26 m), decreased NYHA class by 0.93 (95% CI 1.20–0.67), and improved HRQoL questionnaire by 20.45 points (95% CI 13.77–27.14) with better results for all outcomes in patients with lower EFs.
Conclusion
The present meta-analysis suggests that LADd are favorable in improving 6MWD, NYHA class, and HRQoL in HF across a wide spectrum of ejection fraction, with better outcomes in patients with lower EFs.
Trial registration
CRD42022336077, URL: https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=336077.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
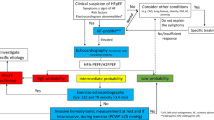
In patients affected by heart failure (HF) with reduced ejection fraction (HFrEF; left ventricular EF ≤ 40%), pharmacological treatment has been proven to favorably affect cardiac remodeling, alleviate symptoms, and enhance cardiac function and prognosis [1]. On the contrary, apart from sodium-glucose co-transporter-2 inhibitors (SGLT2i) [2, 3], no pharmaceutical treatment has been demonstrated to reduce morbidity and mortality in individuals with HF with preserved ejection fraction (HFpEF; EF ≥ 50%). A common symptom of patients with HF, regardless of EF, is dyspnea on efforts, which may be the result of an abrupt development of pulmonary congestion, due to elevated left ventricular and atrial filling pressures [4]. Individual titration of HF pharmacological treatment, especially diuretics, guided by invasive pulmonary artery pressures analysis, may better control congestion and minimize HF hospitalizations [5]; however, pressure-guided HF therapy requires patient adherence, and it is rather difficult and expensive [6]. Similarly to patients with mitral stenosis, in whom a concomitant small congenital atrial septal defect (Lutembacher’s syndrome) is associated with fewer symptoms and better outcomes compared with isolated mitral stenosis [7], various left atrium decompression devices (LADd) are currently under investigation in patients with HF. The present systematic review and meta-regression analysis investigated the feasibility and efficacy of dedicated LADd on symptoms, quality of life, functional status, hemodynamic parameters, and cardiac function in HF individuals across the spectrum of EF (Graphical abstract).
Methods
This meta-analysis was performed based on the Preferred Reporting Items for Systematic Reviews and Meta Analyses guidelines [8] (Table S1) and registered on PROSPERO (CRD42022336077).
Search strategy and study selection
PubMed and Web of Science were searched without any restrictions from inception to 28 May 2022. The search strategy is included in the Supplementary Materials. Two authors separately examined the titles and abstracts of all obtained publications to exclude clearly unrelated research. According to the inclusion criteria, the remaining articles were chosen for full-text examination. The final list of included studies was then reviewed by the authors, and any differences were addressed via discussion. Studies were included if they satisfied the following criteria: (1) a comprehensive study design with rigorous inclusion and exclusion criteria; (2) participants affected by HF; and (3) if a main study had more than one follow-up, only those having the longest follow-up duration were included.
Data extraction and quality assessment
The primary efficacy outcomes were improvement in six-minute walking distance (6MWD), NYHA class and health related quality of life (HRQoL), assessed by specific questionnaire.
The secondary efficacy outcomes were HF hospitalization (HHF), analyzed by comparing the number of HHF occurred in the follow-up period after LADd implantation to the number HHF occurred in the same time lapse before the device implantation, mean right atrial pressure (mRAP), mean pulmonary artery pressure (mPAP), tricuspidal annular plane systolic excursion (TAPSE), pulmonary capillary wedge pressure (PCWP), NT-proBNP variations, and safety concerns of LADd. Device-related adverse events were considered as safety primary outcome, a composite of device embolization, migration or removal, stroke (fatal and non-fatal) or transient ischemic attack, cardiac tamponade, and emergency cardiac surgery; this outcome was derived according to the events reported in each study as device-related.
In addition, a second safety outcome was analyzed, by only considering randomized controlled trial (RCT) that reported a composite of major adverse cardiovascular, cerebral, and renal events (MACCRE), specifically including cardiovascular death, embolic stroke and new onset or worsening kidney function.
The following information was gathered from each included study: baseline characteristics of studies (authors, publication year, journal, country), patients’ characteristics (sample size, gender, age, comorbidities), hemodynamic variables (post-capillary wedge pressure, mean atrial pressure, tricuspid annulus plane excursion, mean pulmonary arterial pressure), functional status changes (6MWD, NT-proBNP, HRQoL), and major adverse events (all-cause mortality, HHF). To analyze the risk of bias, the GRADE tool was used [9] (Table S2). Two reviewers independently estimated means and measures of dispersion from figures in the reports using DigitizeIt, version 2.5 (Braunschweig, Germany), if required. The final values were determined by averaging the opinions of independent reviewers.
Statistical analysis
STATA 17.0 (Stata Corp, College Station, TX, USA) was used. The chi-square test and I2 test were used to investigate heterogeneity, with p ≤ 0.10 or I2 > 50% indicating considerable heterogeneity. A DerSimonian and Laird random-effects model was used. Risk ratios (RR) and 95% confidence intervals (CI) were estimated for binary variables and weighted mean difference (WMD) and 95% CI were determined for the quantitative variables.
In addition, sensitivity analysis, funnel plots, and Egger’s test were performed to assess the stability of estimates and publication bias of included papers. A two-tailed p value of 0.05 was deemed significant.
Random-effects meta-regression analysis was performed to measure the impact of baseline ejection fraction on the effect size for the primary outcomes.
As with prior research using comparable analytic methodologies [10], the pre-procedure patient cohort was designated as the comparison group.
Recent methods [11,12,13] were used to convert data provided as sample size, median, first and third quartiles, or minimum and maximum to mean and its related standard error.
The HRQoL was evaluated by the Minnesota living with heart failure (MLWHF) questionnaire or by the Kansas City cardiomyopathy questionnaire (KCCQ), as both instruments demonstrate HRQoL improvement and deterioration on oppositely directed axes, the MLWHF scores were inverted (higher scores imply superior HRQoL) prior to standardization.
Results
Study characteristics
Of 1200 identified papers, 25 were retrieved for a more detailed evaluation (Fig. S1). According to the inclusion criteria, 2 studies were rejected, while 12 paper reported the results of the same trial at different follow-up duration or were abstracts on the same trial. Eventually, 11 studies were included, published between 2015 and 2022, comprising 547 patients, and with a follow-up time that varied from 3 to 27 months [14,15,16,17,18,19,20,21,22,23,24].
Two trials [14, 16] assessed quality of life by the MLWHF questionnaire and 6 studies [15, 17, 19, 20, 23, 24] by the KCCQ; the two questionnaire scales were converted in order to compare the results, as previously described.
Table 1 provides a summary of the baseline characteristics of the included studies.
Primary outcomes
Ten studies examined the change in submaximal exercise capacity assessed by the 6MWT. The mean baseline 6MWD varied between 242 and 454 m. Among the included studies, the use of LADd provided an average mean 6MWD increase of 43.95 m (95% CI 29.64–58.26 m) (Fig. 1). To further investigate this outcome result, meta regression and cumulative meta-analysis were performed according to EF (Fig. 2). Meta regression analysis showed a consistent effect across the spectrum of EF, with a greater effect in low EFs; cumulative meta-analysis confirmed this observed trend.
Change in 6MWD (mt). Solid squares represent mean differences in trials and have a size proportional to the weight of the difference. The 95% confidence intervals (CI) for individual trials are denoted by lines and those for the pooled mean differences by empty diamonds. 6MWD, six-minute walking distance; CI, confidence interval
Eleven studies examined the change in NYHA class with the use of LADd; the average mean NYHA class decreased by 0.93 (95% CI 1.20–0.67) (Fig. 3). To further explore this finding and to assess the considerable reported heterogeneity, meta regression and cumulative meta-analysis were performed according to EF (Fig. 4). Meta regression analysis showed a consistent effect across the spectrum of EF, with a greater effect in low EFs; cumulative meta-analysis confirmed this observed trend.
Eight studies assessed QoL, the average HRQoL score improved by 20.45 points (95% confidence interval 13.77–27.14) (Fig. 5). To further understand this finding and to assess the considerable reported heterogeneity, meta regression and cumulative meta-analysis were performed according to EF (Fig. 6). Meta regression analysis showed a consistent effect across the spectrum of EF, with a greater effect in low EFs; cumulative meta-analysis confirmed this observed trend.
Change in HRQoL. Solid squares represent mean differences in trials and have a size proportional to the weight of the difference. The 95% confidence intervals (CI) for individual trials are denoted by lines and those for the pooled mean differences by empty diamonds CI, confidence interval; HRQoL, health-related quality of life
There were no significant differences among the different devices in terms of 6MWD, NYHA class change, or HRQoL improvement.
Secondary outcomes
No significant difference was found for the reduction of TAPSE, HHF, NT pro-BNP, mRAP, and mPAP (Fig. S2). This lack of statistical significance may be ascribable to the limited number of studies assessing these specific outcomes. Six studies evaluated PCWP difference; mean PCWP was reduced by 4.67 mmHg by LADd implantation (95% confidence interval: 7.59–1.75) (Fig. S3).
Safety outcomes
All studies examined device-related adverse events, defined as either device embolization, migration or removal, stroke (fatal and non-fatal) or transient ischemic attack, cardiac tamponade, and emergency cardiac surgery, as previously described. LADd implantation did not result in a significant higher risk of device-related adverse events (RR 2.54, 95% CI 0.93–6.96) (Fig. 7). When comparing the two RCT [17] on incidence of MACCRE, LADd implantation did not result in a significant higher risk of MACCRE (RR 0.85, 95% CI 0.45–1.59) (Fig. 7).
Publication bias and sensitivity analysis
Publication bias for all outcomes was assessed. The Egger’s tests revealed no publication bias when corrected for meta-regression analysis moderators (Table S3). To evaluate the consistency of the findings, sensitivity analyses were conducted on all outcomes and were stable (Fig. S4).
Discussion
The present meta-analysis of mostly non-randomized, open-label studies, by investigating the effect of different devices for left atrial decompression in 547 symptomatic patients with HF and a wide spectrum of EFs, showed that the procedure improves submaximal exercise and functional capacity assessed by 6MWD and NYHA class, and improves HRQoL. In particular, the 6MWD, a valuable index of submaximal exercise capacity, increased by approximately 15% and the NYHA class was reduced by approximately 30%. Moreover, device implantation showed a significant improvement of PCWP with no effects on other hemodynamic, prognostic, or echocardiographic endpoints (TAPSE, HHF, NT-proBNP levels, mRAP, and mPAP). There were no significant differences among devices in terms of 6MWD, NYHA class change or HRQoL improvement and device implantation was not related to significant adverse events.
LADd reduce left atrial pressure, by either creating a small artificial atrial septal defect or coronary sinus communication. The potential therapeutic benefit in HF patients of iatrogenic interatrial shunt starts from the observation that the presence of a congenital secundum atrial septal defect in patients with mitral stenosis (Lutembacher syndrome) seemed to be beneficial compared to isolated mitral stenosis, due to the ability to decompress left atrium by shunting blood to the right atrium and to the systemic veins; coronary sinus to left atrium devices similarly allow left atrium decompression, however, without the need of a septal puncture.
In all included studies, the procedural success rates were high for all LADd and most of them were patent at the end of the follow-up period (Table S4). Patients with both reduced and preserved EF were enrolled in the studies included in the present meta-analysis; in particular, the REDUCE LAP-HF II [23] concentrated on patients with preserved or mildly reduced EF, since no therapeutic approaches, apart from the recent efficacy reported for SGLT2i [2, 3], have demonstrated to improve soft and hard endpoints in this population. The pathophysiological substrate of LADd use in preserved and mildly reduced EFs starts from the concept that in HFpEF and in HF with mildly reduced EF patients, with normal or near-normal central venous pressure, but elevated left atrial pressure on efforts, left atrial decompression might be particularly beneficial to alleviate congestion and relief symptoms. However, HFpEF is a clinical entity that includes a great variety of patients with different phenotypes that often needs a personalized therapeutic approach; for this reason, many of the tested therapeutic approaches in this population failed to demonstrate a beneficial effect. Interestingly, our analysis showed that the effect of LADd on 6MWD, NYHA class and HRQoL was consistent across a wide spectrum of EFs with a greater effect in patients with lower EFs (Figs. 2, 4, and 6).
In contrast to previous meta-analysis on the same topic [10, 25], these results underline the efficacy of LAD approach in HFrEF and HFmrEF, with still some controversies in HFpEF, as already reported for most pharmacological approaches. In particular, Lauder et al. [25] reported an improvement in 6MWD, HRQoL, and PCWP, with better outcomes for 6MWD in patients with EF > 40%. In a subsequent meta-analysis, Yi and colleagues [10] reported that the improvement in PCWP was consistent in HF patients, with no difference regarding EF. The reason why a third meta-analysis on the topic was needed after the publication of the REDUCE LAP-HF II trial [23] is related to the clear interest in this specific subject among HF specialists and interventional cardiologists, and, especially, to some new elements that are present in our study. In particular, the inclusion of more recent trials, doubling the total number of patients compared to the aforementioned meta-analyses, and the use of EF as continuous and not dichotomic moderator in meta-regression analyses significantly impacted on meta-analyses results and study data interpretation; thus, adding these components, we demonstrated that the patient-centric outcomes improve across a wide range of EF, with better results in patients with lower EF.
Regarding quality of life, HRQoL score increased by 20.45 points; in general, a shift of 5 points is considered a small, but clinically significant change, while a change of more than 20 points is interpreted as a large significant change [26], as observed in our analysis. The HRQoL is drastically impaired in HF patients; in the studies included in the present meta-analysis, HRQoL was measured using either the MLHFQ or KCCQ, that are both robust and reliable disease specific HRQoL measurements in HF [27, 28]. The gain in HRQoL obtained after LADd implantation overcome those of most approved pharmacological HF treatments. In the DAPA-HF trial, dapagliflozin improved the KCCQ total symptom score by 7.0 points in HFrEF diabetic patients and by 5.4 points in non-diabetics at 8 months [29], whereas, in the recently published DELIVER trial [3], dapagliflozin improved the KCCQ total symptom score by 2.4 points at 8 months in HFpEF subjects. In the EMPEROR-Preserved [2] and EMPEROR-Reduced [30] trials, empagliflozin improved KCCQ by 4.5 points and 5.8 points at 12 months, respectively in HFpEF and HFrEF patients. Similar benefits on HRQoL were only recorded for transvenous edge-to-edge mitral valve repair of secondary mitral regurgitation, in which the KCQQ score increased by 12.50 points [31].
As regards safety concerns, despite different analyzed outcomes among the included studies, no differences in the occurrence of device-related adverse events were found.
Specifically, analyzing the reported adverse outcome in the REDUCE LAP-HF trial [16], at 12-month follow-up, one patient died for combined pneumonia and renal failure and one patient had a fatal stroke. In the REDUCE LAP-HF I trial [17], across the 12 months of follow-up, no device embolization, occlusion, or migration was observed; a second procedure for device removal or occlusion was never required, and no strokes, transient ischemic attacks or atrial fibrillation/atrial flutter events were reported in the device treated arm. The cumulative incidence of major adverse cardiac, cerebrovascular, and renal events was comparable in the device and in the control group (Log-rank p = 0.20). In the REDUCE LAP-HF II trial [17] the composite safety endpoint of cardiovascular mortality, non-fatal stroke, worsening kidney function, major cardiac events, thrombo-embolic complications, new persistent or permanent atrial fibrillation or atrial flutter, and ≥ 30% increase in right ventricular size or ≥ 30% decrease in TAPSE were not significantly different between the device and sham-controlled arms (p = 0.11). Rodes-Cabau et al. [18] reported only one patient experiencing a cardiac tamponade during the in-hospital stay, whereas Guimares et al. [19] reported one patient with advanced HF dying due to an electrical storm. In the PRELIEVE study [21] only one patient experienced post-procedural bleeding and syncope. In the studies by Del Trigo et al. [15], Simard et al. [20], Shang et al. [24], Malek et al. [14], and in the RAISE trial [22], no device-related adverse events were recorded at follow-up. In the present meta-analysis, we analyzed both a composite of averse-related outcomes and a composite of major adverse cardiovascular, cerebral, and renal events just considering the data from RTCs; for both safety outcomes, LADd implantation was neutral.
Thus, LADd implantation is a safe procedure and provide a functional benefit in HF patients, with better evidence in patients with lower EFs. However, despite the benefits of LADd therapy, the present findings should be interpreted with caution until verified by larger, appropriately powered, randomized trials examining the impact on hard endpoints in HFrEF.
Limitations
Our meta-analysis suffers from some limitations. First, most of the included studies were small, single-arm feasibility studies without control groups and with variable follow-up durations. Thus, we cannot rule out non-specific therapeutic effects, such as the placebo effect. Moreover, concerns may exist about the generalizability of our findings due to the limited sample size and considerable heterogeneity of some outcomes. Meta regression analysis according to EF proved to be a significant explanator for the reported heterogeneity. Although the improvements in clinical outcomes, such as 6MWD, NYHA class, and HRQoL are encouraging, they need to be validated by appropriately powered and, ideally, sham-controlled randomized trials. Several trials are now active, like the ALt FLOW US (NCT03523416) [32], RELIEVE-HF (NCT03499236) [33], and PROLONGER [34], recruiting patients with both HFrEF and HFpEF. Second, this meta-analysis is not based on individual patient data. Third, functional outcomes, including the 6MWD, are not simply reliant on cardiopulmonary conditions.
Conclusions
LADd implantation in HF patients is a feasible and safe procedure, associated with considerable improvements in patient-centric outcomes, such as submaximal exercise capacity, NYHA class, and HRQoL across a wide spectrum of EF, with better outcomes in patients with more impaired systolic function. These findings need confirmation by larger sham-controlled trials.
Availability of data and materials
All data and STATA.do files will be available at reasonable request to the corresponding author.
References
McDonagh TA, Metra M, Adamo M et al (2021) 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 42(36):3599–3726
Anker SD, Butler J, Filippatos G et al (2021) Empagliflozin in heart failure with a preserved ejection fraction. N Engl J Med 385(16):1451–1461
Solomon SD, McMurray JJV, Claggett B et al (2022) Dapagliflozin in heart failure with mildly reduced or preserved ejection fraction. N Engl J Med. [published online ahead of print, 2022 Aug 27]
Reddy YNV, Obokata M, Wiley B et al (2019) The haemodynamic basis of lung congestion during exercise in heart failure with preserved ejection fraction. Eur Heart J 40(45):3721–3730
Abraham WT, Stevenson LW, Bourge RC et al (2016) Sustained efficacy of pulmonary artery pressure to guide adjustment of chronic heart failure therapy: complete follow-up results from the CHAMPION randomised trial. Lancet 387(10017):453–461
Abraham WT (2021) Interatrial shunting for the treatment of heart failure: an on-demand, self-regulating left atrial pressure system. Eur J Heart Fail 23:811–813
Sambhi MP, Zimmerman HA (1958) Pathologic physiology of Lutembacher syndrome. Am J Cardiol 2(6):681–686
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71. https://doi.org/10.1136/bmj.n71
Brozek JL, Canelo-Aybar C, Akl EA et al (2021) GRADE Guidelines 30: the GRADE approach to assessing the certainty of modeled evidence-An overview in the context of health decision-making. J Clin Epidemiol 129:138–150
Yi T, Li M, Fan F et al (2022) Haemodynamic changes of interatrial shunting devices for heart failure: a systematic review and meta-analysis. ESC Heart Fail 9(3):1987–1995
Luo D, Wan X, Liu J, Tong T (2018) Optimally estimating the sample mean from the sample size, median, mid-range, and/or mid-quartile range. Stat Methods Med Res 27(6):1785–1805
Wan X, Wang W, Liu J, Tong T (2014) Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol 14:135
Shi J, Luo D, Weng H et al (2020) Optimally estimating the sample standard deviation from the five-number summary. Res Synth Methods 11(5):641–654
Malek F, Neuzil P, Gustafsson F et al (2015) Clinical outcome of transcatheter treatment of heart failure with preserved or mildly reduced ejection fraction using a novel implant. Int J Cardiol 187:227–228
Del Trigo M, Bergeron S, Bernier M et al (2016) Unidirectional left-to-right interatrial shunting for treatment of patients with heart failure with reduced ejection fraction: a safety and proof-of-principle cohort study. Lancet 387(10025):1290–1297
Kaye DM, Hasenfuß G, Neuzil P et al (2016) One-year outcomes after transcatheter insertion of an interatrial shunt device for the management of heart failure with preserved ejection fraction. Circ Heart Fail 9(12):e003662
Shah SJ, Feldman T, Ricciardi MJ et al (2018) One-year safety and clinical outcomes of a transcatheter interatrial shunt device for the treatment of heart failure with preserved ejection fraction in the reduce elevated left atrial pressure in patients with heart failure (REDUCE LAP-HF I) Trial. JAMA Cardiol 3(10):968–977
Rodés-Cabau J, Bernier M, Amat-Santos IJ et al (2018) Interatrial shunting for heart failure: early and late results from the first-in-human experience with the V-wave system. JACC Cardiovasc Interv 11(22):2300–2310
Guimarães L, Bergeron S, Bernier M et al (2020) Interatrial shunt with the second-generation V-Wave system for patients with advanced chronic heart failure. EuroIntervention 15(16):1426–1428
Simard T, Labinaz M, Zahr F et al (2020) Percutaneous atriotomy for levoatrial-to-coronary sinus shunting in symptomatic heart failure: first-in-human experience. JACC Cardiovasc Interv 13(10):1236–1247
Paitazoglou C, Bergmann MW, Özdemir R et al (2021) One-year results of the first-in-man study investigating the Atrial Flow Regulator for left atrial shunting in symptomatic heart failure patients: the PRELIEVE study. Eur J Heart Fail 23(5):800–810
Sun W, Zou H, Yong Y et al (2022) The RAISE trial: a novel device and first-in-man trial. Circ Heart Fail 15(4):e008362
Shah SJ, Borlaug BA, Chung ES et al (2022) Atrial shunt device for heart failure with preserved and mildly reduced ejection fraction (REDUCE LAP-HF II): a randomised, multicentre, blinded, sham-controlled trial. Lancet 399(10330):1130–1140
Shang X, Liu M, Zhong Y et al (2022) Clinical study on the treatment of chronic heart failure with a novel D-shant atrium shunt device. ESC Heart Fail 9(3):1713–1720
Lauder L, Pereira TV, Degenhardt MC et al (2021) Feasibility and efficacy of transcatheter interatrial shunt devices for chronic heart failure: a systematic review and meta-analysis. Eur J Heart Fail 23(11):1960–1970
Spertus J, Peterson E, Conard MW et al (2005) Monitoring clinical changes in patients with heart failure: a comparison of methods. Am Heart J 150(4):707–715
Spertus JA, Jones PG, Sandhu AT, Arnold SV (2020) Interpreting the Kansas City Cardiomyopathy Questionnaire in clinical trials and clinical care. J Am Coll Cardiol 76:2379–2390
Guyatt GH (1993) Measurement of health-related quality of life in heart failure. J Am Coll Cardiol 22:A185–A191
Kosiborod MN, Jhund PS, Docherty KF et al (2020) Effects of dapagliflozin on symptoms, function, and quality of life in patients with heart failure and reduced ejection fraction: results from the DAPA-HF trial. Circulation 141(2):90–99
Packer M, Anker SD, Butler J et al (2020) Cardiovascular and renal outcomes with empagliflozin in heart failure. N Engl J Med 383(15):1413–1424
Stone GW, Lindenfeld J, Abraham WT et al (2018) Transcatheter mitral-valve repair in patients with heart failure. N Engl J Med 379(24):2307–2318
Hibbert B, Zahr F, Simard T et al (2023) Left Atrial to Coronary Sinus Shunting for Treatment of Symptomatic Heart Failure. JACC Cardiovasc Interv S1936–8798(23):00571-X
Nunez-Villota J, Pfeiffer M, Eguchi S, Stone G, Lindenfeld J, Rodes-Cabau J, Anker S, Abraham W, Lee E, Bayes-Genis A, Litwin S, Laufer-Perl M, Boehmer J, Bax J, Gorcsan J on behalf of the RELIEVE-HF Study Group (2022) Improved left and right ventricular structure and function with the V-Wave Ventura Shunt Device in patients with HFrEF and HFpEF: 12-month echocardiographic results from the RELIEVE-HF roll-in cohort. ESC 365. https://esc365.escardio.org/presentation/252970
Lewicki Ł, Kosmalska K, Liedtke S et al (2020) Pomeranian atRial flOw reguLatOr iN conGestive hEart failuRe (PROLONGER): Study protocol. Cardiol J 27(6):879–883
Funding
Open access funding provided by Università degli Studi di Napoli Federico II within the CRUI-CARE Agreement. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
CB: conceptualization, methodology, writing, formal analysis. SP: writing, review and editing. PG: visualization. FM: data curation. SD: methodology. VA: writing. AA: visualization. FC: writing. SD: methodology. IE: writing. MFF: visualization. PPF: writing, review, and editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Competing interests
All authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Basile, C., Paolillo, S., Gargiulo, P. et al. Left atrium decompression devices across the spectrum of ejection fraction in heart failure: an updated systematic review and meta-regression analysis. Heart Fail Rev 28, 1151–1161 (2023). https://doi.org/10.1007/s10741-023-10317-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-023-10317-2