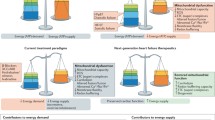
Abstract
Although currently employed therapies for heart failure decrease overall mortality and improve patient quality of life temporarily, the disease is known to progress even for patients who receive all guideline-recommended therapies. This indicates that our concise understanding of heart failure and of disease progression is incomplete, and there is a need for new interventions that may augment, or even supplant, currently available options. A literature review reveals that an exciting, novel area of current research is focused on mitochondria, which are uniquely juxtaposed at the sites of both generation of high-energy molecules and initiation of programmed cell death. Elamipretide is being studied both to maintain cellular biogenetics and prevent reactive oxygen species-induced cell damage by targeting and stabilizing the cardiolipin-cytochrome c supercomplex. Thus far, elamipretide has been shown to increase left ventricular ejection fraction in dog models of heart failure with reduced ejection fraction and to prevent left ventricular remodeling in rats. In early-phase clinical trials, elamipretide administration has not resulted in any severe adverse events, and it has shown promising improvements in cardiac hemodynamics at highest doses. Nonetheless, additional studies are necessary to describe the long-term safety and efficacy of elamipretide.


Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Konstam MA, Dracup K, Baker DW, Bottorff MB, Brooks NH, Dacey RA et al (1995) Heart failure: evaluation and care of patients with left ventricular systolic dysfunction. J Card Fail 1:183–187. https://doi.org/10.1016/1071-9164(95)90021-7
Mann DL (1999) Mechanisms and models in heart failure: a combinatorial approach. Circulation 100:999–1008. https://doi.org/10.1161/01.cir.100.9.999
Foody JM, Farrell MH, Krumholz HM (2002) Beta-blocker therapy in heart failure: scientific review. JAMA 287:883–889. https://doi.org/10.1001/jama.287.7.883
Yusuf S, Pitt B, Davis CE, Hood WB, Cohn JN, Nicklas JM et al (1992) Effect of enalapril on mortality and the development of heart failure in asymptomatic patients with reduced left ventricular ejection fractions. N Engl J Med 327:685–691. https://doi.org/10.1056/NEJM199209033271003
Flather MD, Yusuf S, Køber L, Pfeffer M, Hall A, Murray G et al (2000) Long-term ACE-inhibitor therapy in patients with heart failure or left-ventricular dysfunction: a systematic overview of data from individual patients. ACE-Inhibitor Myocardial Infarction Collaborative Group Lancet 355:1575–1581. https://doi.org/10.1016/s0140-6736(00)02212-1
Pitt B, Poole-Wilson PA, Segal R, Martinez FA, Dickstein K, Camm AJ et al (2000) Effect of losartan compared with captopril on mortality in patients with symptomatic heart failure: randomised trial–the Losartan Heart Failure Survival Study ELITE II. Lancet 355:1582–1587. https://doi.org/10.1016/s0140-6736(00)02213-3
Zannad F, McMurray JJV, Krum H, van Veldhuisen DJ, Swedberg K, Shi H et al (2011) Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med 364:11–21. https://doi.org/10.1056/NEJMoa1009492
McMurray JJV, Solomon SD, Inzucchi SD, Kober L, Kosiborod MN, Martinez FA et al (2019) Dapagliflozin and patients with heart failure and reduced ejection fraction. N Engl J Med 381:1995–2008. https://doi.org/10.1056/NEJMoa1911303
Paul S, Harshaw-Ellis K (2019) Evolving use of biomarkers in the management of heart failure. Cardiol Rev 27:153–159. https://doi.org/10.1097/CRD.0000000000000224
Yusuf S, Pitt B, Davis CE, Hood WB, Cohn JN, Nicklas JM et al (1991) Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. N Engl J Med 325:293–302. https://doi.org/10.1056/NEJM199108013250501
McMurray JJV, Packer M, Desai AS, Gong J, Lefkowitz MP, Rizkala AR et al (2014) Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med 371:993–1004. https://doi.org/10.1056/NEJMoa1409077
Nohria A, Lewis E, Stevenson LW (2002) Medical management of advanced heart failure. JAMA 287:628–640. https://doi.org/10.1001/jama.287.5.628
Dracup K, Baker DW, Dunbar SB, Dacey RA, Brooks NH, Johnson JC et al (1994) Management of heart failure. II. Counseling, education, and lifestyle modifications. JAMA 272:1442–6. https://doi.org/10.1001/jama.1994.03520180066037
Bristow MR, Saxon LA, Boehmer J, Krueger S, Kass DA, De Marco T et al (2004) Cardiac-resynchronization therapy with or without an implantable defibrillator in advanced chronic heart failure. N Engl J Med 350:2140–2150. https://doi.org/10.1056/NEJMoa032423
Ravichandran, PS. Mechanical circulatory support in a nutshell – American College of Cardiology 2015 Nov 13. https://www.acc.org/latest-in-cardiology/articles/2015/11/12/09/28/mechanical-circulatory-support-in-a-nutshell (accessed January 5, 2020)
Miller L, Birks E, Guglin M, Lamba H, Frazier OH (2019) Use of ventricular assist devices and heart transplantation for advanced heart failure. Circ Res 124:1658–1678. https://doi.org/10.1161/CIRCRESAHA.119.313574
Benjamin EJ, Muntner P, Alonso A, Bittencourt MS, Callaway CW, Carson AP et al (2019) Heart Disease and Stroke Statistics-2019 Update: a report from the American Heart Association. Circulation 139:e56-528. https://doi.org/10.1161/CIR.0000000000000659
Underlying Cause of Death, 1999–2017. https://wonder.cdc.gov/controller/datarequest/D76 (accessed January 4, 2020)
Neubauer S (2007) The failing heart–an engine out of fuel. N Engl J Med 356:1140–1151. https://doi.org/10.1056/NEJMra063052
Cooper GM (2000) Actin, myosin, and cell movement. The Cell: A Molecular Approach, 2nd Edition
Herrmann G, Decherd GM (1939) The chemical nature of heart failure. Ann Intern Med 12:1233. https://doi.org/10.7326/0003-4819-12-8-1233
Wollenberger A (1947) On the energy-rich phosphate supply of the failing heart. Am J Physiol 150:733–745. https://doi.org/10.1152/ajplegacy.1947.150.4.733
Olson RE (1959) Myocardial metabolism in congestive heart failure. J Chronic Dis 9:442–464. https://doi.org/10.1016/0021-9681(59)90172-9
Ingwall JS, Weiss RG (2004) Is the failing heart energy starved? On using chemical energy to support cardiac function. Circ Res 95:135–145. https://doi.org/10.1161/01.RES.0000137170.41939.d9
Taegtmeyer H (2000) Metabolism–the lost child of cardiology. J Am Coll Cardiol 36:1386–1388
Taegtmeyer H (2004) Cardiac metabolism as a target for the treatment of heart failure. Circulation 110:894–896. https://doi.org/10.1161/01.CIR.0000139340.88769.D5
Stanley WC, Recchia FA, Lopaschuk GD (2005) Myocardial substrate metabolism in the normal and failing heart. Physiol Rev 85:1093–1129. https://doi.org/10.1152/physrev.00006.2004
Ventura-Clapier R, Garnier A, Veksler V (2004) Energy metabolism in heart failure. J Physiol (Lond) 555:1–13. https://doi.org/10.1113/jphysiol.2003.055095
Dalal JJ, Mishra S (2017) Modulation of myocardial energetics: an important category of agents in the multimodal treatment of coronary artery disease and heart failure. Indian Heart J 69:393–401. https://doi.org/10.1016/j.ihj.2017.04.001
Lopaschuk GD, Rebeyka IM, Allard MF (2002) Metabolic modulation: a means to mend a broken heart. Circulation 105:140–142
Morrow DA, Givertz MM (2005) Modulation of myocardial energetics: emerging evidence for a therapeutic target in cardiovascular disease. Circulation 112:3218–3221. https://doi.org/10.1161/CIRCULATIONAHA.105.581819
Essop MF, Opie LH (2004) Metabolic therapy for heart failure. Eur Heart J 25:1765–1768. https://doi.org/10.1016/j.ehj.2004.08.019
van Bilsen M, Smeets PJH, Gilde AJ, van der Vusse GJ (2004) Metabolic remodeling of the failing heart: the cardiac burn-out syndrome? Cardiovasc Res 61:218–226. https://doi.org/10.1016/j.cardiores.2003.11.014
Münzel T, Camici GG, Maack C, Bonetti NR, Fuster V, Kovacic JC (2017) Impact of oxidative stress on the heart and vasculature: Part 2 of a 3-Part Series. J Am Coll Cardiol 70:212–229. https://doi.org/10.1016/j.jacc.2017.05.035
Bertero E, Maack C (2018) Metabolic remodelling in heart failure. Nat Rev Cardiol 15:457–470. https://doi.org/10.1038/s41569-018-0044-6
Folkers K, Wolaniuk J, Simonsen R, Morishita M, Vadhanavikit S (1985) Biochemical rationale and the cardiac response of patients with muscle disease to therapy with coenzyme Q10. Proc Natl Acad Sci USA 82:4513–4516. https://doi.org/10.1073/pnas.82.13.4513
Mortensen SA, Rosenfeldt F, Kumar A, Dolliner P, Filipiak KJ, Pella D et al (2014) The effect of coenzyme Q10 on morbidity and mortality in chronic heart failure: results from Q-SYMBIO: a randomized double-blind trial. JACC Heart Fail 2:641–649. https://doi.org/10.1016/j.jchf.2014.06.008
Smith RAJ, Hartley RC, Cochemé HM, Murphy MP (2012) Mitochondrial pharmacology. Trends Pharmacol Sci 33:341–352. https://doi.org/10.1016/j.tips.2012.03.010
Szeto HH (2014) First-in-class cardiolipin-protective compound as a therapeutic agent to restore mitochondrial bioenergetics. Br J Pharmacol 171:2029–2050. https://doi.org/10.1111/bph.12461
Robin ED, Wong R (1988) Mitochondrial DNA molecules and virtual number of mitochondria per cell in mammalian cells. J Cell Physiol 136:507–513. https://doi.org/10.1002/jcp.1041360316
Schlame M, Ren M (2009) The role of cardiolipin in the structural organization of mitochondrial membranes. Biochim Biophys Acta 1788:2080–2083. https://doi.org/10.1016/j.bbamem.2009.04.019
Schug ZT, Gottlieb E (2009) Cardiolipin acts as a mitochondrial signalling platform to launch apoptosis. Biochim Biophys Acta 1788:2022–2031. https://doi.org/10.1016/j.bbamem.2009.05.004
Sorice M, Manganelli V, Matarrese P, Tinari A, Misasi R, Malorni W et al (2009) Cardiolipin-enriched raft-like microdomains are essential activating platforms for apoptotic signals on mitochondria. FEBS Lett 583:2447–2450. https://doi.org/10.1016/j.febslet.2009.07.018
Paradies G, Petrosillo G, Paradies V, Ruggiero FM (2010) Oxidative stress, mitochondrial bioenergetics, and cardiolipin in aging. Free Radic Biol Med 48:1286–1295. https://doi.org/10.1016/j.freeradbiomed.2010.02.020
Nickel A, Kohlhaas M, Maack C (2014) Mitochondrial reactive oxygen species production and elimination. J Mol Cell Cardiol 73:26–33. https://doi.org/10.1016/j.yjmcc.2014.03.011
Biala AK, Kirshenbaum LA (2014) The interplay between cell death signaling pathways in the heart. Trends Cardiovasc Med 24:325–331. https://doi.org/10.1016/j.tcm.2014.08.002
Han X, Yang J, Cheng H, Yang K, Abendschein DR, Gross RW (2005) Shotgun lipidomics identifies cardiolipin depletion in diabetic myocardium linking altered substrate utilization with mitochondrial dysfunction. Biochemistry 44:16684–16694. https://doi.org/10.1021/bi051908a
Chicco AJ, Sparagna GC (2007) Role of cardiolipin alterations in mitochondrial dysfunction and disease. Am J Physiol, Cell Physiol 292:C33-44. https://doi.org/10.1152/ajpcell.00243.2006
Shi Y (2010) Emerging roles of cardiolipin remodeling in mitochondrial dysfunction associated with diabetes, obesity, and cardiovascular diseases. J Biomed Res 24:6–15. https://doi.org/10.1016/S1674-8301(10)60003-6
Claypool SM, Koehler CM (2012) The complexity of cardiolipin in health and disease. Trends Biochem Sci 37:32–41. https://doi.org/10.1016/j.tibs.2011.09.003
Ikon N, Ryan RO (2017) Barth syndrome: connecting cardiolipin to cardiomyopathy. Lipids 52:99–108. https://doi.org/10.1007/s11745-016-4229-7
Dey S, DeMazumder D, Sidor A, Foster DB, O’Rourke B (2018) Mitochondrial ROS drive sudden cardiac death and chronic proteome remodeling in heart failure. Circ Res 123:356–371. https://doi.org/10.1161/CIRCRESAHA.118.312708
Errami M, Galindo CL, Tassa AT, Dimaio JM, Hill JA, Garner HR (2008) Doxycycline attenuates isoproterenol- and transverse aortic banding-induced cardiac hypertrophy in mice. J Pharmacol Exp Ther 324:1196–1203. https://doi.org/10.1124/jpet.107.133975
Hori Y, Kunihiro S, Sato S, Yoshioka K, Hara Y, Kanai K et al (2009) Doxycycline attenuates isoproterenol-induced myocardial fibrosis and matrix metalloproteinase activity in rats. Biol Pharm Bull 32:1678–1682. https://doi.org/10.1248/bpb.32.1678
Schulze CJ, Castro MM, Kandasamy AD, Cena J, Bryden C, Wang SH et al (2013) Doxycycline reduces cardiac matrix metalloproteinase-2 activity but does not ameliorate myocardial dysfunction during reperfusion in coronary artery bypass patients undergoing cardiopulmonary bypass. Crit Care Med 41:2512–2520. https://doi.org/10.1097/CCM.0b013e318292373c
Riba A, Deres L, Eros K, Szabo A, Magyar K, Sumegi B et al (2017) Doxycycline protects against ROS-induced mitochondrial fragmentation and ISO-induced heart failure. PLoS ONE 12:e0175195. https://doi.org/10.1371/journal.pone.0175195
Marín-García J, Akhmedov AT, Moe GW (2013) Mitochondria in heart failure: the emerging role of mitochondrial dynamics. Heart Fail Rev 18:439–456. https://doi.org/10.1007/s10741-012-9330-2
Ong S-B, Subrayan S, Lim SY, Yellon DM, Davidson SM, Hausenloy DJ (2010) Inhibiting mitochondrial fission protects the heart against ischemia/reperfusion injury. Circulation 121:2012–2022. https://doi.org/10.1161/CIRCULATIONAHA.109.906610
Elewa MAF, Al-Gayyar MM, Schaalan MF, Abd El Galil KH, Ebrahim MA, El-Shishtawy MM (2015) Hepatoprotective and anti-tumor effects of targeting MMP-9 in hepatocellular carcinoma and its relation to vascular invasion markers. Clin Exp Metastasis 32:479–93. https://doi.org/10.1007/s10585-015-9721-6
Griffin MO, Fricovsky E, Ceballos G, Villarreal F (2010) Tetracyclines: a pleitropic family of compounds with promising therapeutic properties. Review of the literature. Am J Physiol, Cell Physiol 299:C539–48.https://doi.org/10.1152/ajpcell.00047.2010
Reily C, Mitchell T, Chacko BK, Benavides G, Murphy MP, Darley-Usmar V (2013) Mitochondrially targeted compounds and their impact on cellular bioenergetics. Redox Biol 1:86–93. https://doi.org/10.1016/j.redox.2012.11.009
Sabbah HN, Wang M, Zhang K, Gupta RC, Rastogi S (2012) Acute Intravenous infusion of Bendavia (MTP-131), a novel mitochondria-targeting peptide, improves left ventricular systolic function in dogs with advanced heart failure circulation. 126:A15385
Sabbah HN, Gupta RC, Kohli S, Wang M, Hachem S, Zhang K (2016) Chronic therapy with elamipretide (MTP-131), a novel mitochondria-targeting peptide, improves left ventricular and mitochondrial function in dogs with advanced heart failure. Circ Heart Fail 9:e002206. https://doi.org/10.1161/CIRCHEARTFAILURE.115.002206
Dai W, Shi J, Gupta RC, Sabbah HN, Hale SL, Kloner RA (2014) Bendavia, a mitochondria-targeting peptide, improves postinfarction cardiac function, prevents adverse left ventricular remodeling, and restores mitochondria-related gene expression in rats. J Cardiovasc Pharmacol 64:543–553. https://doi.org/10.1097/FJC.0000000000000155
Daubert MA, Yow E, Dunn G, Marchev S, Barnhart H, Douglas PS et al (2017) Novel mitochondria-targeting peptide in heart failure treatment: a randomized, placebo-controlled trial of elamipretide. Circ Heart Fail 10. https://doi.org/10.1161/CIRCHEARTFAILURE.117.004389
Zhao K, Luo G, Zhao G-M, Schiller PW, Szeto HH (2003) Transcellular transport of a highly polar 3+ net charge opioid tetrapeptide. J Pharmacol Exp Ther 304:425–432. https://doi.org/10.1124/jpet.102.040147
Zhao K, Zhao G-M, Wu D, Soong Y, Birk AV, Schiller PW et al (2004) Cell-permeable peptide antioxidants targeted to inner mitochondrial membrane inhibit mitochondrial swelling, oxidative cell death, and reperfusion injury. J Biol Chem 279:34682–34690. https://doi.org/10.1074/jbc.M402999200
Birk AV, Liu S, Soong Y, Mills W, Singh P, Warren JD et al (2013) The mitochondrial-targeted compound SS-31 re-energizes ischemic mitochondria by interacting with cardiolipin. J Am Soc Nephrol 24:1250–1261. https://doi.org/10.1681/ASN.2012121216
Gibson CM, Giugliano RP, Kloner RA, Bode C, Tendera M, Jánosi A et al (2016) EMBRACE STEMI study: a Phase 2a trial to evaluate the safety, tolerability, and efficacy of intravenous MTP-131 on reperfusion injury in patients undergoing primary percutaneous coronary intervention. Eur Heart J 37:1296–1303. https://doi.org/10.1093/eurheartj/ehv597
Armstrong PW, Pieske B, Anstrom KJ, Ezekowitz J, Hernandez AF, Butler J et al (2020) Vericiguat in patients with heart failure and reduced ejection fraction. N Engl J Med 382:1883–1893. https://doi.org/10.1056/NEJMoa1915928
Teerlink JR, Diaz R, Felker M, McMurray JJV, Metra M, Solomon SD et al (2021) Cardiac myosin activation with omecamtiv mecarbil in systolic heart failure. N Engl J Med 384:105–116. https://doi.org/10.1056/NEJMoa2025797
ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). Identifier NCT0278874, Effect of elamipretide on left ventricular function in subjects with stable heart failure and reduced ejection fraction. Cited 2021 4 Nov. Available at https://clinicaltrials.gov/ct2/show/study/NCT02788747?term=elamipretide&cond=Heart+Failure&draw=2&rank=3
ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). Identifier NCT0291466, A phase 2 study to evaluate the cardiac and renal effects of short-term treatment with elamipretide in patients hospitalized with congestion due to heart failure. Cited 2021 4 Nov. Available at https://clinicaltrials.gov/ct2/show/study/NCT02914665?term=elamipretide&cond=Heart+Failure&draw=2&rank=2
Acknowledgements
The authors would like to thank the Departments of Internal Medicine and Cardiology at the Donald and Barbara Zucker School of Medicine at Hofstra/Northwell for their support.
Author information
Authors and Affiliations
Contributions
All authors contributed to study conception and design. CO, AS, and GH drafted the original manuscript. All authors edited and commented on all versions of the manuscript. GH and AA provided administrative support and supervision. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Obi, C., Smith, A.T., Hughes, G.J. et al. Targeting mitochondrial dysfunction with elamipretide. Heart Fail Rev 27, 1925–1932 (2022). https://doi.org/10.1007/s10741-021-10199-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-021-10199-2