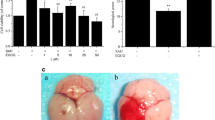
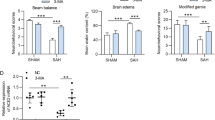
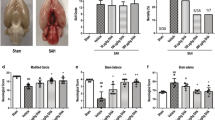
Abstract
ATG5-induced autophagy is triggered in the early stages after SAH, which plays a vital role in subarachnoid hemorrhage (SAH). Acyl-CoA synthetase short-chain family 2 (ACSS2) is not just involved in energy metabolism but also binds to TEFB to form a complex translocated to related autophagy genes to regulate the expression of autophagy-related genes. However, the contribution of ACSS2 to the activation of autophagy in early brain injury (EBI) after SAH has barely been discussed. The purpose of this study was to investigate the alterations of ACSS2 and its neuroprotective effects following SAH. We first evaluated the expression of ACSS2 at different time points (6, 12, 24, and 72 h after SAH) in vivo and primary cortical neurons stimulated by oxyhemoglobin (OxyHb). Subsequently, adeno-associated virus and lentivirus were used to regulate ACSS2 expression to investigate the effect of ACSS2 after SAH. The results showed that the ACSS2 level decreased significantly in the early stages of SAH and was minimized at 24 h post-SAH. After artificial intervention to overexpress ACSS2, ATG5-induced autophagy was further enhanced in EBI after SAH, and neuronal apoptosis was alleviated to protect brain injury. In addition, brain edema and neurological function scores were improved. These results suggest that ACSS2 plays an important role in the neuroprotection against EBI after SAH by increasing ATG5-induce autophagy and inhibiting apoptosis.






Similar content being viewed by others
Abbreviations
- ACSS2:
-
Acyl-CoA synthetase short-chain family 2
- ATG5:
-
Autophagy protein 5
- SAH:
-
Subarachnoid hemorrhage
- EBI:
-
Early brain injury
References
Cao S, Shrestha S, Li J, Yu X, Chen J, Yan F, Ying G, Gu C et al (2017) Melatonin-mediated mitophagy protects against early brain injury after subarachnoid hemorrhage through inhibition of NLRP3 inflammasome activation. Sci Rep 7:2417. https://doi.org/10.1038/s41598-017-02679-z
Chen J, Wang L, Wu C, Hu Q, Gu C, Yan F, Li J, Yan W et al (2014) Melatonin-enhanced autophagy protects against neural apoptosis via a mitochondrial pathway in early brain injury following a subarachnoid hemorrhage. J Pineal Res 56:12–19. https://doi.org/10.1111/jpi.12086
Dikic I, Elazar Z (2018) Mechanism and medical implications of mammalian autophagy. Nat Rev Mol Cell Biol 19:349–364. https://doi.org/10.1038/s41580-018-0003-4
Galluzzi L, Baehrecke EH, Ballabio A, Boya P, Bravo-San Pedro JM, Cecconi F, Choi AM, Chu CT et al (2017) Molecular definitions of autophagy and related processes. EMBO J 36:1811–1836. https://doi.org/10.15252/embj.201796697
Helbok R, Schiefecker AJ, Beer R, Dietmann A, Antunes AP, Sohm F, Fischer M, Hackl WO et al (2015) Early brain injury after aneurysmal subarachnoid hemorrhage: a multimodal neuromonitoring study. Crit Care 19:75. https://doi.org/10.1186/s13054-015-0809-9
Ho WM, Akyol O, Reis H, Reis C, McBride D, Thome C, Zhang J (2018) Autophagy after subarachnoid hemorrhage: can cell death be good? Curr Neuropharmacol 16:1314–1319. https://doi.org/10.2174/1570159X15666171123200646
Jing CH, Wang L, Liu PP, Wu C, Ruan D, Chen G (2012) Autophagy activation is associated with neuroprotection against apoptosis via a mitochondrial pathway in a rat model of subarachnoid hemorrhage. Neuroscience 213:144–153. https://doi.org/10.1016/j.neuroscience.2012.03.055
Lein ES, Hawrylycz MJ, Ao N, Ayres M, Bensinger A, Bernard A, Boe AF, Boguski MS et al (2007) Genome-wide atlas of gene expression in the adult mouse brain. Nature 445:168–176. https://doi.org/10.1038/nature05453
Li X, Yu W, Qian X, Xia Y, Zheng Y, Lee JH, Li W, Lyu J et al (2017) Nucleus-translocated ACSS2 promotes gene transcription for lysosomal biogenesis and autophagy. Mol Cell 66:684-697e689. https://doi.org/10.1016/j.molcel.2017.04.026
Liang Y, Pi H, Liao L, Tan M, Deng P, Yue Y, Xi Y, Tian L et al (2021) Cadmium promotes breast cancer cell proliferation, migration and invasion by inhibiting ACSS2/ATG5-mediated autophagy. Environ Pollut 273:116504. https://doi.org/10.1016/j.envpol.2021.116504
Menzies FM, Fleming A, Caricasole A, Bento CF, Andrews SP, Ashkenazi A, Fullgrabe J, Jackson A et al (2017) Autophagy and neurodegeneration: pathogenic mechanisms and therapeutic opportunities. Neuron 93:1015–1034. https://doi.org/10.1016/j.neuron.2017.01.022
Mews P, Donahue G, Drake AM, Luczak V, Abel T, Berger SL (2017) Acetyl-CoA synthetase regulates histone acetylation and hippocampal memory. Nature 546:381–386. https://doi.org/10.1038/nature22405
Mews P, Egervari G, Nativio R, Sidoli S, Donahue G, Lombroso SI, Alexander DC, Riesche SL et al (2019) Alcohol metabolism contributes to brain histone acetylation. Nature 574:717–721. https://doi.org/10.1038/s41586-019-1700-7
Mizushima N (2020) The ATG conjugation systems in autophagy. Curr Opin Cell Biol 63:1–10. https://doi.org/10.1016/j.ceb.2019.12.001
Moffett JR, Puthillathu N, Vengilote R, Jaworski DM, Namboodiri AM (2020) Acetate revisited: a key biomolecule at the nexus of metabolism, epigenetics, and oncogenesis—part 2: acetate and ACSS2 in health and disease. Front Physiol 11:580171. https://doi.org/10.3389/fphys.2020.580171
Neifert SN, Chapman EK, Martini ML, Shuman WH, Schupper AJ, Oermann EK, Mocco J, Macdonald RL (2021) Aneurysmal subarachnoid hemorrhage: the last decade. Transl Stroke Res 12:428–446. https://doi.org/10.1007/s12975-020-00867-0
Nishida Y, Arakawa S, Fujitani K, Yamaguchi H, Mizuta T, Kanaseki T, Komatsu M, Otsu K et al (2009) Discovery of Atg5/Atg7-independent alternative macroautophagy. Nature 461:654–658. https://doi.org/10.1038/nature08455
Qu XF, Liang TY, Wu DG, Lai NS, Deng RM, Ma C, Li X, Li HY et al (2021) Acyl-CoA synthetase long chain family member 4 plays detrimental role in early brain injury after subarachnoid hemorrhage in rats by inducing ferroptosis. CNS Neurosci Ther 27:449–463. https://doi.org/10.1111/cns.13548
Rass V, Helbok R (2019) Early brain injury after poor-grade subarachnoid hemorrhage. Curr Neurol Neurosci Rep 19:78. https://doi.org/10.1007/s11910-019-0990-3
Runwal G, Stamatakou E, Siddiqi FH, Puri C, Zhu Y, Rubinsztein DC (2019) LC3-positive structures are prominent in autophagy-deficient cells. Sci Rep 9:10147. https://doi.org/10.1038/s41598-019-46657-z
Sarkar C, Zhao Z, Aungst S, Sabirzhanov B, Faden AI, Lipinski MM (2014) Impaired autophagy flux is associated with neuronal cell death after traumatic brain injury. Autophagy 10:2208–2222. https://doi.org/10.4161/15548627.2014.981787
Scrivo A, Bourdenx M, Pampliega O, Cuervo AM (2018) Selective autophagy as a potential therapeutic target for neurodegenerative disorders. Lancet Neurol 17:802–815. https://doi.org/10.1016/s1474-4422(18)30238-2
Shao A, Wang Z, Wu H, Dong X, Li Y, Tu S, Tang J, Zhao M et al (2016) Enhancement of autophagy by histone deacetylase inhibitor trichostatin A ameliorates neuronal apoptosis after subarachnoid hemorrhage in rats. Mol Neurobiol 53:18–27. https://doi.org/10.1007/s12035-014-8986-0
Shi L, Liang F, Zheng J, Zhou K, Chen S, Yu J, Zhang J (2018) Melatonin regulates apoptosis and autophagy via ROS-MST1 pathway in subarachnoid hemorrhage. Front Mol Neurosci 11:93. https://doi.org/10.3389/fnmol.2018.00093
Stavoe AKH, Holzbaur ELF (2019) Autophagy in neurons. Annu Rev Cell Dev Biol 35:477–500. https://doi.org/10.1146/annurev-cellbio-100818-125242
Sugawara T, Ayer R, Jadhav V, Zhang JH (2008) A new grading system evaluating bleeding scale in filament perforation subarachnoid hemorrhage rat model. J Neurosci Methods 167:327–334. https://doi.org/10.1016/j.jneumeth.2007.08.004
Sun CM, Enkhjargal B, Reis C, Zhou KR, Xie ZY, Wu LY, Zhang TY, Zhu QQ et al (2019) Osteopontin attenuates early brain injury through regulating autophagy-apoptosis interaction after subarachnoid hemorrhage in rats. CNS Neurosci Ther 25:1162–1172. https://doi.org/10.1111/cns.13199
Wang Z, Shi XY, Yin J, Zuo G, Zhang J, Chen G (2012) Role of autophagy in early brain injury after experimental subarachnoid hemorrhage. J Mol Neurosci 46:192–202. https://doi.org/10.1007/s12031-011-9575-6
Xu H, Luo J, Ma G, Zhang X, Yao D, Li M, Loor JJ (2018) Acyl-CoA synthetase short-chain family member 2 (ACSS2) is regulated by SREBP-1 and plays a role in fatty acid synthesis in caprine mammary epithelial cells. J Cell Physiol 233:1005–1016. https://doi.org/10.1002/jcp.25954
Yin Y, Sun G, Li E, Kiselyov K, Sun D (2017) ER stress and impaired autophagy flux in neuronal degeneration and brain injury. Ageing Res Rev 34:3–14. https://doi.org/10.1016/j.arr.2016.08.008
Yuan B, Zhou XM, You ZQ, Xu WD, Fan JM, Chen SJ, Han YL, Wu Q et al (2020) Inhibition of AIM2 inflammasome activation alleviates GSDMD-induced pyroptosis in early brain injury after subarachnoid haemorrhage. Cell Death Dis 11:76. https://doi.org/10.1038/s41419-020-2248-z
Zhang XS, Wu Q, Wu LY, Ye ZN, Jiang TW, Li W, Zhuang Z, Zhou ML et al (2016) Sirtuin 1 activation protects against early brain injury after experimental subarachnoid hemorrhage in rats. Cell Death Dis 7:e2416. https://doi.org/10.1038/cddis.2016.292
Zhang X, Wu Q, Zhang Q, Lu Y, Liu J, Li W, Lv S, Zhou M et al (2017) Resveratrol attenuates early brain injury after experimental subarachnoid hemorrhage via inhibition of NLRP3 inflammasome activation. Front Neurosci 11:611. https://doi.org/10.3389/fnins.2017.00611
Zhang X, Lu Y, Wu Q, Dai H, Li W, Lv S, Zhou X, Zhang X et al (2019) Astaxanthin mitigates subarachnoid hemorrhage injury primarily by increasing sirtuin 1 and inhibiting the Toll-like receptor 4 signaling pathway. FASEB J 33:722–737. https://doi.org/10.1096/fj.201800642RR
Zhao H, Ji Z, Tang D, Yan C, Zhao W, Gao C (2013) Role of autophagy in early brain injury after subarachnoid hemorrhage in rats. Mol Biol Rep 40:819–827. https://doi.org/10.1007/s11033-012-2120-z
Zhou X, Wu Q, Lu Y, Zhang X, Lv S, Shao J, Zhou Y, Chen J et al (2019) Crosstalk between soluble PDGF-BB and PDGFRbeta promotes astrocytic activation and synaptic recovery in the hippocampus after subarachnoid hemorrhage. FASEB J 33:9588–9601. https://doi.org/10.1096/fj.201900195R
Funding
This work was grant-supported by the Jiangsu Natural Science Foundation of China (Grant No. SBK2019022915), the National Natural Science Foundation of China (Grant No. 82071328), Jiangsu Medical Priority Talent Project of China (Grant No. ZDRCA2016094) and Jiangsu Health Commission Scientific Research Key Project of China (Grant No. K2019017).
Author information
Authors and Affiliations
Contributions
WH, XZ: Conceptualizations and methodology. WH, LZ, RZ: Investigation. WH, QW, ZZ: Data analysis. WH, CD: Writing—original draft. XZ, XZ: Writing—review & editing. XZ: Resources. All authors of this article have given their permission to publish the article.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no potential conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
He, W., Zhou, X., Wu, Q. et al. Acetyl CoA synthase 2 potentiates ATG5-induced autophagy against neuronal apoptosis after subarachnoid hemorrhage. J Mol Histol 53, 511–521 (2022). https://doi.org/10.1007/s10735-022-10057-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-022-10057-x