Abstract
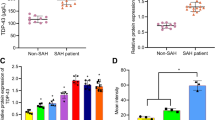
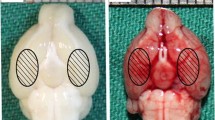
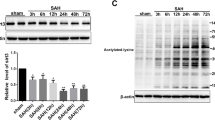
Trichostatin A (TSA), a pan-histone deacetylase inhibitor, exerts multiple neuroprotective properties. This study aims to examine whether TSA could enhance autophagy, thereby reduce neuronal apoptosis and ultimately attenuate early brain injury (EBI) following subarachnoid hemorrhage (SAH). SAH was performed through endovascular perforation method, and mortality, neurological score, and brain water content were evaluated at 24 h after surgery. Western blot were used for quantification of acetylated histone H3, LC3-II, LC3-I, Beclin-1, cytochrome c, Bax, and cleaved caspase-3 expression. Immunofluorescence was performed for colocalization of Beclin-1 and neuronal nuclei (NeuN). Apoptotic cell death of neurons was quantified with double staining of terminal deoxynucleotidyl transferase-mediated uridine 5′-triphosphate-biotin nick end-labeling (TUNEL) and NeuN. The autophagy inhibitor 3-methyladenine (3-MA) was used to manipulate the proposed pathway. Our results demonstrated that TSA reduced brain edema and alleviated neurological deficits at 24 h after SAH. TSA significantly increased acetylated histone H3, the LC3-II/LC3-I ratio, and Beclin-1 while decreased Bax and cleaved caspase-3 in the cortex. Beclin-1 and NeuN, TUNEL, and NeuN, respectively, were colocalized in cortical cells. Neuronal apoptosis in the ipsilateral basal cortex was significantly inhibited after TSA treatment. Conversely, 3-MA reversed the beneficial effects of TSA. These results proposed that TSA administration enhanced autophagy, which contributes to alleviation of neuronal apoptosis, improvement of neurological function, and attenuation of EBI following SAH.







Similar content being viewed by others
Abbreviations
- SAH:
-
Subarachnoid hemorrhage
- TSA:
-
Trichostatin A
- EBI:
-
Early brain injury
- HDAC:
-
Histone deacetylase
- 3-MA:
-
3-Methyladenine
- TUNEL:
-
Terminal deoxynucleotidyl transferase-mediated uridine 5′-triphosphate-biotin nick end-labeling
- LC3:
-
Light chain 3
- Apaf-1:
-
Apoptotic protease-activating factor 1
- Ac-H3:
-
Acetylated histones H3
- CC3:
-
Cleaved caspase-3
References
Sehba FA, Hou J, Pluta RM et al (2012) The importance of early brain injury after subarachnoid hemorrhage. Prog Neurobiol 97:14–37
Chen S, Feng H, Sherchan P et al (2014) Controversies and evolving new mechanisms in subarachnoid hemorrhage. Prog Neurobiol 115:64–91
Cahill J, Calvert JW, Zhang JH (2006) Mechanisms of early brain injury after subarachnoid hemorrhage. J Cereb Blood Flow Metab 26:1341–53
Rubinsztein DC, Codogno P, Levine B (2012) Autophagy modulation as a potential therapeutic target for diverse diseases. Nat Rev Drug Discov 11:709–30
Lee JY, He Y, Sagher O et al (2009) Activated autophagy pathway in experimental subarachnoid hemorrhage. Brain Res 1287:126–35
Wang Z, Shi XY, Yin J et al (2012) Role of autophagy in early brain injury after experimental subarachnoid hemorrhage. J Mol Neurosci 46:192–202
Liu Y, Li J, Wang Z et al (2014) Attenuation of early brain injury and learning deficits following experimental subarachnoid hemorrhage secondary to cystatin C: possible involvement of the autophagy pathway. Mol Neurobiol 49:1043–54
Jing CH, Wang L, Liu PP et al (2012) Autophagy activation is associated with neuroprotection against apoptosis via a mitochondrial pathway in a rat model of subarachnoid hemorrhage. Neuroscience 213:144–53
Chen J, Wang L, Wu C et al (2014) Melatonin-enhanced autophagy protects against neural apoptosis via a mitochondrial pathway in early brain injury following a subarachnoid hemorrhage. J Pineal Res 56:12–9
Grunstein M (1997) Histone acetylation in chromatin structure and transcription. Nature 389:349–52
Shein NA, Shohami E (2011) Histone deacetylase inhibitors as therapeutic agents for acute central nervous system injuries. Mol Med 17:448–56
Hahnen E, Hauke J, Trankle C et al (2008) Histone deacetylase inhibitors: possible implications for neurodegenerative disorders. Expert Opin Investig Drugs 17:169–84
Leng Y, Chuang DM (2006) Endogenous alpha-synuclein is induced by valproic acid through histone deacetylase inhibition and participates in neuroprotection against glutamate-induced excitotoxicity. J Neurosci 26:7502–12
Chen PS, Wang CC, Bortner CD et al (2007) Valproic acid and other histone deacetylase inhibitors induce microglial apoptosis and attenuate lipopolysaccharide-induced dopaminergic neurotoxicity. Neuroscience 149:203–12
Meisel A, Harms C, Yildirim F et al (2006) Inhibition of histone deacetylation protects wild-type but not gelsolin-deficient neurons from oxygen/glucose deprivation. J Neurochem 98:1019–31
Maruoka H, Sasaya H, Sugihara K et al (2011) Low-molecular-weight compounds having neurotrophic activity in cultured PC12 cells and neurons. J Biochem 150:473–5
Marinova Z, Ren M, Wendland JR et al (2009) Valproic acid induces functional heat-shock protein 70 via Class I histone deacetylase inhibition in cortical neurons: a potential role of Sp1 acetylation. J Neurochem 111:976–87
Francisco R, Perez-Perarnau A, Cortes C et al (2012) Histone deacetylase inhibition induces apoptosis and autophagy in human neuroblastoma cells. Cancer Lett 318:42–52
Xie M, Kong Y, Tan W et al (2014) HDAC inhibition blunts ischemia/reperfusion injury by inducing cardiomyocyte autophagy. Circulation 129:1139–51
Gammoh N, Lam D, Puente C et al (2012) Role of autophagy in histone deacetylase inhibitor-induced apoptotic and nonapoptotic cell death. Proc Natl Acad Sci U S A 109:6561–5
Chen S, Ma Q, Krafft PR et al (2013) P2X7 receptor antagonism inhibits p38 mitogen-activated protein kinase activation and ameliorates neuronal apoptosis after subarachnoid hemorrhage in rats. Crit Care Med 41:e466–74
Garcia JH, Wagner S, Liu KF et al (1995) Neurological deficit and extent of neuronal necrosis attributable to middle cerebral artery occlusion in rats. Statistical validation. Stroke 26:627–34, discussion 35
Sugawara T, Ayer R, Jadhav V et al (2008) A new grading system evaluating bleeding scale in filament perforation subarachnoid hemorrhage rat model. J Neurosci Methods 167:327–34
Zhou F, Chen G, Zhang J (2009) Edaravone reduces brain oedema and attenuates cell death after intracerebral haemorrhage in mice. Brain Inj 23:353–7
Chen S, Ma Q, Krafft PR et al (2013) P2X7R/cryopyrin inflammasome axis inhibition reduces neuroinflammation after SAH. Neurobiol Dis 58:296–307
Hong Y, Yan W, Chen S et al (2010) The role of Nrf2 signaling in the regulation of antioxidants and detoxifying enzymes after traumatic brain injury in rats and mice. Acta Pharmacol Sin 31:1421–30
He Z, Ostrowski RP, Sun X et al (2012) Targeting C/EBP homologous protein with siRNA attenuates cerebral vasospasm after experimental subarachnoid hemorrhage. Exp Neurol 238:218–24
Kazantsev AG, Thompson LM (2008) Therapeutic application of histone deacetylase inhibitors for central nervous system disorders. Nat Rev Drug Discov 7:854–68
Ren M, Leng Y, Jeong M et al (2004) Valproic acid reduces brain damage induced by transient focal cerebral ischemia in rats: potential roles of histone deacetylase inhibition and heat shock protein induction. J Neurochem 89:1358–67
Sinn DI, Kim SJ, Chu K et al (2007) Valproic acid-mediated neuroprotection in intracerebral hemorrhage via histone deacetylase inhibition and transcriptional activation. Neurobiol Dis 26:464–72
Shein NA, Grigoriadis N, Alexandrovich AG et al (2009) Histone deacetylase inhibitor ITF2357 is neuroprotective, improves functional recovery, and induces glial apoptosis following experimental traumatic brain injury. FASEB J 23:4266–75
Kim HJ, Rowe M, Ren M et al (2007) Histone deacetylase inhibitors exhibit anti-inflammatory and neuroprotective effects in a rat permanent ischemic model of stroke: multiple mechanisms of action. J Pharmacol Exp Ther 321:892–901
Altay O, Hasegawa Y, Sherchan P et al (2012) Isoflurane delays the development of early brain injury after subarachnoid hemorrhage through sphingosine-related pathway activation in mice. Crit Care Med 40:1908–13
Reed JC (2002) Apoptosis-based therapies. Nat Rev Drug Discov 1:111–21
Li F, Vierstra RD (2012) Autophagy: a multifaceted intracellular system for bulk and selective recycling. Trends Plant Sci 17:526–37
Smith CM, Chen Y, Sullivan ML et al (2011) Autophagy in acute brain injury: feast, famine, or folly? Neurobiol Dis 43:52–9
Zhao H, Ji Z, Tang D et al (2013) Role of autophagy in early brain injury after subarachnoid hemorrhage in rats. Mol Biol Rep 40:819–27
Zhou F, Yang Y, Xing D (2011) Bcl-2 and Bcl-xL play important roles in the crosstalk between autophagy and apoptosis. FEBS J 278:403–13
Kang R, Zeh HJ, Lotze MT et al (2011) The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ 18:571–80
Maiuri MC, Zalckvar E, Kimchi A et al (2007) Self-eating and self-killing: crosstalk between autophagy and apoptosis. Nat Rev Mol Cell Biol 8:741–52
Acknowledgments
This work was supported by Grant 81371369 and 81371433 from the National Natural Science Foundation of China; Grant 20120101120030 from the Doctoral Program of the Ministry of Education; Grant 2013KYA088 from the Zhejiang Provincial Medical Science and Technology Planning Project; and Grant Y13H090007 and LY13H090002 from the Zhejiang Provincial Natural Science Foundation of China.
Conflict of Interest
The authors declare that there is no conflicts of interest regarding the publication of this paper.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Anwen Shao and Zhen Wang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Shao, A., Wang, Z., Wu, H. et al. Enhancement of Autophagy by Histone Deacetylase Inhibitor Trichostatin A Ameliorates Neuronal Apoptosis After Subarachnoid Hemorrhage in Rats. Mol Neurobiol 53, 18–27 (2016). https://doi.org/10.1007/s12035-014-8986-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-014-8986-0