Abstract
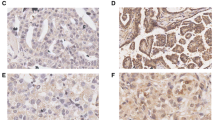
This study focused on determining the role of Spy1 in human epithelial ovarian cancer (EOC). Speedy is a novel cell cycle protein capable of promoting cell proliferation. In this study, western blot and immunohistochemistrical analyses were performed to detect the expression of Spy1 in ovarian cancer. Spy1 protein levels increased with ovarian cancer grade, and Kaplan–Meier curve showed that overexpression of Spy1 was significantly correlated with reduced patient survival. In vitro, Spy1 depletion in ovarian cell lines led to reduced proliferation according to CCK8 and plate colony assays. The expression of Spy1 was positively related to pThr187-p27. Flow cytometry revealed that the reduced expression of Spy1 induced the apoptosis of the EOC cells. In summary, our findings suggested that Spy1 may be a novel independent prognostic predictor of survival for ovarian patients.




Similar content being viewed by others
References
Al Sorkhy M, Ferraiuolo RM, Jalili E, Malysa A, Fratiloiu AR, Sloane BF, Porter LA (2012) The cyclin-like protein Spy1/RINGO promotes mammary transformation and is elevated in human breast cancer. BMC Cancer 12:45. doi:10.1186/1471-2407-12-45
Belletti B et al (2005) p27(kip1) functional regulation in human cancer: a potential target for therapeutic designs. Curr Med Chem 12:1589–1605
Berton S et al (2014) Genetic characterization of p27(kip1) and stathmin in controlling cell proliferation in vivo. Cell Cycle 13:3100–3111. doi:10.4161/15384101.2014.949512
Brown NR, Noble ME, Endicott JA, Johnson LN (1999) The structural basis for specificity of substrate and recruitment peptides for cyclin-dependent kinases. Nat Cell Biol 1:438–443. doi:10.1038/15674
Chen C et al (2015) Expression and clinical role of TCTP in epithelial ovarian cancer. J Mol Histol 46:145–156. doi:10.1007/s10735-014-9607-y
Chu IM, Hengst L, Slingerland JM (2008) The Cdk inhibitor p27 in human cancer: prognostic potential and relevance to anticancer therapy. Nat Rev Cancer 8:253–267. doi:10.1038/nrc2347
Ding Z et al (2015) Spy1 induces de-ubiquitinating of RIP1 arrest and confers glioblastoma’s resistance to tumor necrosis factor (TNF-alpha)-induced apoptosis through suppressing the association of CLIPR-59 and CYLD. Cell Cycle 14:2149–2159. doi:10.1080/15384101.2015.1041688
Fei M, Hang Q, Hou S, Ruan C (2013) Cell adhesion to fibronectin down-regulates the expression of Spy1 and contributes to drug resistance in multiple myeloma cells. Int J Hematol 98:446–455. doi:10.1007/s12185-013-1435-4
Gastwirt RF, McAndrew CW, Donoghue DJ (2007) Speedy/RINGO regulation of CDKs in cell cycle, checkpoint activation and apoptosis. Cell Cycle 6:1188–1193
Golipour A et al (2008) The Spy1/RINGO family represents a novel mechanism regulating mammary growth and tumorigenesis. Cancer Res 68:3591–3600. doi:10.1158/0008-5472.CAN-07-6453
Gulden C, Olopade OI (2010) Risk assessment and genetic testing for ovarian cancer. Am J Roentgenol 194:309–310. doi:10.2214/AJR.09.3999
Hang Q et al (2012) Expression of Spy1 protein in human non-Hodgkin’s lymphomas is correlated with phosphorylation of p27 Kip1 on Thr187 and cell proliferation. Med Oncol 29:3504–3514. doi:10.1007/s12032-012-0224-x
Hartwell LH, Kastan MB (1994) Cell cycle control and cancer. Science 266:1821–1828
Holschneider CH, Berek JS (2000) Ovarian cancer: epidemiology, biology, and prognostic factors. Semin Surg Oncol 19:3–10
Jiang L et al (2015) The role of cyclin G1 in cellular proliferation and apoptosis of human epithelial ovarian cancer. J Mol Histol 46:291–302. doi:10.1007/s10735-015-9622-7
Ke Q et al (2009) Expression and prognostic role of Spy1 as a novel cell cycle protein in hepatocellular carcinoma. Exp Mol Pathol 87:167–172. doi:10.1016/j.yexmp.2009.07.011
Lenormand JL, Dellinger RW, Knudsen KE, Subramani S, Donoghue DJ (1999) Speedy: a novel cell cycle regulator of the G2/M transition. EMBO J 18:1869–1877. doi:10.1093/emboj/18.7.1869
Levkau B et al (1998) Cleavage of p21Cip1/Waf1 and p27Kip1 mediates apoptosis in endothelial cells through activation of Cdk2: role of a caspase cascade. Mol Cell 1:553–563
Lim S, Kaldis P (2013) Cdks, cyclins and CKIs: roles beyond cell cycle regulation. Development 140:3079–3093. doi:10.1242/dev.091744
Lu M, Wang Y, Xu F, Xiang J, Chen D (2015) The prognostic of p27 in ovarian cancer: a meta-analysis. Arch Gynecol Obstet. doi:10.1007/s00404-015-3817-8
Lubanska D, Market-Velker BA, deCarvalho AC, Mikkelsen T, Fidalgo da Silva E, Porter LA (2014) The cyclin-like protein Spy1 regulates growth and division characteristics of the CD133+ population in human glioma. Cancer Cell 25:64–76. doi:10.1016/j.ccr.2013.12.006
McAndrew CW, Gastwirt RF, Meyer AN, Porter LA, Donoghue DJ (2007) Spy1 enhances phosphorylation and degradation of the cell cycle inhibitor p27. Cell Cycle 6:1937–1945
McAndrew CW, Gastwirt RF, Donoghue DJ (2009) The atypical CDK activator Spy1 regulates the intrinsic DNA damage response and is dependent upon p53 to inhibit apoptosis. Cell Cycle 8:66–75
Morgan DO (1997) Cyclin-dependent kinases: engines, clocks, and microprocessors. Annu Rev Cell Dev Biol 13:261–291. doi:10.1146/annurev.cellbio.13.1.261
Polyak K, Waldman T, He TC, Kinzler KW, Vogelstein B (1996) Genetic determinants of p53-induced apoptosis and growth arrest. Genes Dev 10:1945–1952
Porter LA, Dellinger RW, Tynan JA, Barnes EA, Kong M, Lenormand JL, Donoghue DJ (2002) Human Speedy: a novel cell cycle regulator that enhances proliferation through activation of Cdk2. J Cell Biol 157:357–366. doi:10.1083/jcb.200109045
Porter LA, Kong-Beltran M, Donoghue DJ (2003) Spy1 interacts with p27Kip1 to allow G1/S progression. Mol Biol Cell 14:3664–3674. doi:10.1091/mbc.E02-12-0820
Sherr CJ (1996) Cancer cell cycles. Science 274:1672–1677
Sherr CJ, Roberts JM (1999) CDK inhibitors: positive and negative regulators of G1-phase progression. Genes Dev 13:1501–1512
Siegel RL, Miller KD, Jemal A (2015) Cancer statistics, 2015. CA Cancer J Clin 65:5–29. doi:10.3322/caac.21254
Turan G et al (2014) The expression of HER-2/neu (c-erbB2), survivin and cycline D1 in serous ovarian neoplasms: their correlation with clinicopathological variables. J Mol Histol 45:679–687. doi:10.1007/s10735-014-9591-2
Wang Y et al (2012) Overexpression of forkhead box J2 can decrease the migration of breast cancer cells. J Cell Biochem 113:2729–2737. doi:10.1002/jcb.24146
Wang Y et al (2013) Interaction with cyclin H/cyclin-dependent kinase 7 (CCNH/CDK7) stabilizes C-terminal binding protein 2 (CtBP2) and promotes cancer cell migration. J Biol Chem 288:9028–9034. doi:10.1074/jbc.M112.432005
Yao L et al (2014) CRMP1 interacted with Spy1 during the collapse of growth cones induced by Sema3A and acted on regeneration after sciatic nerve crush. Mol Neurobiol. doi:10.1007/s12035-014-9049-2
Zhang Y, Fujita N, Tsuruo T (1999) Caspase-mediated cleavage of p21Waf1/Cip1 converts cancer cells from growth arrest to undergoing apoptosis. Oncogene 18:1131–1138. doi:10.1038/sj.onc.1202426
Zhang L, Shen A, Ke Q, Zhao W, Yan M, Cheng C (2012) Spy1 is frequently overexpressed in malignant gliomas and critically regulates the proliferation of glioma cells. J Mol Neurosci 47:485–494. doi:10.1007/s12031-012-9709-5
Zucchi I et al (2004) Gene expression profiles of epithelial cells microscopically isolated from a breast-invasive ductal carcinoma and a nodal metastasis. Proc Natl Acad Sci USA 101:18147–18152. doi:10.1073/pnas.0408260101
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 81472185).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Shumin Lu and Rong Liu have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Lu, S., Liu, R., Su, M. et al. Spy1 participates in the proliferation and apoptosis of epithelial ovarian cancer. J Mol Hist 47, 47–57 (2016). https://doi.org/10.1007/s10735-015-9646-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-015-9646-z