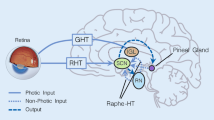
Abstract
Sleep is a globally observable fact, or period of reversible distracted rest, that can be distinguished from arousal by various behavioral criteria. Although the function of sleep is an evolutionarily conserved behavior, its mechanism is not yet clear. The zebrafish (Danio rerio) has become a valuable model for neurobehavioral studies such as studying learning, memory, anxiety, and depression. It is characterized by a sleep-like state and circadian rhythm, making it comparable to mammals. Zebrafish are a good model for behavioral studies because they share genetic similarities with humans. A number of neurotransmitters are involved in sleep and wakefulness. There is a binding between melatonin and the hypocretin system present in zebrafish. The full understanding of sleep and wakefulness physiology in zebrafish is still unclear among researchers. Therefore, to make a clear understanding of the sleep/wake cycle in zebrafish, this article covers the mechanism involved behind it, and the role of the neuromodulator system followed by the mechanism of the HPA axis.









Similar content being viewed by others
Data availability
Not applicable for this submission.
References
Agrawal S, Singh V, Singh C, Singh A (2022) A review on pathophysiological aspects of Sleep Deprivation. CNS Neurol Disord Drug Targets. https://doi.org/10.2174/1871527321666220512092718
Aime M, Adamantidis AR (n.d.) The thalamus and sleep. In: the BOOK The Thalamus, Cambridge University Press
Almeida GZ (2022) Stress-regulating pathways interact with sleep-regulating pathways under sleep deprived conditions. PhD Thesis,. Florida Atlantic University
Altenhofen S, Bonan CD (2022) Zebrafish as a tool in the study of sleep and memory-related disorders. Curr Neuropharmacol 20:540–549
Aranda-Martínez P et al (2022) The zebrafish, an outstanding model for biomedical research in the field of melatonin and human diseases. Int J Mol Sci 23:7438
Azeez IA et al (2021) An overview of the orexinergic system in different animal species. Metab Brain Dis 36:1419–1444
Bao W-W et al (2023) Understanding the neural mechanisms of general anesthesia from interaction with sleep–wake state: a decade of discovery. Pharmacol Rev 75:532–553
Barrios JP et al (2020) Hypothalamic dopamine neurons control sensorimotor behavior by modulating brainstem premotor nuclei in zebrafish. Curr Biol 30:4606–4618
Bernstein DL et al (2018) Hypocretin receptor 1 knockdown in the ventral tegmental area attenuates mesolimbic dopamine signaling and reduces motivation for cocaine. Addict Biol 23:1032–1045
Blows WT (2021) The biological basis of mental health. Routledge
Boutrel B, Koob GF (2004) What keeps us awake: the neuropharmacology of stimulants and wakefulness promoting medications. Sleep 27:1181–1194
Boykin JC (2016) The behavioral, biochemical and genetic effects of sleep deprivation in zebrafish (Danio Rerio). Electronic Theses and Dissertations, pp 1479
Bringmann H (2019) Genetic sleep deprivation: using sleep mutants to study sleep functions. EMBO Rep 20:e46807
Carli G, Farabollini F (2022) Neuromediators and defensive responses including tonic immobility (TI): brain areas and circuits involved. Defence from Invertebrates to Mammals: Focus on Tonic Immobility 271:167
Cassar S et al (2019) Use of zebrafish in drug discovery toxicology. Chem Res Toxicol 33:95–118
Ch’ng S et al (2018) The intersection of stress and reward: BNST modulation of aversive and appetitive states. Prog Neuro-Psychopharmacol Biol Psychiatry 87:108–125
Chu B et al (2021) Physiology, stress reaction. In: StatPearls [Internet]. StatPearls Publishing
Collier AD et al (2022) Embryonic ethanol exposure induces ectopic Hcrt and MCH neurons outside hypothalamus in rats and zebrafish: role in ethanol-induced behavioural disturbances. Addict Biol 27:e13238
Cully M (2019) Zebrafish earn their drug discovery stripes. Nat Rev Drug Discov 18(11):811–813. https://doi.org/10.1038/d41573-019-00165-x
Daskalakis NP, Meijer OC, de Kloet ER (2022) Mineralocorticoid receptor and glucocorticoid receptor work alone and together in cell-type-specific manner: Implications for resilience prediction and targeted therapy. Neurobiol Stress 18:100455. https://doi.org/10.1016/j.ynstr.2022.100455
Dopp J, Ortega A, Davie K, Poovathingal S, Baz ES, Liu S (2023) Single-cell transcriptomics reveals that glial cells integrate homeostatic and circadian processes to drive sleep-wake cycles. bioRxiv, pp 1-45
Duhart JM et al (2023) Many faces of sleep regulation: beyond the time of day and prior wake time. FEBS J 290:931–950
Eban-Rothschild A et al (2018) Neuronal mechanisms for sleep/wake regulation and modulatory drive. Neuropsychopharmacology 43:937–952
Echevarria DJ, Khan KM (2017) Rest in the zebrafish. Int J Comp Psychol 30:32831. https://doi.org/10.46867/ijcp.2017.30.00.15
Erickson E (2020) Investigating the role of Phox2B-expressing glutamatergic parafacial zone neurons in sleep wake control GSBS dissertations and theses. https://doi.org/10.13028/tsyw-vr44
Gaidin SG et al (2020) Activation of alpha-2 adrenergic receptors stimulates GABA release by astrocytes. Glia 68:1114–1130
Ghareghani M et al (2023) Melatonin and vitamin D, two sides of the same coin, better to land on its edge to improve multiple sclerosis. Proc Natl Acad Sci 120:e2219334120
Gjerstad JK et al (2018) Role of glucocorticoid negative feedback in the regulation of HPA axis pulsatility. Stress 21:403–416
Grafe LA, Bhatnagar S (2020) The contribution of orexins to sex differences in the stress response. Brain Res 1731:145893
Griffin J (1996) The origin of dreams: A psychobiological approach. MPhil thesis, London School of Economics and Political Science
Herman JP et al (2020) Brain mechanisms of HPA axis regulation: neurocircuitry and feedback in context Richard Kvetnansky lecture. Stress 23:617–632
Hernández-Pérez OR et al (2019) A synaptically connected hypothalamic magnocellular vasopressin-locus coeruleus neuronal circuit and its plasticity in response to emotional and physiological stress. Front Neurosci 13:196
Holst SC, Landolt HP (2018) Sleep-wake neurochemistry. Sleep Med Clin 13(2):137–146. https://doi.org/10.1016/j.jsmc.2018.03.002
Isa T, Marquez-Legorreta E, Grillner S, Scott EK (2021) The tectum/superior colliculus as the vertebrate solution for spatial sensory integration and action. Curr Biol 31(11):741–762. https://doi.org/10.1016/j.cub.2021.04.001
Jha VM, Jha SK (2020) Sleep: evolution and functions. Springer
Jiang Y et al (2022) Prednisolone induces sleep disorders via inhibition of melatonin secretion by the circadian rhythm in zebrafish. Biomed Pharmacother 147:112590
Jones BE (2020) Arousal and sleep circuits. Neuropsychopharmacology 45:6–20
Kanathur N et al (2010) Circadian rhythm sleep disorders. Clin Chest Med 31:319–325
Kaplan GB, Lakis GA, Zhoba H (2022) Sleep-wake and arousal dysfunctions in post-traumatic stress disorder: Role of orexin systems. Brain Res Bull 186:106–122. https://doi.org/10.1016/j.brainresbull.2022.05.006
Kawashima T (2018) The role of the serotonergic system in motor control. Neurosci Res 129:32–39
Kelly ML (2020) An investigation into sleep in sharks: behavioural and electrophysiological approaches. Thesis The University of Western Australia
Kelly ML et al (2020) Evidence for sleep in sharks and rays: behavioural, physiological, and evolutionary considerations. Brain Behav Evol 94:37–50
Kesner AJ et al (2022) Seeking motivation and reward: roles of dopamine, hippocampus, and supramammillo-septal pathway. Prog Neurobiol 212:102252
Kharkovets T, Hardelin JP, Safieddine S, Schweizer M, El-Amraoui A, Petit C, Jentsch TJ (2000) KCNQ4, a K+ channel mutated in a form of dominant 102 deafness, is expressed in the inner ear and the central auditory pathway. PNAS 97:4333–4338
Kim DW et al (2022) Single-cell analysis of early chick hypothalamic development reveals that hypothalamic cells are induced from prethalamic-like progenitors. Cell Rep 38:110251
Kim JS, Iremonger KJ (2019) Temporally tuned corticosteroid feedback regulation of the stress axis. Trends Endocrinol Metab 30:783–792
Kornum BR, Mignot E (2023) Neurobioloy of sleep and circadian disorders. In: Neurobiology of Brain Disorders. Elsevier, pp 635–658
Kroll F (2022) From disease genes to behavioural screen in zebrafish: early onset Alzheimer’s as case study. PhD Thesis,. UCL (University College London)
Latifi B et al (2018) Sleep-wake cycling and energy conservation: role of hypocretin and the lateral hypothalamus in dynamic state-dependent resource optimization. Front Neurol 9:790
Le Bon O (2020) Relationships between REM and NREM in the NREM-REM sleep cycle: a review on competing concepts. Sleep Med 70:6–16
Lendner JD et al (2020) An electrophysiological marker of arousal level in humans. Elife 9:e55092
Leung LC et al (2019) Neural signatures of sleep in zebrafish. Nature 571:198–204
Li S-B, de Lecea L (2020) The hypocretin (orexin) system: from a neural circuitry perspective. Neuropharmacology 167:107993
Li Y-D et al (2021) Ventral pallidal GABAergic neurons control wakefulness associated with motivation through the ventral tegmental pathway. Mol Psychiatry 26:2912–2928
Lightman SL et al (2020) Dynamics of ACTH and cortisol secretion and implications for disease. Endocr Rev 41:bnaa002
Lin J-G et al (2018) Histamine H1 receptor antagonists facilitate electroacupuncture analgesia. Am J Chin Med 46:55–68
Lu Q, Kim JY (2022) Mammalian circadian networks mediated by the suprachiasmatic nucleus. FEBS J 289:6589–6604
Luppi P-H, Fort P (2019) Sleep–wake physiology. Handb Clin Neurol 160:359–370
Luppi P-H et al (2019) Inhibitory mechanisms in the dorsal raphe nucleus and locus coeruleus during sleep. In: Handbook of behavioral state control. CRC Press, pp 195–211
Lyons DG, Rihel J (2020) Sleep circuits and physiology in non-mammalian systems. Curr Opin Physio 15:245–255
Mader EC Jr, Mader ACL (2016) Sleep as spatiotemporal integration of biological processes that evolved to periodically reinforce neurodynamic and metabolic homeostasis: The 2m3d paradigm of sleep. J Neurol Sci 367:63–80
Malikowska-Racia N, Salat K (2019) Recent advances in the neurobiology of posttraumatic stress disorder: a review of possible mechanisms underlying an effective pharmacotherapy. Pharmacol Res 142:30–49
Maroteaux L, Kilic F (2018) Frontiers of serotonin beyond the brain. Pharmacol Res 140:1–6
Martire VL et al (2020) Stress & sleep: a relationship lasting a lifetime. Neurosci Biobehav Rev 117:65–77
Mearns DS et al (2020) Deconstructing hunting behavior reveals a tightly coupled stimulus-response loop. Curr Biol 30:54–69
Mehta R et al (2020) REM sleep loss–induced elevated noradrenaline could predispose an individual to psychosomatic disorders: a review focused on proposal for prediction, prevention, and personalized treatment. EPMA J 11:529–549
Mi P et al (2019) Melatonin protects embryonic development and maintains sleep/wake behaviors from the deleterious effects of fluorene-9-bisphenol in zebrafish (Danio rerio). J Pineal Res 66:e12530
Mosser EA et al (2019) Identification of pathways that regulate circadian rhythms using a larval zebrafish small molecule screen. Sci Rep 9:1–14
Mu P, Huang YH (2019) Cholinergic system in sleep regulation of emotion and motivation. Pharmacol Res 143:113–118
Müller TE et al (2020) Role of the serotonergic system in ethanol-induced aggression and anxiety: a pharmacological approach using the zebrafish model. Eur Neuropsychopharmacol 32:66–76
Nevárez N, de Lecea L (2018) Recent advances in understanding the roles of hypocretin/orexin in arousal, affect, and motivation. F1000Res. 7:1000–1421. https://doi.org/10.12688/f1000research.15097.1
Oesch LT, Adamantidis AR (2021) How REM sleep shapes hypothalamic computations for feeding behavior. Trends Neurosci 44:990–1003
Oikonomou G et al (2019) The serotonergic raphe promote sleep in zebrafish and mice. Neuron 103:686–701
Osorio-Forero A (2022) An active locus coeruleus in sleep: towards a dynamic structure of NREM sleep. PhD Thesis, UNIL, pp 1–95
Paller KA et al (2021) Memory and sleep: how sleep cognition can change the waking mind for the better. Annu Rev Psychol 72:123–150
Panula P et al (2021) The histamine system in zebrafish brain: organization, receptors, and behavioral roles. In: The functional roles of histamine receptors. Springer, pp 291–302
Patke A et al (2020) Molecular mechanisms and physiological importance of circadian rhythms. Nat Rev Mol Cell Biol 21:67–84
Pfaff D (2019) How brain arousal mechanisms work: paths toward consciousness. Cambridge University Press
Pinheiro-da-Silva J et al (2018) Sleep deprivation impairs cognitive performance in zebrafish: a matter of fact? Behav Processes 157:656–663
Ramachandran S, Rajagopal S (2019) Zebrafish: a model for marine peptide based drug screening. Springer
Rao R et al (2021) Modeling the influence of chronic sleep restriction on cortisol circadian rhythms, with implications for metabolic disorders. Metabolites 11:483
Rasch B, Born J (2013) About sleep's role in memory. Physiol Rev 93(2):681–766. https://doi.org/10.1152/physrev.00032.2012
Sanford L (2019) Characterization of intracellular Zn2+ dynamics and Zn2+-dependent gene expression in primary dissociated hippocampal neurons. Desserted Thesis, University of Colorado Boulder
Sharples L (2020) Disrupted-in-schizophrenia 1 (disc1) regulates development of the hypothalamus and its associated behaviours. PhD Thesis,. University of Sheffield
Shine JM (2019) Neuromodulatory influences on integration and segregation in the brain. Trends Cogn Sci 23:572–583
Simmons SJ (2018) Hypocretin/orexin and the ventral midbrain: topography and function associated with psychostimulant-taking and affect. Temple University
Simor P et al (2020) The microstructure of REM sleep: why phasic and tonic? Sleep Med Rev 52:101305
Singh R, Biswas DA, Biswas D (2023) Physiological role of orexin/hypocretin in the human body in motivated behavior: a comprehensive review. Cureus 15(1):e34009. https://doi.org/10.7759/cureus.34009
Smith SM, Vale WW (2022) The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin Neurosci 8(4):383–395. https://doi.org/10.31887/DCNS.2006.8.4/ssmith
Stetler C et al (2004) Uncoupling of social zeitgebers and diurnal cortisol secretion in clinical depression. Psychoneuroendocrinology 29:1250–1259
Takahashi N et al (2020) Polygenic risk score analysis revealed shared genetic background in attention deficit hyperactivity disorder and narcolepsy. Transl Psychiatry 10:284
Téblick A et al (2022) Impact of hydrocortisone and of CRH infusion on the hypothalamus-pituitary-adrenocortical axis of septic male mice. Endocrinology 163:bqab222
Tiligada E, Ennis M (2020) Histamine pharmacology: from Sir Henry Dale to the 21st century. Br J Pharmacol 177:469–489
Top D, Young MW (2018) Coordination between differentially regulated circadian clocks generates rhythmic behavior. Cold Spring Harb Perspect Biol 10:a033589
Trang A, Khandhar PB (2021) Physiology, acetylcholinesterase. In: StatPearls [Internet]. StatPearls Publishing
Van Drunen R, Eckel-Mahan K (2021) Circadian rhythms of the hypothalamus: from function to physiology. Clocks sleep 3:189–226
Van Egroo M, Koshmanova E, Vandewalle G, Jacobs HI (2022) Importance of the locus coeruleusnorepinephrine system in sleep-wake regulation: Implications for aging and Alzheimer's disease. Sleep Med Rev 62:101592. https://doi.org/10.1016/j.smrv.2022.101592
Vargas I, Lopez-Duran N (2020) The cortisol awakening response after sleep deprivation: Is the cortisol awakening response a “response” to awakening or a circadian process? J Health Psychol 25:900–912
Venner A et al (2019) Reassessing the role of histaminergic tuberomammillary neurons in arousal control. J Neurosci 39:8929–8939
Vitkauskas M, Mathuru AS (2020) Total recall: lateral habenula and psychedelics in the study of depression and comorbid brain disorders. Int J Mol Sci 21:6525
War R et al (2022) Zebrafish as an emerging alternative tool for studying anxiety disorders. J Adv Sci Res 13:13–20
Wiggin TD (2015) The spinal locomotor circuit of the larval zebrafish: anatomical organization and functional components. PhD Thesis,. University of Minnesota
Yoshikawa T et al (2021) Histaminergic neurons in the tuberomammillary nucleus as a control centre for wakefulness. Br J Pharmacol 178:750–769
Yusan RT, Fadlilah SH (2023) Review sleep and sleep deprivation. Med Health J 2:148–169
Yutao BAI (2022) Towards a higher-throughput chemobehavioural phenomics using small model organisms. PhD Thesis,. RMIT University
Zada D, Appelbaum L (2020) Behavioral criteria and techniques to define sleep in zebrafish. In: Behavioral and Neural Genetics of Zebrafish. Elsevier, pp 141–153
Zada D et al (2019) Sleep increases chromosome dynamics to enable reduction of accumulating DNA damage in single neurons. Nat Commun 10:895
Zerbi V et al (2019) Rapid reconfiguration of the functional connectome after chemogenetic locus coeruleus activation. Neuron 103:702–718
Zhang X et al (2020) Inactivation of the ventral pallidum by GABA A receptor agonist promotes non-rapid eye movement sleep in rats. Neurochem Res 45:1791–1801
Zhdanova IV (2006) Sleep in zebrafish. Zebrafish 3:215–226
Zhdanova IV (2011) Sleep in zebrafish. Zebrafish 3(2):215–226. https://doi.org/10.1089/zeb.2006.3.215
Zheng X et al (2021) Environmental chemicals affect circadian rhythms: an underexplored effect influencing health and fitness in animals and humans. Environ Int 149:106159
Zimmerman JE et al (2008) Conservation of sleep: insights from non-mammalian model systems. Trends Neurosci 31:371–376
Zisapel N (2018) New perspectives on the role of melatonin in human sleep, circadian rhythms and their regulation. Br J Pharmacol 175:3190–3199
Acknowledgements
All authors acknowledge the Institute for support.
Funding
As provided by the Institute.
Author information
Authors and Affiliations
Contributions
Rima Singh and Deepali Sharma wrote the main manuscript text and prepared figures Anoop Kumar and Charan Singh revise the manuscript. Arti Singh design the layout and critically revise the manuscriptAll authors reviewed the manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singh, R., Sharma, D., Kumar, A. et al. Understanding zebrafish sleep and wakefulness physiology as an experimental model for biomedical research. Fish Physiol Biochem 50, 827–842 (2024). https://doi.org/10.1007/s10695-023-01288-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-023-01288-0