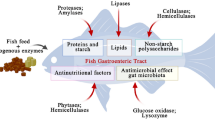
Abstract
We studied the effects of Yarrowia lipolytica biomass on digestive enzymes, blood biochemical profile, energy metabolism enzymes, and proximate meat composition of Nile tilapias. The experiment was entirely randomized with four replications. The animals (n = 20 per repetition) were fed with 0%, 3%, 5%, and 7% of biomass for 40 days and then blood and liver were analyzed. There was an increase in the activities of chymotrypsin (5, 7% groups), trypsin (3, 5% groups), and sucrase (7% group) compared to the respective control groups. On the other hand, maltase activity was significantly reduced for all yeast biomass treatments, while the supplementation did not influence lipase and amylase activities. Moreover, the blood triacylglycerol concentrations were increased in the 7% group, while any treatment modified blood total cholesterol, glycemia, and hepatic glycogen content. Y. lipolytica biomass promoted significant increases in meat protein and lipid contents without changes in moisture and ash parameters. Furthermore, Y. lipolytica biomass promoted increases in hexokinase (3% group), phosphofructokinase (5, 7% groups), glucose-6-phosphate dehydrogenase (5% group), citrate synthase (3% group), aspartate aminotransferase and alanine aminotransferase (3% group), and glutamate dehydrogenase (3, 5% groups) compared to the respective control groups. At the same time, no changes were observed in the activity of glucose-6-phosphatase. Y. lipolytica biomass supplementation in tilapias’ diet can modulate the digestive system and improve nutrient disponibility to the cells. Moreover, the changes in the metabolic profile and in energy metabolism can be correlated with the improvement of meat composition. Therefore, the Y. lipolytica biomass has a great potential to be used as a feed ingredient for Nile tilapias.









Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Abass DA, Obirikorang KA, Bb C, Edziyie RE, Skov PV (2018) Dietary supplementation of yeast (Saccharomyces cerevisiae) improves growth, stress tolerance, and disease resistance in juvenile Nile tilapia (Oreochromis niloticus). Aquacult Internat 26:843–855. https://doi.org/10.1007/s10499-018-0255-1
Abdel-Tawwab M, Am A-R, Nem I (2008) Evaluation of commercial live bakers’ yeast, Saccharomyces cerevisiae as a growth and immunity promoter for Fry Nile tilapia, Oreochromis niloticus (L.) challenged in situ with Aeromonas hydrophila. Aquaculture 280:185–189. https://doi.org/10.1016/j.aquaculture.2008.03.055
Adel M, Safari R, Yeganeh S, Binajj M, Ghiasi M, Ahmadyand S (2017) Effect of dietary GroBiotic®- A supplementation as a prebiotic on the intestinal microflora, growth performance, haemato-serological parameters, survival rate, and body composition in juvenile beluga (Huso huso Linnaeus, 1754). Aquacult Nut 23:492–499. https://doi.org/10.1111/anu.12417
Agboola JO, Øverland M, Skrede A, Hansen JØ (2021) Yeast as major protein-rich ingredient in aquafeeds: a review of the implications for aquaculture production. Rev Aquacult 13:949–970. https://doi.org/10.1111/raq.12507
Alam MS, Liang XF, Liu L, He S, Kuang Y, Hoseinifar SH, Dawar FU (2019) Growth and metabolic response of Chinese perch to different dietary protein-to-energy ratios in artificial diets. Int J Mol Sci 20:5983. https://doi.org/10.3390/ijms20235983
Ama G, Hahh M, Maeh W, Th E-A (2012) Effect of using baker’s yeast and exogenous digestive enzymes as growth promoters on growth, feed utilization and hematological indices of Nile tilapia, Oreochromis niloticus Fingerlings. J Agricult Sci Technol B 2:15–28
Andlid T, Vazquez JR, Gustafsson L (1998) Yeast isolated from the intestine of rainbow trout adhere to and grow in intestinal mucus. Mol Mar Biol Biotech 7:115–126
AOAC - Association of Official Analytical Chemists (1995) Official methods of analysis of the Association of the Analytical Chemists, 16th edn Washington
Ar Á-S, Nolasco-Soria H, Peña-Rodríguez A, Mejía-Ruíz H (2018) In vitro digestibility of Yarrowia lipolytica yeast and growth performance in whiteleg shrimp Litopenaeus vannamei. Turk J Fish Aquat Sci 18:395–404. https://doi.org/10.4194/1303-2712-v18_3_05
Ayiku S, Shen JF, Tan BP, Dong XH, Liu HY (2020) Effects of dietary yeast culture on shrimp growth, immune response, intestinal health, and disease resistance against Vibrio harveyi. Fish Shellf Immunol 102:286–295. https://doi.org/10.1016/j.fsi.2020.04.036
Baldwin J, Elias JP, RMG W, Donovan DA (2007) Energy metabolism in the tropical abalone, Haliotis asinina Linné: comparisons with temperate abalone species. J Exp Mar Biol Ecol 342:213–225. https://doi.org/10.1016/j.jembe.2006.09.005
Barth G, Gaillardin C (1996) Yarrowia lipolytica. In: Wolf K (ed) Nonconventional Yeasts in Biotechnology: a handbook. Springer, Berlin, Heidelberg, pp 318–388. https://doi.org/10.1007/978-3-642-79856-6_10
BCW H (1959) A modified spectrophotometric determination of chymotrypsin, trypsin, and thrombin. Canad J Biochem Physiol 37:1393–1399
Beopoulos A, Cescut J, Haddouche R, Uribelarrea JL, Molina-Jouve C, Nicaud JM (2009) Yarrowia lipolytica as a model for bio-oil production. Prog Lip Res 48:375–387. https://doi.org/10.1016/j.plipres.2009.08.005
Beopoulos A, Mrozova Z, Thevenieau F, Mt LD, Hapala I, Papanikolaou S, Chardot T, Nicaud JM (2008) Control of lipid accumulation in the yeast Yarrowia lipolytica. J App Environm Microbiol 74:7779–7789. https://doi.org/10.1128/AEM.01412-08
Bitencourt TB, Cazarolli LH, Silva VG, Souza FA, Konopka DN (2021) Avaliação da biomassa produzida a partir de fermentação por Yarrowia lipolytica de resíduo agroindustrial de mandioca (Manihot esculenta) em distintas concentrações de glicose. J Biotech Biodiv 9:142–148. https://doi.org/10.20873/jbb.uft.cemaf.v9n2.bitencourt
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Canad J Biochem Physiol 27:911–917. https://doi.org/10.1139/o59-099
Boonanuntanasarn S, Ditthab K, Jangprai A, Nakharuthai C (2019) Effects of microencapsulated Saccharomyces cerevisiae on growth, hematological indices, blood chemical, and immune parameters and intestinal morphology in striped catfish, Pangasianodon hypophthalmus. Prob Antimicrob Prot 11:427–437. https://doi.org/10.1007/s12602-018-9404-0
Botman D, Tigchelaar W, CJF VN (2014) Determination of phosphate-activated glutaminase activity and its kinetics in mouse tissues using metabolic mapping (quantitative enzyme histochemistry). J Histochem Cytochem 62:813–826. https://doi.org/10.1369/0022155414551177
Bradford M (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry 72:248–254. https://doi.org/10.1006/abio.1976.9999
Carrillo-Farnés O, Forrellat-Barrios A, Guerrero-Galván S, Veja-Villasante F (2007) A review of digestive enzyme activity in penaeid shrimps. Crustaceana 80:257–275
Chen M, Chen X-Q, Tian LX, Liu YJ, Niu J (2020) Enhanced intestinal health, immune responses, and ammonia resistance in Pacific white shrimp (Litopenaeus vannamei) fed dietary hydrolyzed yeast (Rhodotorula mucilaginosa) and Bacillus licheniformis. Aquacult Rep 17:100385. https://doi.org/10.1016/j.aqrep.2020.100385
Ciardello M, Camardella L, Di Prisco G (1995) Glucose-6-phosphate dehydrogenase from the blood cells of two Antarctic teleosts: correlation with cold adaptation. Bioch Biophys Acta 1250:76–82. https://doi.org/10.1016/0167-4838(95)00046-w
Coroian C, Miresan V, Raducu C, Costea C, Constantinescu R, Coroian A, Bentea M, Simeanu D, Radu-Rusu C, Latiu C, Cocan D (2019) Oxidative stress and haematological response in Rainbow Trout (Oncorhynchus mykiss) fed with brewer yeast (Saccharomyces Cerevisiae) as nutritional supplement. Rev Chim 70:3727–3733. https://doi.org/10.37358/RC.19.10.7634
Czech A, Merska M, Ognik K (2014) Blood immunological and biochemical indicators in turkey hens fed diets with a different content of the yeast Yarrowia Lipolytica. Ann Anim Sci 14:935–946. https://doi.org/10.2478/aoas-2014-0057
Czech A, Smolczyk A, Ognik K, Wlazło L, Nowakowicz-Dębek B, Kiesz M (2018) Effect of dietary supplementation with Yarrowia lipolytica or Saccharomyces cerevisiae yeast and probiotic additives on haematological parameters and the gut microbiota in piglets. Res Vet Sci 119:221–227. https://doi.org/10.1016/j.rvsc.2018.06.007
Dahlqvist A (1984) Assay of intestinal disaccharidases. Scand J Clin Lab Invest 44:169–172. https://doi.org/10.3109/00365518409161400
Debnath D, Pal AK, Sahu NP, Yengkokpam S, Baruah K, Choudhury D, Venkateshwarlu G (2007) Digestive enzymes and metabolic profile of Labeo rohita fingerlings fed diets with different crude protein levels. Comp Biochem Physiol part B 146:107–114. https://doi.org/10.1016/j.cbpb.2006.09.008
Dimitroglou A, Davies S, Sweetman J (2008) The effects of dietary mannan oligosaccharides on the intestinal histology of rainbow trout (Oncorhynchus mykiss). Comp Biochem Physiol part A 150:63. https://doi.org/10.1016/j.cbpa.2008.04.069
Dimitroglou A, Merrifield DL, Spring P, Sweetman J, Moate R, Davies SJ (2010) Effects of mannan oligosaccharide (MOS) supplementation on growth performance, feed utilization, intestinal histology, and gut microbiota of gilthead sea bream (Sparus aurata). Aquaculture 300:182–188. https://doi.org/10.1016/j.aquaculture.2010.01.015
Doherty D (1970) L-glutamate dehydrogenases (yeast). Met Enzymol 17:850–856. https://doi.org/10.1016/0076-6879(71)17294-1
Dong LF, Tong T, Zhang Q, Wang Q, Xu MZ, Yu HR, Wang J (2018) Effects of dietary carbohydrate to lipid ratio on growth, feed utilization, body composition, and digestive enzyme activities of golden pompano (Trachinotus ovatus). Aquacult Nut 24:341–347. https://doi.org/10.1111/anu.12565
Encarnação P (2016) Functional feed additives in aquaculture feeds. In: Formulation A (ed) Sergio F. Academic Press, Nates, pp 217–237. https://doi.org/10.1016/B978-0-12-800873-7.00005-1
Enes P, Panserat S, Kaushik S, Oliva-Teles A (2009) Nutritional regulation of hepatic glucose metabolism in fish. Fish Physiol Biochem 35:519–539. https://doi.org/10.1007/s10695-008-9259-5
Falco F, Stincone P, Cammarata M, Brandelli A (2020) Amino acids as the main energy source in fish tissues. Aquacult Fish Stud 3:1–11
FAO. Food and Agriculture Organization of the United Nations. (2022) The state of world fisheries and aquaculture (SOFIA) 2022. p. 244, Rome, Italy.
Fathi AR, Krautheim A, Lucke S, Becker K, RHJ S (2002) Nonradioactive technique to measure protein phosphatase 2A-like activity and its inhibition by drugs in cell extracts. Anal Biochem 310:208–214. https://doi.org/10.1016/s0003-2697(02)00377-9
Fernández Gimenez AV, García-Carreño FL, Navarrete Del Toro MA, Fenucci JL (2002) Digestive proteinases of Artemesia longinaris (Decapoda, Penaeidae) and relationship with molting. Comp Biochem Physiol Part B 132:593–598. https://doi.org/10.1016/s1096-4959(02)00080-5
Gálvez-López D, Chávez-Meléndez B, Vásquez-Ovando A, Rosas-Quijano R (2019) The metabolism and genetic regulation of lipids in the oleaginous yeast Yarrowia lipolytica. Braz J Microbiol 50:23–31. https://doi.org/10.1007/s42770-018-0004-7
García-Carreño FL, Hernández-Cortés MP, Haard N (1994) Enzymes with peptidase and proteinase activity from the digestive system of a fresh water and a marine decapod. J Agricult Food Chem 42:145–146. https://doi.org/10.1021/jf00043a013
Gioda CR, Pretto A, Freitas CS, Leitemperger J, Baldisserotto B, Salbego J (2017) Different feeding habits influence the activity of digestive enzymes in freshwater fish. Ciência Rural 47:e20160113. https://doi.org/10.1590/0103-8478cr20160113
Gonçalves LU, Carvalho M, EMM V (2010) Utilização de levedura íntegra e seus derivados em dietas para juvenis de tilápia do Nilo. Ciência Rural 40:1173–1179. https://doi.org/10.1590/S0103-84782010000500027
Hemre G-I, Mommsen TP, Krogdahl Å (2002) Carbohydrates in fish nutrition: effects on growth, glucose metabolism, and hepatic enzymes. Aquacult Nut 8:175–194. https://doi.org/10.1046/j.1365-2095.2002.00200.x
JFB M, Lundstedt LM, Metón I, Baanante IV, Moraes G (2006) Effects of dietary levels of protein on nitrogenous metabolism of Rhamdia quelen (Teleostei: Pimelodidae). Comp Biochem Physiol Part A 145:181–187. https://doi.org/10.1016/j.cbpa.2006.06.007
Krisman CR (1962) A method for the colorimetric estimation of glycogen with iodine. Anal Biochem 4:17–23. https://doi.org/10.1016/0003-2697(62)90014-3
Kubitza F (2011) O status atual e as tendências da tilapicultura no Brasil. Rev Pan da Aquicult 21:10–19
Lara-Flores M, Olvea-Novoa MA, Guzman-Mendez BE (2003) Use of bacteria Streptococcus faecium and Lactobacillus acidophilus, and the yeast Saccharomyces cerevisiae as growth promoters in Nile tilapia (Oreochromis niloticus). Aquaculture 216:193–201. https://doi.org/10.1016/S0044-8486(02)00277-6
Mazdak C (2018) Engineering Yarrowia lipolytica for use in Biotechnological applications: a review of major achievements and recent innovations. Mol Biotech 60:621–635. https://doi.org/10.1007/s12033-018-0093-4
Merska M, Czech A, Ognik K (2013) The effect of different doses of dried yeast Yarrowia lipolytica on production effects of turkey hens and hematological indicators of blood. Annales Universitatis Mariae Curie-Sklodowska Sectio EE Zootechnica 31:35–41
Montoya-Mejía M, García-Ulloa M, Hernández-Llamas A, Nolasco-Soria H, Rodríguez-González H (2017) Digestibility, growth, blood chemistry, and enzyme activity of juvenile Oreochromis niloticus fed isocaloric diets containing animal and plant byproducts. Rev Bras Zoot 46:873–882. https://doi.org/10.1590/S1806-92902017001200001
Moon TW (2001) Glucose intolerance in teleost fish: fact or fiction? Comp Biochem Physiol Part B 129:243–249. https://doi.org/10.1016/s1096-4959(01)00316-5
Neuls L, Souza VJ, Romão S, Bitencourt TB, CJR R, JEG P, Cazarolli LH (2021) Immunomodulatory effects of Yarrowia lipolytica as a food additive in the diet of Nile tilápia. Fish Shellf Immunol 119:272–279. https://doi.org/10.1016/j.fsi.2021.10.011
Opiyo MA, Jumbe J, Ngugi CC, Charo-Karisa H (2019) Different levels of probiotics affect growth, survival, and body composition of Nile tilapia (Oreochromis niloticus) cultured in low input ponds. Sci Afr 4:e00103. https://doi.org/10.1016/j.sciaf.2019.e00103
Özlüer-Hunt A, Özkan-Yilmaz F, Engin K, Berköz M, Sg G, Yalin S, Şahin N (2014) The effects of fish meal replacement by yeast-based nucleotides on growth, body composition, and digestive enzyme activity in rainbow trout juveniles (Oncorhynchus mykiss). Isr J Aquacult 66:964–973. https://doi.org/10.46989/001c.20745
Patsios SI, Dedousi A, Sossidou EN, Zdragas A (2020) Sustainable animal feed protein through the cultivation of Yarrowia lipolytica on agro-industrial wastes and by-products. Sustainability 12:1–23. https://doi.org/10.3390/su12041398
Pereira DF, Cazarolli LH, Lavado C, Mengatto V, MSRB F, Guedes A, Pizzolatti MG, FRMB S (2011) Effects of flavonoids on α-glucosidase activity: Potential targets for glucose homeostasis. Nutrition 27:1161–1167. https://doi.org/10.1016/j.nut.2011.01.008
Peres H, JMA F, PLP C, Furuya WM, MMP S, Oliva-Teles A, Pezzato LE, Barros MM (2022) Growth performance and metabolic responses of Nile tilapia fed diets with different protein to energy ratios. Aquaculture 547:737493. https://doi.org/10.1016/j.aquaculture.2021.737493
Polakof S, Panserat S, Soengas JL, Moon TW (2012) Glucose metabolism in fish: a review. J Comp Physiol part B 182:1015–1045. https://doi.org/10.1007/s00360-012-0658-7
Prasad L, Nayak BB, Srivastava PP, Reddy AK, MPS K (2013) Use of Brewer’s yeast Saccharomyces cerevisiae as growth promoter in giant freshwater prawn (Macrobrachium rosenbergii de Man) post larvae. Turk J Fish Aquat Sci 13:447–452. https://doi.org/10.4194/1303-2712-v13_3_07
Ran C, Huang L, Hu J, Tacon P, He S, Li Z, Wang Y, Liu Z, Xu L, Yang Y, Zhou Z (2016) Effects of dietary live and heat-inactive baker’s yeast on growth, gut health, and disease resistance of Nile tilapia under high rearing density. Fish Shellf Immunol 56:263–271. https://doi.org/10.1016/j.fsi.2016.07.001
Ruiz-Herrera J, García-Maceira P, Lc C-B, Valentín E, Sentandreu R (2003) Cell wall composition and structure of Yarrowia lipolytica transposon mutants affected in calcofluor sensitivity. Antonie van Leeuwenhoek 84:229–238. https://doi.org/10.1023/A:1026007923908
Saborowski R, Buchholz F (2002) Metabolic properties of Northern krill, Meganyctiphanes norvegica, from different climatic zones. II Enzyme characteristics and activities. Mar Biol 140:557–565. https://doi.org/10.1007/s00227-001-0734-0
Sáenz De Rodrigáñez MA, Díaz-Rosales P, Chabrillón M, Smidt H, Arijo S, León-Rubio JM, Alarcón FJ, Balebona MC, Ma M, Cara JB, Moyano FJ (2008) Effects of dietary administration of probiotics on growth and intestine function of juvenile Senegalese sole (Solea senegalensis, Kaup 1858). Aquact Nut 15:177–185. https://doi.org/10.1111/j.1365-2095.2008.00581.x
Sánchez-Muros MJ, Gárcia-Rejón L, Gárcia-Salguero L, De La Higuera M, Lupiáñez JA (1998) Long-term nutritional effects on the primary liver and kidney metabolism in rainbow trout. Adaptive response to starvation and a high-protein, carbohydrate-free diet on glutamate dehydrogenase and alanine aminotransferase kinetics. Int J Biochem Cell Biol 30:55–63. https://doi.org/10.1016/s1357-2725(97)00100-3
Seenivasan C, Bhavan PS, Radhakrishnan S (2011) Effect of probiotics (BinifitTM) on survival, growth, biochemical constituents, and energy budget of the freshwater prawn Macrobrachium rosenbergii postlarvae. Elixir Aquacult 41:5919–5927
Seixas-Filho JT (2003) Revisão sobre as enzimas digestivas nos peixes teleostei e seus métodos de determinação. Augustus v:08
Seixas-Filho JT (2004) Uma revisão sobre o papel do carboidrato e da proteína no metabolismo de peixes com hábitos alimentar carnívoro e onívoro. Augustus. 09:18
Seixas-Filho JT, Brás JM, ATM G, MGA O, Donzele JL, Menin E (2000) Anatomia funcional e morfometria dos intestinos e dos cecos pilóricos do Teleostei (Pisces) de água doce Brycon orbignyanus (Valenciennes, 1849). Rev Bras Zoot 29:313–324. https://doi.org/10.1590/S1516-35982000000200001
Smutná M, Vorlová L, Svobodová Z (2002) Pathobiochemistry of Ammonia in the Internal Environment of Fish (Review). Acta Vet BRNO 71:169–181. https://doi.org/10.2754/avb200271020169
Souza FA, Silva VG, Tormen SH, Tamura C, Jesus BV, Koester DL, Bitencourt TB (2019) Use of agro-industrial waste for the production of microbial lipids by Yarrowia lipolytica qu69. Braz J Develop 5:8801–8810. https://doi.org/10.34117/bjdv5n7-087
Stein HH, Kill DY (2006) Reduced use of antibiotic growth promoters in diets fed to weanling pigs: dietary tools, part 2. Anim Biotech 17:217–231. https://doi.org/10.1080/10495390600957191
Torrecillas S, Montero D, Izquierdo M (2014) Improved health and growth of fish fed mannan oligosaccharides: potential mode of action. Fish Shellf Immunol 36:525–544. https://doi.org/10.1016/j.fsi.2013.12.029
Ugolev AM, Kuz’mina VV (1994) Fish enterocyte hydrolases. Nutrition adaptations. Comp Biochem Physiol Part A 107:187–193. https://doi.org/10.1016/0300-9629(94)90292-5
Wm S, Ls C, López-Olmeda JF, NCS C, Fac S, Oliveira CG, Guilherme HO, Bahiense RN, Luz RK, PAP R (2020) Dietary protein modulates digestive enzyme activities and gene expression in red tilapia juveniles. Animal 14:1802–1810. https://doi.org/10.1017/S1751731120000543
Ye J-D, Wang K, Li F-D, Sun Y-Z (2011) Single or combined effects of fructo- and mannan oligosaccharide supplements and Bacillus clausii on the growth, feed utilization, body composition, digestive enzyme activity, innate immune response, and lipid metabolism of the Japanese flounder Paralichthys. Aquacul Nut 17:e902–e911. https://doi.org/10.1111/j.1365-2095.2011.00863.x
Zhao L, Wang W, Huang X, Guo T, Wen W, Feng L, Wei L (2017) The effect of replacement of fish meal by yeast extract on the digestibility, growth, and muscle composition of the shrimp Litopenaeus vannamei. Aquacult Res 48:311–320. https://doi.org/10.1111/are.12883
Zimmermann S, Fitzsimmons K (2004) Tilapicultura intensiva. In: CYRINO JE, URBINATI EC, FRACALOSSI DM, CASTAGNOLLI N (Ed) Tópicos especiais em piscicultura de água doce tropical intensiva. 1 ed. TecArt, São Paulo, pp. 239–266.
Acknowledgements
The authors are grateful to Professor Patricia Valente from the Federal University of Rio Grande do Sul (UFRGS, Brazil) for donating the Yarrowia lipolytica QU69 strain and Professor Thiago Bergler Bitencourt from the Federal University of Fronteira Sul by providing the Yarrowia lipolytica–fermented biomass.
Funding
This work was supported by Federal University of Fronteira Sul and Fundação Araucária [grant no. 739/GR/UFFS/2019 - Concession of social inclusion scholarships (PIBIS) - Fundação Araucária].
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, conceptualization, data collection, and analysis were performed by Milena Cia Retcheski, Luiz Vitor Maximowski, Keveen Jhonathan Soares Escorsin, Jéssica Kimie de Almeida Rosa Kurosaki, Silvia Romão, Thiago Bergler Bitencourt, Jorge Erick Garcia Parra, Luisa and Helena Cazarolli. The first draft of the manuscript was written by Luisa Helena Cazarolli and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This declaration is not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Retcheski, M.C., Maximowski, L.V., Escorsin, K.J.S. et al. Yarrowia lipolytica biomass—a potential additive to boost metabolic and physiological responses of Nile tilapia. Fish Physiol Biochem 49, 655–670 (2023). https://doi.org/10.1007/s10695-023-01219-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-023-01219-z