Abstract
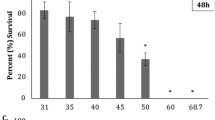
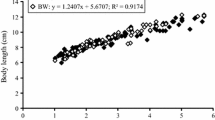
Reducing water salinity towards iso-osmotic conditions is a common practice applied in euryhaline fish farming to limit osmoregulation costs and enhance growth. In this respect, the present study investigated the timing of salinity reduction in an abrupt manner during European eel (Anguilla anguilla) larval culture by examining associated impacts on morphological and molecular levels. Larvae from 3 different parental combinations (families) were reared at constant 36 psu for 6 days (control) or subjected to a direct reduction to 18 psu on 1, 2, or 3 days post-hatch. Overall, salinity reduction enhanced growth and survival, resulting from more efficient energy resource utilization. In the control group, expression of growth-related igf2 remained constant, demonstrating a steady growth progression, while igf1 expression increased over time only for the salinity reduced treatments, potentially qualifying as a useful biomarker for growth performance. Even though each parental combination seems to have a different capacity to cope with salinity alterations, as observed by family-driven water-transport-related aquaporin (aqp1, aqp3) gene expression, it could be inferred that the abrupt salinity change is generally not stressful, based on non-upregulated heat shock proteins (hsp70, hsp90). However, the applied salinity reduction (irrespective of timing) induced the development of pericardial edema. As such, we conclude that despite the positive effect of salinity reduction on early growth and survival, the long-term benefit for eel larval culture lies in establishing a protocol for salinity reduction, at a precise developmental time point, without causing pericardial malformations.





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Benini E, Politis SN, Sørensen SR et al (2018) Effect of parental origin on early life history traits of European eel. Reprod Dom Anim 53:1149–1158. https://doi.org/10.1111/rda.13219
Bœuf G, Payan P (2001) How should salinity influence fish growth? Comp Biochem Physiol Part C 130:411–423
Bone Q, Moore RH (2008) Osmoregulation and ion balance. In: Owen E (ed) Biology of fishes: Third Edition. Taylor & Francis Group In, pp 161–207
Borgnia M, Nielsen S, Engel A, Agre P (1999) Cellular and molecular biology of the aquaporin water channels. Annu Rev Biochem 68:425–458
Butts IAE, Sørensen SR, Politis SN et al (2014) Standardization of fertilization protocols for the European eel, Anguilla anguilla. Aquaculture 426–427:9–13. https://doi.org/10.1016/j.aquaculture.2014.01.020
Cutler CP, Cramb G (2001) Molecular physiology of osmoregulation in eels and other teleosts: the role of transporter isoforms and gene duplication. Comp Biochem Physiol Part A 130:551–564
Deane EE, Kelly SP, Luk JCY, Woo NYS (2002) Chronic salinity adaptation modulates hepatic heat shock protein and insulin-like growth factor I expression in Black Sea Bream. Mar Biotechnol 4:193–205. https://doi.org/10.1007/s1012602-0091-5
Gaumet F, Boeuf G, Sever A et al (1995) Effects of salinity on the ionic balance and growth of juvenile turbot. J Fish Biol 47(5):865–876
Hellemans J, Mortier G, Paepe A De et al (2007) qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data. Genome Biol 8.https://doi.org/10.1186/gb-2007-8-2-r19
ICES (2020) European eel (Anguilla anguilla) throughout its natural range. In: Report of the ICES Advisory Committee, 2020. ICES Advice 2020
Imsland AK, Foss A, Gunnarsson S et al (2001) The interaction of temperature and salinity on growth and food conversion in juvenile turbot (Scophthalmus maximus). Aqua 198:353–367
Kinyage JPH, Pedersen PB, Pederse L-F (2019) Effects of abrupt salinity increase on nitrification processes in a freshwater moving bed biofilter. Aquac Eng 84:91–98. https://doi.org/10.1016/j.aquaeng.2018.12.005
Kottmann JS, Jørgensen MGP, Bertolini F et al (2020) Differential impacts of carp and salmon pituitary extracts on induced oogenesis, egg quality, molecular ontogeny and embryonic developmental competence in European eel. PLoS ONE 15:e0235617. https://doi.org/10.1371/journal.pone.0235617
Kurokawa T, Okamoto T, Gen K et al (2008) Influence of water temperature on morphological deformities in cultured larvae of Japanese eel, Anguilla japonica, at completion of yolk resorption. J World Aquac Soc 39:726–735
Kurokawa T, Shibahara H, Gen K et al (2013) Determination of periods of sensitivity to low-salinity and low-temperature conditions during the early development of cultured Japanese eel Anguilla japonica larvae with respect to the rate of morphological deformity at completion of yolk resorption. Fish Sci 79:673–680. https://doi.org/10.1007/s12562-013-0636-3
Kuroki M, Seo MY, Okamura A et al (2016) Morphofunctional features of ionocytes in Japanese eel Anguilla japonica leptocephali acclimated to half-diluted and full-strength seawater. Ichthyol Res 63:487–495. https://doi.org/10.1007/s10228-016-0520-0
Lee KM, Yamada Y, Okamura A et al (2013) Hyposmoregulatory ability and ion- and water-regulatory mechanisms during the leptocephalus stages of Japanese eel Anguilla japonica. Fish Sci 79:77–86. https://doi.org/10.1007/s12562-012-0576-3
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real- time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lyssenko C, Wheaton F (2006) Impact of rapid impulse operating disturbances on ammonia removal by trickling and submerged-upflow biofilters for intensive recirculating aquaculture. Aquacult Eng 35(1):38–50
Martinez A-S, Cutler CP, Wilson GD et al (2005a) Cloning and expression of three aquaporin homologues from the European eel (Anguilla anguilla): effects of seawater acclimation and cortisol treatment on renal expression. Biol Cell 97:615–627
Martinez A-S, Cutler CP, Wilson GD et al (2005b) Regulation of expression of two aquaporin homologs in the intestine of the European eel: effects of seawater acclimation and cortisol treatment. Am J Physiol Regul Integr Comp Physiol 288:1733–1743. https://doi.org/10.1152/ajpregu.00747.2004
McCormick SD (2001) Endocrine control of osmoregulation in teleost fish. Am Zool 41:781–794
Morgan JD, Iwama GK (1991) Effects of salinity on growth, metabolism, and ion regulation in juvenile rainbow and steelhead trout (Oncorhynchus mykiss) and fall Chinook salmon (Oncorynchus tshawytscha). Can J Fish Aquat Sci 48:2083–2094
Neff BD, Pitcher TE (2005) Genetic quality and sexual selection: an integrated framework for good genes and compatible genes. Mol Ecol 14:19–38. https://doi.org/10.1111/j.1365-294X.2004.02395.x
Okamoto T, Kurokawa T, Gen K et al (2009) Influence of salinity on morphological deformities in cultured larvae of Japanese eel, Anguilla japonica, at completion of yolk resorption. Aquaculture 293:113–118. https://doi.org/10.1016/j.aquaculture.2009.04.005
Okamura A, Yamada Y, Horie N et al (2007) Effects of water temperature on early development of Japanese eel Anguilla japonica. Fish Sci 73:1241–1248. https://doi.org/10.1111/j.1444-2906.2007.01461.x
Okamura A, Yamada Y, Mikawa N et al (2009) Growth and survival of eel leptocephali (Anguilla japonica) in low-salinity water. Aquaculture 296:367–372. https://doi.org/10.1016/j.aquaculture.2009.08.039
Okamura A, Yamada Y, Mikawa N et al (2011) Notochord deformities in reared Japanese eel Anguilla japonica larvae. Aquaculture 317:37–41. https://doi.org/10.1016/j.aquaculture.2011.04.024
Okamura A, Yamada Y, Mikawa N et al (2016) Effect of salinity on occurrence of notochord deformities in Japanese eel Anguilla japonica larvae. Aquac Int 24:549–555. https://doi.org/10.1007/s10499-015-9944-1
Okamura A, Horie N, Mikawa N et al (2018) Influence of temperature and feeding regimes on growth and notochord deformity in reared Anguilla japonica leptocephali. Fish Sci 84:505–512. https://doi.org/10.1007/s12562-018-1188-3
Politis SN, Butts IAE, Tomkiewicz J (2014) Light impacts embryonic and early larval development of the European eel, Anguilla anguilla. J Exp Mar Bio Ecol 461:407–415. https://doi.org/10.1016/j.jembe.2014.09.014
Politis SN, Mazurais D, Servili A et al (2017) Temperature effects on gene expression and morphological development of European eel, Anguilla anguilla larvae. P L O S One 12:e0182726. https://doi.org/10.1371/journal.pone.0182726
Politis SN, Syropoulou E, Benini E et al (2021) Performance thresholds of hatchery produced European eel larvae reared at different salinity regimes. Aquaculture 539:736651. https://doi.org/10.1016/j.aquaculture.2021.736651
Politis SN, Mazurais D, Servili A et al (2018a) Salinity reduction benefits European eel larvae: insights at the morphological and molecular level. PLOS One 13.https://doi.org/10.1371/journal.pone.0198294
Politis SN, Sørensen SR, Mazurais D et al (2018b) Molecular ontogeny of first-feeding European eel larvae. Front Physiol 9.https://doi.org/10.3389/fphys.2018b.01477
Rahmah S, Liew HJ, Napi N, Rahmat SA (2020) Metabolic cost of acute and chronic salinity response of hybrid red tilapia Oreochromis sp. larvae. Aquac Rep 16:100233. https://doi.org/10.1016/j.aqrep.2019.100233
Reinecke M, Björnsson BT, Dickhoff WW et al (2005) Growth hormone and insulin-like growth factors in fish: where we are and where to go. Gen Comp Endocrinol 142:20–24. https://doi.org/10.1016/j.ygcen.2005.01.016
Righton DA, Metcalfe JD (2011) Eel migrations. In: Encyclopedia of fish physiology. Elsevier Inc., pp 1937–1944
Sasai S, Kaneko T, Tsukamoto K (1998) Extrabranchial chloride cells in early life stages of the Japanese eel, Anguilla japonica. Ichthyol Res 45:95–98
Sasai S, Katoh F, Kaneko T, Tsukamoto K (2007) Ontogenic change of gill chloride cells in leptocephalus and glass eel stages of the Japanese eel, Anguilla japonica. Mar Biol 150:487–496. https://doi.org/10.1007/s00227-006-0355-8
Schmidt J (1923) The breeding places of the eel. Philos Trans R Soc Lond Ser B 211:179–208
Scott GR, Richards JG, Forbush B et al (2004) Changes in gene expression in gills of the euryhaline killifish Fundulus heteroclitus after abrupt salinity transfer. Am J Physiol Cell Physiol 287:300–309
Seo MY, Kuroki M, Okamura A et al (2015) Occurrence of larval and adult types of ion-secreting ionocytes in Japanese eel Anguilla japonica. Ichthyol Res 62:487–494. https://doi.org/10.1007/s10228-015-0463-x
Sørensen SR, Skov PV, Lauesen P et al (2014) Microbial interference and potential control in culture of European eel (Anguilla anguilla) embryos and larvae. Aquaculture 426–427:1–8. https://doi.org/10.1016/j.aquaculture.2014.01.011
Sørensen SR, Anthony I, Butts E et al (2016a) Effects of salinity and sea salt type on egg activation, fertilization, buoyancy and early embryology of European eel, Anguilla anguilla. Zygote 24:121–138. https://doi.org/10.1017/S0967199414000811
Sørensen SR, Tomkiewicz J, Munk P et al (2016b) Ontogeny and growth of early life stages of captive-bred European eel. Aquaculture 456:50–61. https://doi.org/10.1016/j.aquaculture.2016.01.015
Tandler A, Anav FA, Choshniak I (1995) The effect of salinity on growth rate, survival and swimbladder inflation in gilthead seabream, Sparus aurata, larvae. Aqua 135:343–353
Tomkiewicz J, Kofoed TM, Pedersen JS (2011) Assessment of testis development during induced spermatogenesis in the European eel Anguilla anguilla. Mar Coast Fish 3(1):106–118. https://doi.org/10.1080/19425120.2011.556902
Tomkiewicz J, Politis SN, Sørensen SR, Butts IAE, Kottmann JS (2019) European eel - an integrated approach to establish eel hatchery technology in Denmark. In: Don A, Coulson P (eds) Eels - biology, monitoring, management, culture and exploitation: Proceedings of the First International Eel Science Symposium. 5M Publishing, pp 340–374
Tse WKF, Au DWT, Wong CKC (2006) Characterization of ion channel and transporter mRNA expressions in isolated gill chloride and pavement cells of seawater acclimating eels. Biochem Biophys Res Commun 346(346):1181–1190. https://doi.org/10.1016/j.bbrc.2006.06.028
Tsukamoto K (1992) Discovery of the spawning area for Japanese eel. Nature 356:789–791
Varsamos S, Nebel C, Charmantier G (2005) Ontogeny of osmoregulation in postembryonic fish: a review. Comp Biochem Physiol Part A 141:401–429. https://doi.org/10.1016/j.cbpb.2005.01.013
Acknowledgements
We would like to thank Paraskevas Koumpiadis and Annika Toth for broodstock husbandry and provision of gametes for the experiment, Eftychia Maria Goniou for embryonic rearing, and Eugenia Capatina, Joanna Miest, and Adrian Loh for assistance in molecular work.
Funding
This study received funding from the Innovation Fund Denmark under grant agreement no. 7076-00125B (ITS-EEL) and from “ENV-Fonden.”
Author information
Authors and Affiliations
Contributions
JT, SP, SS, and IB provided funding, while JT, SP, and SS designed the study. JT established the assisted broodstock protocols providing gametes for the experiment. SS constructed and tailored the larval systems and rearing tanks. ES, SP, and EB conducted the experiment and collected samples. SP and EB carried out the gene expression analysis. IB performed the statistical analysis. SP and SS made illustrations. ES wrote original manuscript draft. All the authors contributed to data interpretation and manuscript revision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
All fish were handled according to the European Union regulations concerning the protection of experimental animals (Dir 86/609/EEC). The experimental protocol for the present study was approved by the Danish Ministry of Food, Agriculture and Fisheries (permit number: 2015–15-0201–00696). In particular, broodstook used were anesthetized with ethyl p-aminobenzoate (benzocaine) prior to any handling, while efforts were made to minimize animal handling stress.
Consent for publication
All the authors approved the submitted version of this manuscript.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Syropoulou, E., Benini, E., Sørensen, S.R. et al. Early and abrupt salinity reduction impacts European eel larval culture. Fish Physiol Biochem 48, 355–366 (2022). https://doi.org/10.1007/s10695-022-01056-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-022-01056-6