Abstract
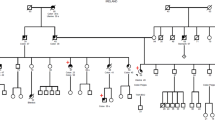
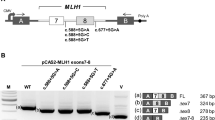
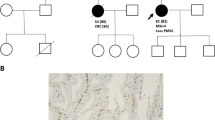
Lynch syndrome (hereditary non-polyposis colorectal cancer) is an inherited disease caused by germ-line mutation in mismatch repair genes such as MLH1, MSH2, and MSH6. The mutations include missense and nonsense mutations, small insertions and deletions, and gross genetic alterations including large deletions and duplications. In addition to these genetic changes, mutations in introns are also involved in the pathogenesis. However, it is sometimes difficult to interpret correctly the pathogenicity of variants in exons as well as introns. To evaluate the effect of splice-site mutations in two Lynch syndrome patients, we carried out a functional splicing assay using minigenes. Consequently, this assay showed that the mutation of c.1731+5G>A in MLH1 led to exon15 skipping, and that the mutation of c.211+1G>C in MSH2 created an activated cryptic splice-site 17-nucleotides upstream in exon1. These aberrant splicing patterns were not observed when wild type sequence was used for the assay. We also obtained concordant results by RT-PCR experiments with transcripts from the patients. Furthermore, additional functional splicing assays using two different intronic mutations described in earlier studies revealed splicing alterations that were in complete agreement with the reports. Therefore, functional splicing assay is helpful for evaluating the effects of genetic variants on splicing.



Similar content being viewed by others
Abbreviations
- HNPCC:
-
Hereditary non-polyposis colorectal cancer
- MLH1 :
-
mutL homolog 1 gene
- MSH2 :
-
mutS homolog 2 gene
- RT-PCR:
-
Reverse transcription-PCR
- NMD:
-
Nonsense-mediated RNA decay
- ESE:
-
Exonic splicing enhancer
- UV:
-
Uncharacterized variant
References
Lynch HT, de la Chapelle A (1999) Genetic susceptibility to non-polyposis colorectal cancer. J Med Genet 36(11):801–818
Peltomaki P (2005) Lynch syndrome genes. Fam Cancer 4(3):227–232
Syngal S, Fox EA, Li C et al (1999) Interpretation of genetic test results for hereditary nonpolyposis colorectal cancer: implications for clinical predisposition testing. JAMA 282(3):281–282
Cartegni L, Chew SL, Krainer AR (2002) Listening to silence and understanding nonsense: exonic mutations that affect splicing. Nat Rev Genet 3(4):285–298
Hentze MW, Kulozik AE (1999) A perfect message: RNA surveillance and nonsense-mediated decay. Cell 96(3):307–310
Tournier I, Vezain M, Martins A et al (2008) A large fraction of unclassified variants of the mismatch repair genes MLH1 and MSH2 is associated with splicing defects. Hum Mutat 29(12):1412–1424
Burge C, Karlin S (1997) Prediction of complete gene structures in human genomic DNA. J Mol Biol 268(1):78–94
Reese MG, Eeckman FH, Kulp D et al (1997) Improved splice site detection in genie. J Comput Biol 4(3):311–323
Hebsgaard SM, Korning PG, Tolstrup N et al (1996) Splice site prediction in Arabidopsis thaliana DNA by combining local and global sequence information. Nucleic Acids Res 24(17):3439–3452
Nalla VK, Rogan PK (2005) Automated splicing mutation analysis by information theory. Hum Mutat 25(4):334–342
Divina P, Kvitkovicova A, Buratti E et al (2009) Ab initio prediction of mutation-induced cryptic splice-site activation and exon skipping. Eur J Hum Genet Jan 14 (Epub ahead of print)
Sahashi K, Masuda A, Matsuura T et al (2007) In vitro and in silico analysis reveals an efficient algorithm to predict the splicing consequences of mutations at the 5′ splice sites. Nucleic Acids Res 35(18):5995–6003
Krüger S, Plaschke J, Jeske B et al (2003) Identification of six novel MSH2 and MLH1 germline mutations in HNPCC. Hum Mutat 21(4):445–446
Casey G, Lindor NM, Papadopoulos N et al (2005) Conversion analysis for mutation detection in MLH1 and MSH2 in patients with colorectal cancer. JAMA 293(7):799–809
Brow DA (2002) Allosteric cascade of spliceosome actination. Annu Rev Genet 36:333–360
Lesser CF, Guthrie C (1993) Mutations in U6 snRNA that alter splice site specificity: implications for the active site. Science 262(5142):1982–1988
Kandels-Lewis S, Seraphin B (1993) Involvement of U6 snRNA in 5′ splice site selection. Science 262(5142):2035–2039
Roca X, Olson AJ, Rao AR et al (2008) Features of 5′-splice-site efficiency derived from disease-causing mutations and comparative genomics. Genome Res 18(1):77–87
Nakai K, Sakamoto H (1994) Construction of a novel database containing aberrant mutations of mammalian genes. Gene 141(2):171–177
Krawczak M, Thomas NS, Hundrieser B et al (2007) Single base-pair substitutions in exon-intron junctions of human genes: nature, distribution, and consequences for mRNA splicing. Hum Mutat 28(2):150–158
Thanaraj TA, Robinson AJ (2000) Prediction of exact boundaries of exons. Brief Bioinform 1(4):343–356
Carmel I, Tal S, Vig I et al (2004) Comparative analysis detects dependencies among the 5′ splice-site positions. RNA 10(5):828–840
Kerber RA, Neklason DW, Samowitz WS et al (2005) Frequency of familial colon cancer and hereditary nonpolyposis colorectal cancer (Lynch syndrome) in a large population database. Fam Cancer 4(3):239–244
Barnetson RA, Tenesa A, Farrington SM et al (2006) Identification and survival of carriers of mutations in DNA mismatch-repair genes in colon cancer. N Enql J Med 354(26):2751–2763
Stella A, Wagner A, Shito K et al (2001) A nonsense mutation in MLH1 causes exon skipping in three unrelated HNPCC families. Cancer Res 61(19):7020–7024
Pagenstecher C, Wehner M, Friedl W et al (2006) Aberrant splicing in MLH1 and MSH2 due to exonic and intronic variants. Hum Genet 119(1–2):9–22
Auclair J, Busine MP, Navarro C et al (2006) Systematic mRNA analysis for the effect of MLH1 and MSH2 missense and silent mutations on aberrant splicing. Hum Mutat 27(2):145–154
Vreeswijk MP, Kraan JN, van der Klift HM et al (2009) Intronic variants in BRCA1 and BRCA2 that affect RNA splicing can be reliably selected by splice-site prediction programs. Hum Mutat 30(1):107–114
Lastella P, Resta N, Miccolis I et al (2004) Site directed mutagenesis of hMLH1 exonic splicing enhancers does not correlate with splicing disruption. J Med Genet 41(6):e72
Sharp A, Pichert G, Lucassen A et al (2004) RNA analysis reveals splicing mutations and loss of expression defects in MLH1 and BRCA1. Hum Mutat 24(3):272
Acknowledgments
This work was supported in part by Grant-in-Aid (#17015009) for Scientific Research from The Ministry of Education, Culture, Sports, Science and Technology Japan, and by a research grant from Mitsui Life Social Welfare Foundation.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10689_2009_9280_MOESM1_ESM.pdf
Supplementary Fig. 1. (A) A schematic representation of plasmids containing wild type or mutant DNA fragment of case 1. (B) RT-PCR analysis of transcripts in cells transfected with wild type or mutant plasmids using murine Gapdh-specific (upper panel) or human GAPDH-specific (lower panel) primers. RT-PCR products from the cells transfected with pcDNAmGapdh1 (lane1), pcDNAmGapdh1-MLH1-ex8wt (lane2), pcDNAmGapdh1-MLH1-ex8mut(G) (lane3), pcDNAmGapdh1-MLH1-ex8mut(C) (lane4), and mock (lane5) were separated on an agarose gel. Negative PCR experiment without DNA is shown in lane6
10689_2009_9280_MOESM2_ESM.pdf
Supplementary Fig. 2. (A) A schematic representation of plasmids containing wild type and mutant DNA fragment of case 2. (B) RT-PCR analysis of transcripts from the cells transfected with wild type or mutant plasmids using MLH1 exon1- and murine Gapdh exon3-specific primers (upper panel) or human GAPDH-specific primers (lower panel). RT-PCR products from the cells transfected with pcDNAmGapdh1 (lane1), pcDNAmGapdh1-MLH1-ex1wt (lane2), pcDNAmGapdh1-MLH1-ex1mut (lane3), and mock (lane4) were separated on an agarose gel. Negative PCR experiment without DNA is shown in lane5. The products of pcDNAmGapdh1-MLH1-ex1mut revealed aberrant bands in addition to the band corresponding to the wild type products
Rights and permissions
About this article
Cite this article
Naruse, H., Ikawa, N., Yamaguchi, K. et al. Determination of splice-site mutations in Lynch syndrome (hereditary non-polyposis colorectal cancer) patients using functional splicing assay. Familial Cancer 8, 509–517 (2009). https://doi.org/10.1007/s10689-009-9280-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10689-009-9280-6