Abstract
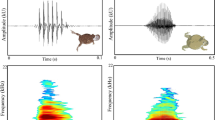
According to the concept of ritualization, acquisition of signal function (i.e., species recognition) leads to formalization of specific signal structure (i.e., species-specific pattern). By contrast, loss of species recognition may result in specific pattern collapse of the signal, from a temporally regular pattern to an irregular form. Several studies have reported the loss of species recognition driven by release from selective pressure against hybridization under isolation from close relatives, but no study has reported collapse of a regular signal pattern that serves in species recognition. In nocturnal lizards of the genus Gekko, several species pairs hybridize when brought together, whereas other species pairs never hybridize, despite being sympatric. I investigated the relationship between the existence of natural hybridization, courtship call similarity and species recognition ability in eight species of Gekko. I found that males of all eight species court using calls, and four of these species have species-specific regular patterns in the courtship calls, whereas the other four have no temporal patterns. In playback experiments, females of the species-specific pattern call species discriminated species by the call. On the other hand, in the patternless call species, females did not discriminate species by the call, suggesting that natural hybridization has resulted from the loss of species recognition ability in the patternless call species. In insular Gekko isolated from congeners, acoustic species recognition might have been lost, followed by pattern collapse of the courtship calls. This unique example of the signal pattern collapse would provide a key to answer basic questions concerning the causes and consequences of signal evolution.








Similar content being viewed by others
References
Andersson M (1994) Sexual selection. Princeton University Press, Princeton
Behm JE, Ives AR, Boughman JW (2010) Breakdown in postmating isolation and the collapse of a species pair through hybridization. Am Nat 175:11–26
Bradbury JW, Vehrencamp SL (1998) Principles of animal communication. Sinauer, Sunderland
Brumm H, Slabbekoorn H (2005) Acoustic communication in noise. Adv Stud Behav 35:151–209
Chen J, Jono T, Cui J, Yue X, Tang Y (2016) The acoustic properties of low intensity vocalizations match hearing sensitivity in the webbed-toed gecko, Gekko subpalmatus. PLoS ONE 11:e0146677
Croucher PJ, Jones RM, Searle JB et al (2007) Contrasting patterns of hybridization in large house spiders (Tegenaria atrica group, Agelenidae). Evolution 61:1622–1640
Dobzhansky T (1940) Speciation as a stage in evolutionary divergence. Am Nat 74:312–321
Ewing AW (1989) Arthropod bioacoustics: neurobiology and behavior. Comstock, Ithaca
Feng J, Han D, Bauer AM, Zhou K (2007) Interrelationships among gekkonid geckos inferred from mitochondrial and nuclear gene sequences. Zool Sci 24:656–665
Frankenberg E (1974) Vocalization of males of three geographical forms of Ptyodactylus from Israel (Reptilia: Sauria: Gekkoninae). J Herpetol 8:59–70
Frankenberg E (1982) Vocal behavior of the Mediterranean house gecko, Hemidactylus turcicus. Copeia 1982:770–775
Fugère V, Krahe R (2010) Electric signals and species recognition in the wave-type gymnotiform fish Apteronotus leptorhynchus. J Exp Biol 213:225–236
Gerhardt HC, Huber F (2002) Acoustic communication in Insects and Anurans: common problems and diverse solutions. University of Chicago Press, Chicago
Gerhardt HC, Klump GM (1988) Masking of acoustic signals by the chorus background noise in the green tree frog: a limitation on mate choice. Anim Behav 36:1247–1249
Gröning J, Hochkirch A (2008) Reproductive interference between animal species. Q Rev Biol 83:257–282
Hibbitts TJ, Whiting MJ, Stuart-Fox DM (2007) Shouting the odds: vocalization signals status in a lizard. Behav Ecol Sociobiol 61:1169–1176
Hochkirch A, Gröning J, Bücker A (2007) Sympatry with the devil: reproductive interference could hamper species coexistence. J Anim Ecol 76:633–642
Hödel G, Gerhardt HC (2003) Reproductive character displacement in the acoustic communication system of green tree frogs (Hyla cinerea). Evolution 57:894–904
Hoskin CJ, Higgie M, McDonald KR et al (2005) Reinforcement drives rapid allopatric speciation. Nature 437:1353–1356
Howard DJ (1993) Reinforcement: origin, dynamics, and fate of an evolutionary hypothesis. In: Harrison RG (ed) Hybrid zones and the evolutionary process. Oxford University Press, New York, pp 46–69
Ikeuchi I (2004) Male and female reproductive cycles of the Japanese gecko, Gekko japonicus, in Kyoto, Japan. J Herpetol 38:269–274
Jono T (in press) Observations of reproductive behavior of Gekko hokouensis and G. vertebralis in captivity. Akamata
Jono T, Inui Y (2012) Secret calls from under the eaves: acoustic behavior of the Japanese house gecko, Gekko japonicus. Copeia 2012:145–149
Kaneshiro KY (1980) Sexual isolation, speciation and the direction of evolution. Evolution 34:437–444
Kaneshiro KY (1983) Sexual selection and direction of evolution in the biosystematics of Hawaiian Drosophilidae. Annu Rev Entomol 28:161–178
Lemmon EM, Lemmon AR (2010) Reinforcement in chorus frogs: lifetime fitness estimates including intrinsic natural selection and sexual selection against hybrids. Evolution 64:1748–1761
Maenosono T, Toda M (2007) Distributions and terrestrial reptiles in the Ryukyu Archipelago: a review of published records. Akamata 18:28–46
Marcellini DL (1974) Acoustic behavior of the gekkonid lizard, Hemidactylus frenatus. Herpetologica 30:44–52
Marcellini DL (1978) The acoustic behavior of lizards. In: Greenberg N, MacLean PD (eds) Behavior and neurobiology of lizards: an interdisciplinary colloquium. National Institute of Mental Health, Bethesda, pp 287–300
Matsuo T (2005) Amphibians and reptiles in Nagasaki Prefecture. Nagasaki Sinbun-Sya, Nagasaki
McPeek MA, Gavrilets S (2006) The evolution of female mating preferences: differentiation from species with promiscuous males can promote speciation. Evolution 60:1967–1980
Morita T (1990) The study of Gekko on Satsuma Peninsula and several neighboring islets. Shizen-Aigo 16:6–9
Okada S, Toda M (1998) New records of Gekko tawaensis from southwest Japan. Hibakagaku 189:1–12
Okada S, Izawa M, Ota H (2002) Growth and reproduction of Gekko hokouensis (Reptilia: Squamata) on Okinawajima Island of the Ryukyu Archipelago, Japan. J Herpetol 36:473–479
Ord TJ, Peters RA, Clucas B et al (2007) Lizards speed up visual displays in noisy motion habitats. Proc R Soc B Biol Sci 274:1057–1062
Ota H, Tanaka S (1996) Gekkonidae and Eublepharidae. In: Sengoku S, Matsui M, Nakata N (eds) The encyclopaedia of animals in Japan 5, amphibians, reptiles, chondrichthyes. Heibonsya, Tokyo, pp 65–71
Pfennig KS, Ryan MJ (2006) Reproductive character displacement generates reproductive isolation among conspecific populations: an artificial neural network study. Proc R Soc B Biol Sci 273:1361–1368
Phelps SM, Rand AS, Ryan MJ (2006) A cognitive framework for mate choice and species recognition. Am Nat 167:28–42
Price JJ (2013) Why is birdsong so repetitive? Signal detection and the evolution of avian singing modes. Behaviour 150:995–1013
R Development Core Team (2011) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Regalado R (2003) Roles of visual, acoustic, and chemical signals in social interactions of the tropical house gecko (Hemidactylus mabouia). Carib J Sci 39:307–320
Robertson HM, Paterson HE (1982) Mate recognition and mechanical isolation in Enallagma damselflies (Odonata: Coenagrionidae). Evolution 1982:243–250
Sætre GP, Moum T, Bures S et al (1997) A sexually selected character displacement in flycatchers reinforces premating isolation. Nature 387:589–592
Scott JL, Kawahara AY, Skevington JH et al (2010) The evolutionary origins of ritualized acoustic signals in caterpillars. Nat Commun 1:1–9
Searcy WA, Nowicki S (2005) The evolution of animal communication: reliability and deception in signaling systems. Princeton University Press, Princeton
Seehausen O, van Alphen JJ, Witte F (1997) Cichlid fish diversity threatened by eutrophication that curbs sexual selection. Science 277:1808–1811
Servedio MR, Noor MA (2003) The role of reinforcement in speciation: theory and data. Annu Rev Ecol Evol Syst 34:339–364
Shibaike Y, Takahashi Y, Arikura I et al (2009) Chromosome evolution in the lizard genus Gekko (Gekkonidae, Squamata, Reptilia) in the East Asian islands. Cytogenet Genome Res 127:182–190
Shibata Y (1983) Two species in ‘Japanese gecko’. Nat Study 29:27–29
Tang Y, Zhuang L, Wang Z (2001) Advertisement calls and their relation to reproductive cycles in Gekko gecko (Reptilia, Lacertilia). Copeia 2001:248–253
Tinghitella RM, Zuk M (2009) Asymmetric mating preferences accommodated the rapid evolutionary loss of a sexual signal. Evolution 63:2087–2098
Toda M (2008) External characters useful to discriminate Gekko hokouensis and its cryptic undescribed species of the Okinawa Group, Japan: a preliminary report. Akamata 19:23–30
Toda M (2011) Natural hybridization among Gekko species in insular East Asia. Bull Herpetol Soc Jpn 2011:76–77
Toda M, Hikida T (2011) Possible incursions of Gekko hokouensis (Reptilia: Squamata) into non-native area: an example from Yakushima Island of the northern Ryukyus, Japan. Curr Herpetol 30:33–39
Toda M, Hikida T, Ota H (2001a) Discovery of sympatric cryptic species within Gekko hokouensis (Gekkonidae; Squamata) from the Okinawa Island, Japan, by use of allozyme data. Zool Scr 30:1–11
Toda M, Okada S, Ota H et al (2001b) Biochemical assessment of evolution and taxonomy of the morphologically poorly diverged geckos, Gekko yakuensis and G. hokouensis (Reptilia: Squamata) in Japan, with special reference to their occasional hybridization. Biol J Linn Soc 73:153–165
Toda M, Hikida T, Okada S et al (2003) Contrasting patterns of genetic variation in the two sympatric geckos Gekko tawaensis and G. japonicus (Reptilia: Squamata) from western Japan, as revealed by allozyme analyses. Heredity 90:90–97
Toda M, Okada S, Hikida T et al (2006) Extensive natural hybridization between two geckos, Gekko tawaensis and Gekko japonicus (Reptilia: Squamata), throughout their broad sympatric area. Biochem Genet 44:1–17
Toda M, Sengoku S, Hikida T et al (2008) Descriptions of two new species of the genus Gekko (Squamata, Gekkonidae) from the Takara and Amami Island Groups in the Ryukyu Archipelago, Japan. Copeia 2:452–466
Tokunaga S (1988) A gecko of the genus Gekko from Taka-shima Island, Hirado, Nagasaki, Japan (Reptilia: Lacertilia). Jap J Herpetol 12:127–130
Uy JAC, Endler JA (2004) Modification of the visual background increases the conspicuousness of golden-collared manakin displays. Behav Ecol 15:1003–1010
Wellenreuther M, Tynkkynen K, Svensson EI (2009) Simulating range expansion: male species recognition and loss of premating isolation in damselflies. Evolution 64:242–252
Werner YL, Chernis E, Granot R (2008) The call of the Sri Lankan golden gecko Calodactylodes illingworthorum, ecological parallel of the fan-toed geckos, genus Ptyodactylus (Reptilia: Sauria: Gekkonidae). Bioacoustics 18:35–49
West-Eberhard MJ (1983) Sexual selection, social competition, and speciation. Q Rev Biol 1983:155–183
Wiley RH (1983) The evolution of communication: information and manipulation. Anim Behav 2:156–189
Wiley RH (2006) Signal detection and animal communication. Adv Stud Behav 36:217–247
Wiley RH, Richards DG (1982) Adaptations for acoustic communication in birds: sound transmission and signal detection. In: Kroodsma DE, Miller EH (eds) acoustic communication in birds, vol 1. Academic Press, New York, pp 132–181
Wollerman L, Wiley RH (2002) Background noise from a natural chorus alters female discrimination of male calls in a Neotropical frog. Anim Behav 63:15–22
Zuk M, Kolluru GR (1998) Exploitation of sexual signals by predators and parasitoids. Q Rev Biol 73:415–438
Zuk M, Rotenberry JT, Tinghitella RM (2006) Silent night: adaptive disappearance of a sexual signal in a parasitized population of field crickets. Biol Lett 2006:521–524
Acknowledgments
I thank A. Mori, M. Toda, H. Numata, M. Hasegawa and T. Morisaka for invaluable comments on data analysis and the manuscript. I also thank A. H. Savitzky and A. M. Bauer for reviewing the manuscript; Y. Tang, I. Ikeuchi and D. Muramatsu for helpful comments on an earlier version of the manuscript, and Y. Koizumi for assistance in maintaining geckos. Geckos were handled under Guideline for Animal Experiments of Kyoto University. This work was supported in part by the Global COE Program (A06) to Kyoto University, the JSPS Research Fellowships for Young Scientists (No. 231388) to T.J. and the Fellowship for Young International Scientists of the Chinese Academy of Sciences (No. Y4J3011100) to T.J.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jono, T. Absence of temporal pattern in courtship signals suggests loss of species recognition in gecko lizards. Evol Ecol 30, 583–600 (2016). https://doi.org/10.1007/s10682-016-9829-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-016-9829-4