Abstract
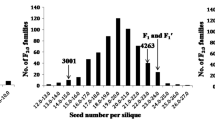
Seed size is directly involved in determining the yield of groundnut. Here, we report the identification of a main QTL for large seed size using a F2 population of ‘TG 89 × ICGV 15007’. A total of 122 F2 individuals were genotyped with a total 345 markers which included 297 of ddRAD-seq SNP, 37 AhMITE, 9 SSR and 2 gene specific markers to construct a genetic linkage map spanning 3891.3 cM. QTL analysis revealed two major QTLs, one on linkage group A05 explaining 18.07% phenotypic variation and the other on A02 explaining 11.95% phenotypic variation. The major additive effect of qHKW_A05 was contributed by mutant allele. It was located between marker AhMITE470 and SNP_05_103140987 having a map interval 8.6 cM, corresponding to 4.53 Mbp (99.18–103.71 Mbp) in the genome. In close proximity to this QTL peak, an ortholog of BIG SEEDS 1 (Arahy.5M7JWE) located at 102.47 Mbp. Arahy.5M7JWE belongs to AhTIFY protein having key role in regulating organ size and growth. The study also reports generation of allele specific PCR based SNP markers and validation of its usage in marker assisted selection.






Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Alyr MH, Pallu J, Sambou A, Nguepjop JR, Seye M, Tossim HA, Djiboune YR, Sane D, Rami JF, Fonceka D (2020) Fine-mapping of a wild genomic region involved in pod and seed size reduction on chromosome A07 in peanut (Arachis hypogaea L.). Genes 11(12):1402. https://doi.org/10.3390/genes11121402
Arya SS, Salve AR, Chauhan S (2016) Peanuts as functional food: a review. J Food Sci Technol 53(1):31–41. https://doi.org/10.1007/s13197-015-2007-9
Badigannavar AM, Mondal S, Bhad PG (2020) Radiation based induced mutagenesis in Trombay groundnuts: Developments and accomplishments. IANCAS Bulletin Vol XV 1:45–49
Bai Y, Meng Y, Huang D, Qi Y, Chen M (2011) Origin and evolutionary analysis of the plant-specific TIFY transcription factor family. Genomics 98:128–136. https://doi.org/10.1016/j.ygeno.2011.05.002
Barmukh R, Roorkiwal M, Dixit GP, Bajaj P, Kholova J, Smith MR, Chitikineni A, Bharadwaj C, Sreeman SM, Rathore A, Tripathi S (2022) Characterization of ‘QTL-hotspot’ introgression lines reveals physiological mechanisms and candidate genes associated with drought adaptation in chickpea. J Exp Bot 73(22):7255–7272. https://doi.org/10.1093/jxb/erac348
Bera SK, Kamdar JH, Kasundra SV, Dash P, Maurya AK, Jasani MD, Chandrashekar AB, Manivannan N, Vasanthi RP, Dobariya KL, Pandey MK, Janila P, Radhakrishnan T, Varshney RK (2018) Improving oil quality by altering levels of fatty acids through marker-assisted selection of ahfad2 alleles in peanut (Arachis hypogaea L.). Euphytica 214(9):162–176. https://doi.org/10.1371/journal.pone.0226252
Bertioli DJ, Jenkins J, Clevenger J, Dudchenko O, Gao D, Seijo G, Leal-Bertioli S, Ren L, Farmer AD, Pandey MK, Samoluk SS (2019) The genome sequence of segmental allotetraploid peanut Arachis hypogaea. Nat Genet 51(5):877–884. https://doi.org/10.1038/s41588-019-0405-z
Byzova MV, Franken J, Aarts MG, De Almeida-Engler J, Engler G, Mariani C, Campagne MMVL, Angenent G (1999) Arabidopsis STERILE APETALA, a multifunctional gene regulating inflorescence, flower, and ovule development. Genes Dev 13(8):1002–1014. https://doi.org/10.1101/gad.13.8.1002
Che R, Tong H, Shi B, Liu Y, Fang S, Liu D, Xiao Y, Hu B, Liu L, Wang H, Zhao M, Chu C (2015) Control of grain size and rice yield by GL2-mediated brassinosteroid responses. Nat Plants 2(1):1–8. https://doi.org/10.1038/nplants.2015.195
Chen W, Jiao Y, Cheng L, Huang L, Liao B, Tang M, Ren X, Zhou X, Chen Y, Jiang H (2016) Quantitative trait locus analysis for pod-and kernel-related traits in the cultivated peanut (Arachis hypogaea L.). BMC Genet 17:25. https://doi.org/10.1186/s12863-016-0337-x
Chu Y, Chee P, Isleib TG, Holbrook CC, Ozias-Akins P (2020) Major seed size QTL on chromosome A05 of peanut (Arachis hypogaea) is conserved in the US mini core germplasm collection. Mol Breed 40(1):1–16. https://doi.org/10.1007/s11032-019-1082-4
Dodia SM, Joshi B, Gangurde SS, Thirumalaisamy PP, Mishra GP, Narandrakumar D, Soni P, Rathnakumar AL, Dobaria JR, Sangh C, Chitikineni A (2019) Genotyping-by-sequencing based genetic mapping reveals large number of epistatic interactions for stem rot resistance in groundnut. Theor Appl Genet 132(4):1001–1016. https://doi.org/10.1007/s00122-018-3255-7
Elshire RJ, Glaubitz JC, Sun Q, Poland SA, Kawamoto K, Buckler ES, Mitchel SE (2011) A robust, simple genotyping-by-sequencing (GBS) approach for high diversity species. PLoS ONE 6:e19379. https://doi.org/10.1371/journal.pone.0019379
FAOSTAT (2020) Food and Agricultural Organization of the United Nations. Data available at https://www.fao.org/faostat/en/#data/QCL. Accessed 17 Jun 2022.
Fonceka D, Tossim HA, Rivallan R, Vignes H, Faye I, Ndoye O, Moretzsohn MC, Bertioli DJ, Glaszmann JC, Courtois B, Rami JF (2012) Fostered and left behind alleles in peanut: interspecific QTL mapping reveals footprints of domestication and useful natural variation for breeding. BMC Plant Biol 12(1):1–6. https://doi.org/10.1186/1471-2229-12-26
Gangurde SS, Wang H, Yaduru S, Pandey MK, Fountain JC, Chu Y, Isleib T, Holbrook CC, Xavier A, Culbreath AK, Ozias-Akins P, Varshney RK, Guo B (2020) Nested-association mapping (NAM)-based genetic dissection uncovers candidate genes for seed and pod weights in peanut (Arachis hypogaea). Plant Biotechnol J 18:1457–1471. https://doi.org/10.1111/pbi.13311
Gayathri M, Shirasawa K, Varshney RK, Pandey MK, Bhat RS (2018) Development of new AhMITE1 markers through genome-wide analysis in peanut (Arachis hypogaea L.). BMC Res Notes 11(1):10
Ge L, Yu J, Wang H, Luth D, Bai G, Wang K, Chen R (2016) Increasing seed size and quality by manipulating BIG SEEDS1 in legume species. Proc Natl Acad Sci 113(44):12414–12419. https://doi.org/10.1073/pnas.1611763113
Hake AA, Shirasawa K, Yadawad A, Nayak SN, Mondal S, Badigannavar AM, Nadaf HL, Gowda MVC, Bhat RS (2017) Identification of transposable element markers associated with yield and quality traits from a large population of independent mutants in peanut (Arachis hypogaea L.). Euphytica 213(12):1–13. https://doi.org/10.1007/s10681-017-2070-6
Hammer Ø, Harper DAT, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontol Electron 4(1):1–9
Huang L, He H, Chen W, Ren X, Chen Y, Zhou X, Xia Y, Wang X, Jiang X, Liao B, Jiang H (2015) Quantitative trait locus analysis of agronomic and quality-related traits in cultivated peanut (Arachis hypogaea L.). Theor Appl Genet 128:1103–1115. https://doi.org/10.1007/s00122-015-2493-1
IBPGR, ICRISAT (1985) Descriptors for Groundnut (Revised). International Board for Plant Genetic Resources, Rome and International Crop Research Institute for the Semi-Arid Tropics, Patancheru, India
Jadhav MP, Patil MD, Hampannavar M, Venkatesh PD, Shirasawa K, Pasupuleti J, Pandey MK, Varshney RK, Bhat RS (2021a) Enhancing oleic acid content in two commercially released peanut varieties through marker-assisted backcross breeding. Crop Sci 61(4):2435–2443
Jadhav MP, Gangurde SS, Hake AA, Yadawad A, Mahadevaiah SS, Pattanashetti SK, Gowda MV, Shirasawa K, Varshney RK, Pandey MK, Bhat RS (2021b) Genotyping-by-sequencing based genetic mapping identified major and consistent genomic regions for productivity and quality traits in peanut. Front Plant Sci 12:1–15. https://doi.org/10.3389/fpls.2021.668020
Kale MD, Mouli C, Murty GSS, Rao MVP (1997) Development of a new groundnut variety “TG-26” by using induced mutants in cross breeding. Mutat Breed Newslett (IAEA) 43:25–27
Kale DM, Badigannavar AM, Murty GSS (2000) Development of new large pod Trombay groundnut (Arachis hypogaea) selections. Indian J Agric Sci 70(6):365–369
Kolekar RM, Sukruth M, Shirasawa K, Nadaf HL, Motagi BN, Lingaraju S, Patil PV, Bhat RS (2017) Marker-assisted backcrossing to develop foliar disease resistant genotypes in TMV 2 variety of peanut (Arachis hypogaea L.). Plant Breeding 136(6):948–953
Kosambi DD (1944) The estimation of map distance from recombination values. Ann Eugen 12:172–175. https://doi.org/10.1111/j.1469-1809.1943.tb02321.x
Li N, Liu Z, Wang Z, Ru L, Gonzalez N, Baekelandt A, Pauwels L, Goossens A, Xu R, Zhu Z, Inzé D (2018) STERILE APETALA modulates the stability of a repressor protein complex to control organ size in Arabidopsis thaliana. PLoS Genet 14:e1007218. https://doi.org/10.1371/journal.pgen.1007218
Luo H, Ren X, Li Z, Xu Z, Li X, Huang L, Zhou X, Chen Y, Chen W, Lei Y, Liao B, Pandey MK, Varshney RK, Guo B, Jiang X, Liu F, Jiang H (2017) Co-localization of major quantitative trait loci for pod size and weight to a 37 cM interval on chromosome A05 in cultivated peanut (Arachis hypogaea L.). BMC Genom 18(1):1–12. https://doi.org/10.1111/pbi.13050
Luo H, Guo J, Ren X, Chen W, Huang L, Zhou X, Chen Y, Liu N, Xiong F, Lei Y, Liao B, Jiang H (2018) Chromosomes A07 and A05 associated with stable and major QTLs for pod weight and size in cultivated peanut (Arachis hypogaea L.). Theor Appl Genet 131:267–282. https://doi.org/10.1007/s00122-017-3000-7
Luo H, Guo J, Yu B, Chen W, Zhang H, Zhou X, Chen Y, Huang L, Liu N, Ren X, Yan L (2021) Construction of ddRADseq-based high-density genetic map and identification of quantitative trait loci for trans-resveratrol content in Peanut seeds. Front Plant Sci 12:644402. https://doi.org/10.3389/fpls.2021.644402
Meng L, Li H, Zhang L, Wang J (2015) QTL IciMapping: integrated software for genetic linkage map construction and quantitative trait locus mapping in biparental populations. Crop J 3(3):269–283. https://doi.org/10.1016/j.cj.2015.01.001
Mondal S, Petwal VC, Badigannavar AM, Bhad PG, Verma VP, Goswami SG, Dwivedi J (2017) Electron beam irradiation revealed genetic differences in radio-sensitivity and generated mutants in groundnut (Arachis hypogaea L.). Appl Radiat Isotopes 122:78–83. https://doi.org/10.1016/j.apradiso.2017.01.016
Nawade B, Mishra GP, Radhakrishnan T, Sangh C, Dobariya JR, Kundu R (2019) Development of high oleic peanut lines through marker-assisted introgression of mutant ahFAD2 alleles and its fatty acid profiles under open-filed and controlled conditions. 3 Biotech 9(6):1–6. https://doi.org/10.1007/s13205-019-1774-9
Pandey MK, Roorkiwal M, Singh VK, Ramalingam A, Kudapa H, Thudi M, Chitikineni A, Rathore A, Varshney RK (2016) Emerging genomic tools for legume breeding: current status and future prospects. Front Plant Sci 7:455. https://doi.org/10.3389/fpls.2016.00455
Patil SH (1975) A new groundnut variety for export. Nuclear India 13:7–8
Peterson BK, Weber JN, Kay EH, Fisher HS, Hoekstra HE (2012) Double digest RADseq: an inexpensive method for de novo SNP discovery and genotyping in model and non-model species. PLoS ONE 7:e37135. https://doi.org/10.1371/journal.pone.0037135
Poland JA, Brown PJ, Sorrells ME, Jannink JL (2012) Development of high-density genetic maps for barley and wheat using a novel two-enzyme genotyping-by-sequencing approach. PLoS ONE 7:e32253. https://doi.org/10.1371/journal.pone.0032253
Schneider M, Gonzalez N, Pauwels L, Inzé D, Baekelandt A (2021) The PEAPOD pathway and its potential to improve crop yield. Trends Plant Sci 26(3):220–236. https://doi.org/10.1016/j.tplants.2020.10.012
Sen S (2022) Genome-wide TIFY family in Arachis hypogaea in the perspective of legume JAZs. J Crop Sci Biotechnol 4:1–24. https://doi.org/10.1007/s12892-022-00145-5
Sen S, DasGupta M (2021) Involvement of Arachis hypogaea Jasmonate ZIM domain/TIFY proteins in root nodule symbiosis. J Plant Res 134(2):307–326. https://doi.org/10.1007/s10265-021-01256-w
Settaluri VS, Kandala CV, Puppala N, Sundaram (2012) Peanuts and their nutritional aspects—a review. Food Nutr Sci 3(12):1644–1650. https://doi.org/10.4236/fns.2012.312215
Sharma VP (2017) Oilseed production in india the problems and prospects. Springer, Ahmedabad, New Delhi, pp 17–54
Shasidhar Y, Variath MT, Vishwakarma MK, Manohar SS, Gangurde SS, Sriswathi M, Sudini HK, Dobariya KL, Bera SK, Radhakrishnan T, Pandey MK, Janila P, Varshney RK (2020) Improvement of three Indian popular groundnut varieties for foliar disease resistance and high oleic acid using SSR markers and SNP array in marker-assisted backcrossing. The Crop J 8:1–15. https://doi.org/10.1016/j.cj.2019.07.001
Shirasawa K, Koilkonda P, Aoki K, Hirakawa H, Tabata S, Watanabe M, Hasegawa M, Kiyoshima H, Suzuki S, Kuwata C, Naito Y (2012a) In silico polymorphism analysis for the development of simple sequence repeat and transposon markers and construction of linkage map in cultivated peanut. BMC Plant Biol 12(1):1–13. https://doi.org/10.1186/1471-2229-12-80
Shirasawa K, Hirakawa H, Tabata S, Hasegawa M, Kiyoshima H, Suzuki S, Sasamoto S, Watanabe A, Fujishiro T, Isobe S (2012b) Characterization of active miniature inverted-repeat transposable elements in the peanut genome. Theor Appl Genet 124:1365–1373
Sun X, Liu D, Zhang X, Li W, Liu H, Hong W, Jiang C, Guan N, Ma C, Zeng H, Xu C (2013) SLAF-seq: an efficient method of large-scale de novo SNP discovery and genotyping using high-throughput sequencing. PLoS ONE 8:e58700. https://doi.org/10.1371/journal.pone.0058700
Toomer OT (2018) Nutritional chemistry of the peanut (Arachis hypogaea). Critical Rev Food Sci Nutr 58(17):3042–3053. https://doi.org/10.1080/10408398.2017.1339015
Varshney RK, Pandey MK, Janila P, Nigam SN, Sudini H, Gowda MVC, Sriswathi M, Radhakrishnan T, Manohar SS, Nagesh P (2014) Marker-assisted introgression of a QTL region to improve rust resistance in three elite and popular varieties of peanut (Arachis hypogaea L.). Theor Appl Genet 127(8):1771–1781. https://doi.org/10.1007/s00122-014-2338-3
Venuprasad R, Aruna R, Nigam SN (2011) Inheritance of traits associated with seed size in groundnut (Arachis hypogaea L.). Euphytica 181:169–177. https://doi.org/10.1007/s10681-011-0390-5
Voorrips RE (2002) Mapchart: software for the graphical presentation of linkage map and QTL. J Hered 93:77–78. https://doi.org/10.1093/jhered/93.1.77
Wang Z, Li N, Jiang S, Gonzalez N, Huang X, Wang Y, Inzé D, Li Y (2016) SCFSAP controls organ size by targeting PPD proteins for degradation in Arabidopsis thaliana. Nat Commun 7(1):1–11. https://doi.org/10.1038/ncomms11192
Wang Z, Huai D, Zhang Z, Cheng K, Kang Y, Wan L, Yan L, Jiang H, Lei Y, Liao B (2018) Development of a high-density genetic map based on specific length amplified fragment sequencing and its application in quantitative trait loci analysis for yield related traits in cultivated peanut. Front Plant Sci 9:827. https://doi.org/10.3389/fpls.2018.00827
Wang Z, Yan L, Chen Y, Wang X, Huai D, Kang Y, Jiang H, Liu K, Lei Y, Liao B (2022) Detection of a major QTL and development of KASP markers for seed weight by combining QTL-seq, QTL-mapping and RNA-seq in peanut. Theor Appl Genet 135(5):1779–1795. https://doi.org/10.1007/s00122-022-04069-0
Wangkumhang P, Chaichoompu K, Ngamphiw C, Ruangrit U, Chanprasert J, Assawamakin A, Tongsima S (2007) WASP: a Web-based Allele-Specific PCR assay designing tool for detecting SNPs and mutations. BMC Genom 8:1–9. https://doi.org/10.1186/1471-2164-8-275
Yang H, Luo L, Li Y, Li H, Zhang X, Zhang K, Zhu S, Li X, Li Y, Wan Y (2023) Fine mapping of qAHPS07 and functional studies of AhRUVBL2 controlling pod size in peanut (Arachis hypogaea L.). Plant Biotechnol J. https://doi.org/10.1111/pbi.14076
Zhang S, Hu X, Miao H, Chu Y, Cui F, Yang W, Wang C, Shen Y, Xu T, Zhao L, Zhang J (2019) QTL identification for seed weight and size based on a high-density SLAF-seq genetic map in peanut (Arachis hypogaea L.). BMC Plant Biol 19(1):1–5. https://doi.org/10.1186/s12870-019-2164-5
Zhao Y, Zhang C, Chen H, Yuan M, Nipper R, Prakash CS, Zhuang W, He G (2016) QTL mapping for bacterial wilt resistance in peanut (Arachis hypogaea L.). Mol Breed 36(2):1–11. https://doi.org/10.1007/s11032-015-0432-0
Zhou X, Guo J, Pandey MK, Varshney RK, Huang L, Luo H, Liu N, Chen W, Lei Y, Liao B, Jiang H (2021) Dissection of the genetic basis of yield-related traits in the Chinese peanut mini-core collection through genome-wide association studies. Front Plant Sci 12:664. https://doi.org/10.3389/fpls.2021.637284
Zhuang W, Chen H, Yang M, Wang J, Pandey MK, Zhang C, Chang WC, Zhang L, Zhang X, Tang R, Garg V (2019) The genome of cultivated peanut provides insight into legume karyotypes, polyploid evolution and crop domestication. Nat Genet 51(5):865–876. https://doi.org/10.1038/s41588-019-0402-2
Zuo JF, Niu Y, Cheng P, Feng JY, Han SF, Zhang YH, Shu G, Wang Y, Zhang YM (2019) Effect of marker segregation distortion on high density linkage map construction and QTL mapping in Soybean (Glycine max L.). Heredity 123(5):579–592. https://doi.org/10.1038/s41437-019-0238-7
Acknowledgements
Authors acknowledge Director, Bioscience Group and Head, Nuclear Agriculture and Biotechnology Division for encouragement and support. This work is a part of an ongoing IAEA-coordinated research project on “Radiation-induced Crop Diversity and Genetic Associations for Accelerating Variety Development” (D24015; IAEA Research Agreement No.: 25064).
Funding
This work is funded by in house funding of Bhabha Atomic Research Centre, Department of Atomic Energy, Government of India.
Author information
Authors and Affiliations
Contributions
All authors developed the plant materials and mapping population used in this study. PGB has performed all the laboratory experiments, linkage map construction, QTL analysis and other data analysis and wrote first draft of the manuscript. SM conceptualize the work, help in data analysis and edited the manuscript. AMB assisted in field experimentations, data analysis and edited the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.

Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bhad, P.G., Mondal, S. & Badigannavar, A.M. A SNP-based linkage mapping revealed a mutant-origin major quantitative trait locus for seed size in A05 chromosome of groundnut (Arachis hypogaea L.). Euphytica 219, 85 (2023). https://doi.org/10.1007/s10681-023-03212-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10681-023-03212-2