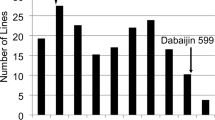
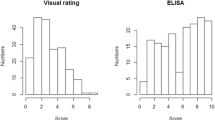
Abstract
Brown spot (BS) is a destructive foliar disease in tobacco (Nicotiana tabacum L.) and is caused by Alternaria alternata. BS poses a serious threat to tobacco production worldwide. To develop molecular markers that are tightly linked to BS resistance for marker-assisted selection (MAS), F2, F2:3 and BC3F2:3 populations were developed from a cross between a source of BS resistance Jingyehuang (JYH) and a BS susceptible flue-cured variety NC82. One hundred eighty-one F2 individuals, 180 F2:3 lines and 256 BC3F2:3 lines were evaluated for field resistance under different environments and quantitative trait loci (QTL) were identified by linkage mapping. A major QTL was mapped on chromosome15; this QTL explained 8.6–18.0% of the phenotypic variation under different conditions. Furthermore, 219 accessions were evaluated for their responses to BS at two sites, and association mapping (AM) was used to verify the chromosomal region harboring the major QTL. The AM results showed that six significant marker-trait associations were detected at two sites. Among these markers, the marker Indel53 within the specific chromosomal region exhibited the most significant association with resistance to BS and explained 20.0 and 21.5% of the phenotypic variation at the two sites, respectively. An approximately 2-Mb physical interval at the locus of marker Indel53 contained 31 predicted genes; quantitative real-time PCR results suggested that two of these genes (Nitab 4.5_0000264g0050.1 and Nitab 4.5_0000264g0130.1) were probable candidate genes for resistance to BS. In summary, our results suggested that the novel major QTL from tobacco variety JYH for resistance to BS provided partial effective resistance against A. alternata and was useful for MAS of resistance to BS in tobacco breeding.





Similar content being viewed by others
References
Bindler G, Vand HR, Gunduz I, Plieske J, Ganal M, Rossi L, Gadani F, Donini P (2007) A microsatellite marker based linkage map of tobacco. Theor Appl Genet 114:341–349
Bindler G, Plieske J, Bakaher N, Gunduz I, Ivanov N, Vand HR, Ganal M, Donini P (2011) A high density genetic map of tobacco (Nicotiana tabacum L.) obtained from large scale microsatellite marker development. Theor Appl Genet 123:219–230
Bolwell GP, Wojtaszek P (1997) Mechanisms for the generation of reactive oxygen species in plant defence: a broad perspective. Physiol Mol Plant Pathol 51:347–366
Bradbury PJ, Zhang Z, Kroon DE, Casstevens TM, Ramdoss Y, Buckler ES (2007) TASSEL: software for association mapping of complex traits in diverse samples. Bioinformatics 23:2633–2635
Broggi LE, González HHL, Resnik SL, Pacin A (2007) Alternaria alternata prevalence in cereal grains and soybean seeds from Entre Rios, Argentina. Rev Iberoam Micol 24:47–51
Chaplin JF (1966) Comparison of tobacco black shank (Phytophthora parasitica var. nicotianae) resistance from four sources. Tob Sci 10:55–58
Cheng LR, Wang Y, Meng LJ, Hu X, Cui YR, Sun Y, Zhu LH, Ali J, Xu JL, Li ZK (2012) Identification of salt-tolerant QTLs with strong genetic background effect using two sets of reciprocal introgression lines in rice. Genome 55:45–55
Cheng LR, Yang AG, Jiang CH, Ren M, Zhang Y, Feng QF, Wang SM, Guan YS, Luo CG (2015) Quantitative trait loci mapping for plant height in tobacco using linkage and association mapping methods. Crop Sci 55:641–647
Chung KR (2012) Stress response and pathogenicity of the necrotrophic fungal pathogen Alternaria alternata. Scientifica 2012:635431
Churchill GA, Doerge RW (1994) Empirical threshold values for quantitative trait mapping. Genetics 138:963–971
Duan S, Xiao M, Wei C, Wan W, He Y, Ma X, Ma Y, Ni L, Tan Y, Wang Y (2016) Transcriptomic profile of tobacco in response to Alternaria longipes and Alternaria alternata infections. Sci Rep UK 6:25635
Edwards KD, Fernandez-Pozo N, Drake-Stowe K, Humphry M, Evans AD, Bombarely A, Allen F, Hurst R, White B, Kernodle SP, Bromley JR, Sanchez-Tamburrino JP, Lewis RS, Mueller LA (2017) A reference genome for Nicotiana tabacum enables map-based cloning of homeologous loci implicated in nitrogen utilization efficiency. BMC Genom 18:448
Gao F, Han X, Wu J, Zheng S, Shang Z, Sun D, Zhou R, Li B (2012) A heat-activated calcium-permeable channel—Arabidopsis cyclic nucleotide-gated ion channel 6—is involved in heat shock responses. Plant J 70:1056–1069
Giancaspro A, Giove SL, Zito D, Blanco A, Gadaleta A (2016) Mapping QTLs for fusarium head blight resistance in an interspecific wheat population. Front Plant Sci 7:1381
Guo YF, Zhu XC, Kong FY, Shi JK, Wang N (1997) Comparison of resistance in various brown spot resistant varieties. China Tob Sci 1:1–6
Guo YF, Zhu XC, Shi JK, Kong FY, Wang N, Wang CL (1998) Genetic studies on resistance to brown spot disease in tobacco. China Tob Sci 3:1–6
Guo YF, Shi JK, Kong FY, Wang N, Wang CL, He JM, Zhu XC (2000) Diallel cross analysis on inheritance of resistance components to tobacco brown spot disease. China Tob Sci 21:17–20
Julio E, Denoyes-Rothan B, Verrier JL, de Borne F (2006) Detection of QTLs linked to leaf and smoke properties in Nicotiana tabacum based on a study of 114 recombinant inbred lines. Mol Breed 18:69–91
Kushalappa AC, Yogendra Kalenahalli N, Karre KN (2016) Plant innate immune response: qualitative and quantitative resistance. Crit Rev Plant Sci 35:38–55
Lewis RS, Milla SR, Kernodle SP (2007) Analysis of an introgressed Nicotiana tomentosa genomic region affecting leaf number and correlated traits in Nicotiana tabacum. Theor Appl Genet 114:841–854
Li ZK, Yu SB, Lafitte HR, Huang N, Courtois B, Hittalmani S, Vijayakumar CH, Liu GF, Wang GC, Shashidhar HE, Zhuang JY, Zheng KL, Singh VP, Sidhu JS, Srivantaneeyakul S, Khush GS (2003) QTL × environment interactions in rice. I. heading date and plant height. Theor Appl Genet 108:141–153
Li H, Ye G, Wang J (2007) A modified algorithm for the improvement of composite interval mapping. Genetics 175:361–374
Lu YL, Zhang SH, Shah T, Xie CX, Hao ZF, Li XH, Farkhari M, Ribaut JM, Cao MJ, Rong TZ, Xu YB (2010) Joint linkage-linkage disequilibrium mapping is a powerful approach to detecting quantitative trait loci underlying drought tolerance in maize. Proc Natl Acad Sci USA 107:19585–19590
Lu Y, Wang MN, Chen XM, See D, Chao SM, Jing JX (2014) Mapping of Yr62 and a small-effect QTL for high-temperature adult-plant resistance to stripe rust in spring wheat PI 192252. Theor Appl Genet 127:1449–1459
Ma W, Berkowitz GA (2011) Ca2+ conduction by plant cyclic nucleotide gated channels and associated signaling components in pathogen defense signal transduction cascades. New Phytol 190:566–572
Meena M, Zehra A, Dubey MK, Aamir M, Gupta VK, Upadhyay RS (2016) Comparative evaluation of biochemical changes in tomato (Lycopersicon esculentum Mill.) infected by Alternaria alternata and its toxic metabolites (TeA, AOH, and AME). Front. Plant Sci 7:1408
Meng L, Li H, Zhang L, Wang J (2015) QTL IciMapping: integrated software for genetic linkage map construction and quantitative trait locus mapping in biparental populations. Crop J 3:269–283
Moller IM, Jensen PE, Hansson A (2007) Oxidative modifications to cellular components in plants. Annu Rev Plant Biol 58:459–481
Nakano H, Sasaki K, Mine Y, Takahata K, Lee O, Sugiyama N (2016) Quantitative trait loci (QTL) controlling plant architecture traits in a Solanum lycopersicum × S-pimpinellifolium cross. Euphytica 211:353–367
Nishi T, Tajima T, Noguchi S, Ajisaka H, Negishi H (2003) Identification of DNA markers of tobacco linked to bacterial wilt resistance. Theor Appl Genet 106:765–770
Rotem J, Eidt J, Wendt U, Kranz J (1988) Relative effects of Alternaria alternata and A. macrospora on cotton crops in Israel. Plant Pathol 37:16–19
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc 3:1101–1108
Sierro N, Battey JND, Ouadi S, Bakaher N, Bovet L, Willig A, Goepfert S, Peitsch MC, Ivanov NV (2014) The tobacco genome sequence and its comparison with those of tomato and potato. Nat Commun 5:3833
Simmons EG (2003) Alternaria themes and variations (310–335) species on malvaceae. Mycotaxon 88:163–217
Slavov S, Mayama S, Atanassov A (2004) Toxin production of Alternaria Alternata tobacco pathotype. Biotechnol Biotechnol Equip 18:90–95
Stavely JR, Chaplin JF, Gwynn GR (1984) Registration of BEL 921 brown spot resistant flue-cured tobacco germplasm. Crop Sci 24:830–831
Tong ZJ, Jiao TL, Wang FQ, Li MY, Leng XD, Gao YL, Li YP, Xiao BG, Wu WR (2012) Mapping of quantitative trait loci conferring resistance to brown spot in flue-cured tobacco (Nicotiana tabacum L.). Plant Breed 131:335–339
Van Ooijen J (2011) Multipoint maximum likelihood mapping in a full-sib family of an outbreeding species. Genet Res 93(5):343–349
Vontimitta V, Lewis RS (2012a) Growth chamber evaluation of a tobacco ‘Beinhart 1000’ × ‘Hicks’ mapping population for quantitative trait loci affecting resistance to multiple races of Phytophthora nicotianae. Crop Sci 52:91–98
Vontimitta V, Lewis RS (2012b) Mapping of quantitative trait loci affecting resistance to Phytophthora nicotianae in tobacco (Nicotiana tabacum L.) line Beinhart-1000. Mol Breed 29:89–98
Voorrips RE (2002) MapChart: software for the graphical presentation of linkage maps and QTLs. J Hered 93:77–78
Wang H, Huang Y, Wang J, Chen X, Wei K, Wang M, Shang S (2016a) Activities of azoxystrobin and difenoconazole against Alternaria alternata and their control efficacy. Crop Prot 90:54–58
Wang Q, Wang L, Zhu G, Cao K, Fang W, Chen C, Wang X (2016b) DNA marker-assisted evaluation of fruit acidity in diverse peach (Prunus persica) germplasm. Euphytica 210(3):413–426
Wilkinson SR, John MK (2003) The role of glutathione peroxidases in trypanosomatids. Biol Chem 384:517–525
Wu M, Li Y, Chen D, Liu H, Zhu D, Yan X (2016) Genome-wide identification and expression analysis of the IQD gene family in moso bamboo (Phyllostachys edulis). Sci Rep UK 6:24520
Xu H, Wang Y, Zhao P, Zhang Y, Xu R, Li D (2011) A cAMP-dependent protein kinase gene, aapk1, is required for mycelia growth, toxicity and pathogenicity of Alternaria alternata on tobacco. J Phytopathol 159:208–216
Yin C, Li H, Li S, Xu L, Zhao Z, Wang J (2015) Genetic dissection on rice grain shape by the two-dimensional image analysis in one japonica × indica population consisting of recombinant inbred lines. Theor Appl Genet 128:1969–1986
Zhang L, Li H, Li Z, Wang J (2008) Interactions between markers can be caused by the dominance effect of quantitative trait loci. Genetics 180(2):1177–1190
Acknowledgements
This work was supported by grants from the Agricultural Science and Technology Innovation Program (ASTIP–TRIC01).
Author information
Authors and Affiliations
Contributions
MMS, CGZ, CHJ and YSZ performed the experiment. LRC and MR analyzed the experimental data. CHJ, CGZ, MMS, YZ, DL, QZ, RMG and XLH participated in the field trails. YYW, AGY and LRC designed the experiment. LRC and YSZ wrote this manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
All authors have read the manuscript and declare that they have no conflict of interest.
Ethical standards
We declare that these experiments comply with the ethical standards in China.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material S1
Schematic diagram of QTL mapping procedure for resistance to BS disease in this study (TIFF 24836 kb)
Supplementary material S2
List of the 219 tobacco accessions (XLSX 15 kb)
Supplementary material S3
Information of polymorphic Indel markers in the F2:3 population for LM (XLSX 10 kb)
Supplementary material S4
Marker information in the BC3F2:3 population for LM (XLSX 9 kb)
Supplementary material S5
Information of Indel markers for AM (XLSX 10 kb)
Supplementary material S6
The results of qRT-PCR of 31 annotation genes in the region of interest (XLSX 15 kb)
Rights and permissions
About this article
Cite this article
Sun, M., Cheng, L., Jiang, C. et al. Identification of a major QTL affecting resistance to brown spot in tobacco (Nicotiana tabacum L.) via linkage and association mapping methods. Euphytica 214, 195 (2018). https://doi.org/10.1007/s10681-018-2244-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10681-018-2244-x