Abstract
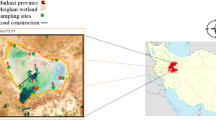
The microalga Dunaliella salina has been broadly studied for different purposes such as beta-carotene production, toxicity assessment and salinity tolerance, yet research on the habitat suitability of this alga has rarely been reported. The present research aims to apply a suitable monitoring and modelling methods (two critical steps in ecological researches) to predict the abundance of D. salina. The abundance of D. salina was predicted by decision tree model (J48 algorithm) in 10 different monitoring sites during 1-year study period (2016–2017) in the Meighan wetland, one of the valuable hypersaline wetlands in Iran. The abundance of alga (as output of model) together with various water quality and physical-habitat wetland characteristics (as inputs of model) were monthly and repeatedly monitored in two different depths (one from the surface layer and another one from the depth of maximum 50 cm) which in total resulted in 240 instances (120 instances for each depth). Based on trial and error, a sevenfold cross-validation resulted in the highest predictive performances of the model (CCI > 75% and Cohen’s Kappa > 0.65). According to the model’s prediction, the number of sunny hours might be one of the most important driving parameters to meet the habitat requirements of alga in the hypersaline wetland. Model also predicted that an increase in dissolved oxygen and sodium concentrations might increase the abundance of D. salina in the salt wetland. In contrast, an increase in total suspended solids concentration and monthly precipitation might lead to a decrease in the abundance of alga. Chi-square test of independence showed a significant difference between the abundance of the D. salina and spatio-temporal patterns in the wetland (Pearson chi-square statistic = 221.7, p = 0.001) so warm seasons (spring and summer) had more contribution to the sampling of the species than cold seasons (autumn and winter). The difference in the abundance of the species in different sampling sites can be attributed due to the various anthropogenic activities.
Access this article
We’re sorry, something doesn't seem to be working properly.
Please try refreshing the page. If that doesn't work, please contact support so we can address the problem.






Similar content being viewed by others
References
Al Dhaheri, S., & Saji, A. (2013). Water quality and brine shrimp (Artemia sp.) population in Al Wathba Lake, Al Wathba Wetland Reserve, Abu Dhabi Emirate, UAE. International Journal of Biodiversity and Conservation, 5, 281–288.
Ambelu, A., Lock, K., & Goethals, P. (2010). Comparison of modelling techniques to predict macroinvertebrate community composition in rivers of Ethiopia. Ecological Informatics, 5, 147–152.
Amini, M., Amini Khoei, Z., & Erfanifar, E. (2019). Nitrate (NO3 −) and phosphate (PO4 3−) removal from aqueous solutions by microalgae Dunaliella salina. Biocatalysis and Agricultural Biotechnology, 19, 101097. https://doi.org/10.1016/j.bcab.2019.101097.
APHA/AWWA/WEF, (1998). Standard methods for the examination of water and wastewater, 19th ed. Washington, DC, USA.
Basil, J. A., Premkumar, D. R. D., Lipton, A. P., & Marian, M. P. (1987). Artemia in the salt pans of Vedaranyam, southern India. In P. Sorgeloos, D. A. Bengtson, W. Decleir, & E. Jaspers (Eds.), Artemia research and its application. Vol. 3, ecology, culturing, use in aquaculture (pp. 141–143). Wetteren, Belgium: Universa Press.
Bellinger, E. G. (1992). A key to common algae: freshwater, estuarine and some coastal species (4th ed.). London: Institution of Water Engineers and Scientists 95 p.
Ben Naceur, H., Ben Rejeb Jenhani, A., & Romdhane, M. S. (2012). Impacts of salinity, temperature, and pH on the morphology of Artemia salina (Branchiopoda: Anostraca) from Tunisia. Zoological Studies, 51, 453–462.
Bhosale, P. (2004). Environmental and cultural stimulants in the production of carotenoids from microorganisms. Applied Microbiology and Biotechnology, 63, 351–361.
Bonnefond, H., Moelants, N., Talec, A., Bernard, O., & Sciandra, A. (2016). Concomitant effects of light and temperature diel variations on the growth rate and lipid production of Dunaliella salina. Algal Research, 14, 72–78.
Borowitzka, M. A. (2013). High-value products from microalgae-their development and commercialisation. Journal of Applied Phycology, 25, 743–756.
Borowitzka, M. A., Borowitzka, L. J., & Kessly, D. (1990). Effects of salinity increase on carotenoid accumulation in the green alga Dunaliella salina. The Journal of Applied Phycology, 2, 111–119.
Borowitzka, M.A., & Borowitzka, L.J. (1988). Limits to growth and carotenogenesis in laboratory and large-scale outdoor cultures of Dunaliella salina. In: Stadler, T., Mollion, J., Verdus, M.C.,
Breiman, L., Friedman, J., Olshen, R., & Stone, C. (1984). 'Classification and regression trees.' (Wadsworth and Brooks: Pacific Groove CA, USA).
Brown, A. D., & Borowitzka, L. J. (1979). Halotolerance of Dunaliella. In M. Levandowsky & S. H. Hutner (Eds.), Biochemistry and physiology of protozoa (2nd ed., pp. 1,139–1,190). New York: Academic Press.
Chen, H., Jiang, J. G., & Wu, G. H. (2009). Effects of salinity changes on the growth of Dunaliella salina and its isozyme activities of glycerol-3-phosphate dehydrogenase. Journal of Agricultural and Food Chemistry, 57, 6178–6182.
Chen, H., & Jiang, J. G. (2009). Osmotic responses of Dunaliella to the changes of salinity. The Journal of Cellular Physiology, 219, 251–258.
Cifuentes, A. S., González, M. A., & Parra, O. O. (1996). The effect of salinity on the growth and carotenogenesis in two Chilean strains of Dunaliella salina Teodoresco. Biological Research, 29, 227–236.
Coesel, S. N., Baumgartner, A. C., Teles, L. M., Ramos, A., Henriques, N. M., Cancela, L., & Varela, J. C. S. (2008). Nutrient limitation is the main regulatory factor for carotenoid accumulation and for Psy and Pds steady state transcript levels in Dunaliella salina (Chlorophyta) exposed to high light and salt stress. Marine Biotechnology, 10, 602–611.
Cohen, J. (1960). A coefficient of agreement for nominal scale. Educational and Psychological Measurement, 20, 37–46.
Cowan, A. K., Rose, P. D., & Horne, L. G. (1992). Dunaliella salina: a model system for studying the response of plant cells to stress. Journal of Experimental Botany, 43, 1535–1547.
European Council (2000) Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a framework for Community action in the field of water policy. Official Journal of the European Communities L327, 22 December 2000, Brussels.
El-Baz, F. K., Aly, H. F., & Salama, A. A. A. (2020). Toxicity assessment of the green Dunaliella salina microalgae. Toxicology Reports, 6, 850–861.
Everaert, G., Bennetsen, E., & Goethals, P. (2016). An applicability index for reliable and applicable decision trees in water quality modelling. Ecological Informatics, 32, 1–6.
Fu, W., Paglia, G., Magnúsdóttir, M., Steinarsdóttir, E. A., Gudmundsson, S., Palsson, B. Ø., Andrésson, Ó. S., & Brynjólfsson, S. (2014). Effects of abiotic stressors on lutein production in the green microalga Dunaliella salina. Microbial Cell Factories, 13, 1–9. https://doi.org/10.1186/1475-2859-13-3.
Fielding, A. H., & Bell, J. F. (1997). A review of methods for the assessment of prediction errors in conservation presence/absence models. Environmental Conservation, 24, 38–49.
Gallego-Cartagena, E., Castillo-Ramírez, M., & Martínez-Burgos, W. (2019). Effect of stressful conditions on the carotenogenic activity of a Colombian strain of Dunaliella salina. Saudi Journal of Biological Sciences, 26, 1325–1330.
Ghasemzadeh, F., Matinfar, A., Jamili, S., & Zare, A. (2005). Study of Artemia sp. production in Gonabad salt-waters, Kavir-e-Namak basin, eastern Iran. Iranian Journal of Fisheries Sciences, 14, 99–118 (in Farsi).
Gerloff, J., & Cholnoky, B. J. (1970). Diatomaceae II. Berlin: J. Cramer Verlag.
Gomez, P. I., & Gonzalez, M. A. (2005). The effect of temperature and irradiance on the growth and carotenogenic capacity of seven strains of Dunaliella salina (Chlorophyta) cultivated under laboratory conditions. Biological Research, 3, 151–162.
Hesami, H., Zarkami, R., & Agh, N. (2018). Habitat suitability of Artemia parthenogenetica in the Meighan wetland (Markazi province) using multivariate analysis. Journal of Animal Researches, 30, 552–563 (in Farsi).
Hoang, T. H., Lock, K., Mouton, A., & Goethals, P. L. M. (2010). Application of classification trees and support vector machines to model the presence of macro invertebrates in rivers in Vietnam. Ecological Informatics, 5, 140–146.
Jin, E., & Polle, J. E. W. (2009). Carotenoid biosynthesis in Dunaliella (Chlorophyta). In A. Ben-Amotz, J. E. W. Polle, & D. V. Subba Rao (Eds.), The alga Dunaliella (pp. 147–172). Einfield, NH: Biodiversity, Physiology Genomics and Biotechnology. Science Publishers.
Katz, A., Jimenez, C., & Pick, U. (1995). Isolation and characterization of a protein associated with carotene globules in the alga Dunaliella bardawil. Plant Physiology, 108, 1657–1664.
Lock, K., & Goethals, P. (2012). Habitat suitability modelling for mayflies (Ephemeroptera) in Flanders (Belgium). Ecological Informatics, 17, 30–35.
Oren, A. (2014). The ecology of Dunaliella in high-salt environments. Journal of Biological Research (Thessaloniki), 21, 23. https://doi.org/10.1186/s40709-014-0023-y.
Padmaja, T. D. (1972). Studies on coccoid blue-green algae, II. In T. V. Desikachary (Ed.), Taxonomy and biology of blue-green algae (pp. 75–127). Chennai: University of Madras Press.
Persoone, G., & Sorgeloos, P. (1980). General aspects of the ecology and biogeography of Artemia. In G. Persoone et al. (Eds.), The brine shrimp Artemia: Proceedings of the international symposium on the brine shrimp Artemia salina, Corpus Christi, Texas, USA, August 20-23, 1979: 3. Ecology, culturing, use in aquaculture (pp. 3–24).
Pick, U. (1998). Dunaliella: a model extremophilic alga. The Israel Journal of Plant Sciences, 46, 131–139.
Polle, J. E. W., Tran, D., & Ben-Amotz, A. (2009). History, distribution, and habitats of algae of the genus Dunaliella Teodoresco (Chlorophyceae). In A. Ben-Amotz, J. E. W. Polle, & D. V. Subba Rao (Eds.), The alga Dunaliella (pp. 1–14). Einfield, NH: Biodiversity, Physiology Genomics and Biotechnology. Science Publishers.
Quinlan, J. R. (1993). C4.5, Program for machine learning, pp. 302. San Francisco: Morgan Kaufmann Publishers.
Ramos, A. A., Polle, J., Tran, D., Cushman, J. C., Jin, E., & Varela, J. C. (2011). The unicellular green alga Dunaliella salina Teod. s a model for abiotic stress tolerance: genetic advances and future perspectives. Algae, 26(1), 3–20.
Saha, S. K., Kazipet, N., & Murray, P. (2018). The carotenogenic Dunaliella salina CCAP 19/20 produces enhanced levels of carotenoid under specific nutrients limitation. BioMed Research International, 2018(1), 1–11. https://doi.org/10.1155/2018/7532897.
Sui, Y., & Vlaeminck, S. E. (2018). Effects of salinity, pH and growth phase on the protein productivity by Dunaliella salina. Journal of Chemical Technology & Biotechnology, 94, 1032–1040.
Sadeghi, R., Zarkami, R., Sabetraftar, K., & Van Damme, P. (2012a). Use of support vector machines (SVMs) to predict distribution of an invasive water fern Azolla filiculoides (Lam.) in Anzali wetland, southern Caspian Sea, Iran. Ecological Modelling, 244, 117–126.
Sadeghi, R., Zarkami, R., Sabetraftar, K., & Van Damme, P. (2012b). Application of classification trees to model the distribution pattern of a new exotic species Azolla filiculoides (Lam.) in Selkeh Wildlife Refuge, Anzali wetland, Iran. Ecological Modelling, 243, 8–17.
Sadeghi, R., Zarkami, R., Sabetraftar, K., & Van Damme, P. (2013). Application of genetic algorithm and greedy stepwise to select input variables in classification tree models for the prediction of habitat requirements of Azolla filiculoides (lam.) in Anzali wetland, Iran. Ecological Modelling, 251, 44–53.
Sadeghi, R., Zarkami, R., & Van Damme, P. (2014). Modelling habitat preference of an alien aquatic fern, Azolla filiculoides (Lam.), in Anzali wetland (Iran) using data-driven methods. Ecological Modelling, 284, 1–9.
Shaish, A., Ben-Amotz, A., & Avron, M. (1992). Biosynthesis of β-carotene in Dunaliella. Methods in Enzymology, 213, 439–444.
Sorgeloos, P., & Lavens, P. (2004). Manual on the production and use of live food for aquaculture (FAO) fisheries technical paper, No. 361. Rome, FAO. 295p. Belgium: Laboratory of Aquaculture and Artemia Reference Center University of Ghent.
Sorgeloos, P., Lavens, P., Leger, P., Tackaert, W., & Versichele, D. (1986). Manual for the culture and use of brine shrimp Artemia in aquaculture. Ghent, Belgium: State University of Ghent 319 pp.
Thuiller, W., Lafourcade, B., Engler, R., & Araújo, M. (2009). BIOMOD -a platform for ensemble forecasting of species distributions. Ecography., 32, 369–373.
Tourenq, C., Barcelo, I., & Drew, C. (2004). Annual report on water quality and Artemia monitoring at Al Wathba (p. 28). Internal Report: TERC-ERWDA.
Vorst, P., Baard, R. L., Mur, L. R., Korthals, H. J., & van den Ende, H. (1994). Effect of growth arrest on carotene accumulation and photosynthesis in Dunaliella. Microbiology, 140, 1411–1417.
Wang, Y., Zhang, C. H., Lin, M. M., & Ge, Y. (2016). A symbiotic bacterium differentially influences arsenate absorption and transformation in Dunaliella salina under different phosphate regimes. Journal of Hazardous Materials, 318, 443–451.
Wayne, A., Wurtsbaugh, W. A., & Gliwicz, Z. M. (2001). Limnological control of brine shrimp population dynamics and cyst production in the Great Salt Lake, Utah. Hydrobiologia, 466, 119–132.
Williams, W. D. (2002). Environmental threats to salt lakes and the likely status of inland saline ecosystems in 2025. Environmental Conservation, 29, 154–167.
Witten, I. H., Frank, E., & Mark, A. (2011). Data Mining: Practical machine learning tools and techniques (3rd ed.p. 629). San Francisco: Morgan Kaufmann.
Xu, Y., Ibrahim, I. M., & Harvey, P. J. (2016). The influence of photoperiod and light intensity on the growth and photosynthesis of Dunaliella salina (chlorophyta) CCAP 19/30. Plant Physiology and Biochemistry, 106, 305–315.
Zarkami, R., Goethals, P., & De Pauw, N. (2010). Use of classification tree methods to study the habitat requirements of tench (Tinca tinca) (L., 1758). Caspian Journal of Environmental Science, 8, 55–63.
Zarkami, R., Sadeghi, R., & Goethals, P. (2012). Use of fish distribution modelling for river management. Ecological Modelling, 230, 44–49.
Zarkami, R., Sadeghi, R., & Goethals, P. (2014). Modelling occurrence of roach “Rutilus rutilus” in streams. Aquatic Ecology, 48, 161–177.
Zarkami, R., Moradi, M., Sadeghi, R., Bani, A., & Abbasi, K. (2018). Input variable selection with greedy stepwise search algorithm for analysing the probability of fish occurrence: a case study for Alburnoides mossulensis in the Gamasiab River, Iran. Ecological Engineering, 118, 104–110.
Zarkami, R., Darizin, Z., Sadeghi, R., Bani, A., & Ghane, A. (2019). Use of data-driven model to analyse the occurrence patterns of an indicator fish species in river: a case study for Alburnoides eichwaldii (De Filippi, 1863) in Shafaroud River, north of Iran. Ecological Engineering, 133, 10–19.
Acknowledgements
The authors would like to thank Pourya Bahri for building the satellite imagery of sampling sites. He is currently following his Master of Science study in the department of environmental science, faculty of natural resources, University of Guilan, Iran.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zarkami, R., Hesami, H. & Sadeghi Pasvisheh, R. Assessment, monitoring and modelling of the abundance of Dunaliella salina Teod in the Meighan wetland, Iran using decision tree model. Environ Monit Assess 192, 172 (2020). https://doi.org/10.1007/s10661-020-8148-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-020-8148-y