Abstract
European canker is one of the most devastating fungal diseases of apple in most temperate regions. The causal agent, Neonectria ditissima, infects trees through wounds in the bark forming cankers that girdle the stem and eventually cause tree death. Timely protection of the trees is challenged by stagnation of symptom expression after infections for a long period of time. The objective of this research is to use a novel TaqMan PCR assay to detect and quantify N. ditissima during the asymptomatic colonization of apple wood. Pruning wounds on branches of the cultivars Elstar and Gala were inoculated with N. ditissima and wood discs were sampled at 2–6, 10–14, and 30–34 mm distance from the inoculation site after 3 hours, 2 weeks, 4 weeks and 8 weeks for the detection and quantification of the pathogen. The TaqMan PCR assay detected N. ditissima in 51% of the inoculated apple tree samples. This was more sensitive than the culturing method detecting N. ditissima in 11% of the samples. An accumulation of N. ditissima DNA up to 34 mm distance from the inoculation site was observed without development of visible symptoms. To our knowledge this is the first time colonization of N. ditissima was detected and quantified in the absence of symptoms of European canker. The implications of this research are discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
European canker caused by Neonectria ditissima and apple scab caused by Venturia inaequalis, are the most important fungal diseases in apple growing regions in the world with maritime climates (Weber, 2014). In the Netherlands, apple is the second most valuable fruit crop after pear (CBS, 2021) with 6150 hectares of orchards and a production of 220 million kilos of fruit in 2020. In addition, Dutch nurseries produce approximately 6.5 million apple trees annually, mostly through clonal propagation (G. Mellema, Naktuinbouw, Netherlands, 25 May 2021, pers. comm.). European canker has been reported in Dutch orchards since the 18th century (Scheer, 1980) and it is still a major concern. The main measures to control European canker to date are (1) pruning out infected branches when weather conditions are less conducive to infection, (2) fungicide applications after pruning, leaf fall and picking, and (3) painting of wounds (Weber, 2014; Weber & Børve, 2021). Environmental thresholds contributing to infection have been used as a warning system (Latorre et al., 2002) and to estimate regional risk for European canker development (Beresford & Kim, 2010; Latorre et al., 2002), but control measures are still not sufficiently effective and more information is needed to refine the management strategies.
Infection occurs through various wound types including leaf scars, pruning, and picking wounds. It varies by region which wound type contributes most to the numbers of infections in the field (Amponsah et al., 2015; Crowdy, 1952; Dubin & English, 1974; Swinburne, 1975; Xu & Ridout, 1998). Timely protection of the trees is challenged by stagnation of symptom expression for a long period of time when conditions are suboptimal (Amponsah et al., 2015; McCracken et al., 2003; Walter et al., 2016; Xu & Ridout, 1998). Moreover, infections in the propagation phase could stay symptomless and only become visible up to three years later (McCracken et al., 2003).
The interactions of N. ditissima with the host before symptom expression are unclear, limiting our knowledge about the disease. Systemic spread (colonization inside the tree under the bark) of N. ditissima in the wood causing new cankers at different spots of the tree has been proposed (Crowdy, 1949; Weber, 2014) and may be an explanation for certain observations on spatial disease patterns such as the positive correlation in canker development between neighboring leaf scars in the same shoot (Xu et al., 2021). N. ditissima in symptomatic plant material, such as necrotic canker lesions, dead and stunted buds and fruit rot, can be detected through several methods including microbiological culturing methods (Amponsah et al., 2014; Amponsah et al., 2015; McCracken et al., 2003; Olivieri et al., 2021;Wesche & Weber, 2022), immunolocalization (Dewey et al., 1995) and molecular tools such as PCR and qPCR (Ghasemkhani et al., 2016; Langrell, 2002; Langrell & Barbara, 2001). Asymptomatic presence of N. ditissima in wood tissue after inoculations has been confirmed using culturing (Olivieri et al., 2021; Walter et al., 2016; Xu et al., 2021) where detection was achieved at a maximum depth of 5–10 mm from the inoculation site after 2 weeks (Olivieri et al., 2021). Using qPCR, Olivieri et al. (2021) confirmed asymptomatic colonization in wood up to 15 mm from a symptomatic inoculation site at 16 and 21 weeks after inoculation of pruning wounds and leaf scars, respectively. Recently, however a novel TaqMan PCR assay with higher sensitivity, developed by Elena et al. (2022), quantified N. ditissima after incubation of inoculated pruning wounds after 4 weeks, demonstrating the potential to not only detect, but also quantify N. ditissima during the colonization process at an earlier stage than previously reported. The objective of this research is therefore to use the TaqMan PCR assay (Elena et al., 2022) to detect and quantify N. ditissima during the asymptomatic colonization of apple wood over time. Outcomes of this research will contribute to a better understanding of the colonization process of N. ditissima in apple wood and may aid in the development of improved management strategies to reduce European canker.
Materials and methods
Plant material
The progress of colonization of N. ditissima through time was studied in the apple cultivars Elstar (moderately resistant) and Gala (susceptible). A total of 40 two-year-old potted trees of each cultivar asymptomatic to European canker were used, i.e. 20 trees inoculated with N. ditissima and 20 control trees inoculated with sterile water. The trees were placed in 2 blocks (N. ditissima/water) with 20 trees per cultivar and 3 rows (± 10 m) in between the blocks at the Wageningen University and Research Station in Randwijk (GPS coordinates 51.937706, 5.704645). The site had a concrete floor at a location distant from potential splash-dispersed conidia. Trees were potted in fertilized substrates (Multicote HK 8 mnd N14 + P8 + K20, Legro) and watered through a drip irrigation system. One application of Pirimor (pirimicarb) was applied to control aphids.
Inoculum preparation and inoculations
Inoculum was prepared from three single-spored isolates of N. ditissima, WURR18, 21 and 23, obtained from symptomatic ‘Jonagold’ branches in Randwijk, the Netherlands, in 2017 (WURR18) and 2018 (WURR 21 and 23). The isolates were thawed from storage in -80 °C and incubated on potato dextrose agar (PDA) for 5–7 days at 20 °C in the dark. To stimulate sporulation the isolates were transferred to modified Matsushima medium (Dubin & English, 1974; Matsushima, 1961) and incubated for three weeks with 16 hours light and eight hours darkness per day (Scheper et al., 2014). Spore suspensions were prepared by flooding the culture dish with sterile distilled water and disrupting the spores using a sterile scalpel. The suspension was poured through one layer of cheesecloth to remove mycelial fragments. The concentration of macro- and microconidia was determined using a haemocytometer and adjusted to a concentration of 104 conidia/ml. The isolates produced mostly microconidia (98%) and few macroconidia (2%). The suspension was prepared using a mixture of equal conidia ratios of the three isolates. To ascertain a good spread of the conidia on the inoculated surface 0.01% of surfactant Tween20 (Sigma-Aldrich, Darmstadt, Germany) was added. Inoculations were performed through pruning wounds on one-year old branches (growth of the previous year). The inoculations were performed outside, at dry weather with temperatures of 20–25 °C on August 8th 2018. Five randomly selected branches per tree (diameter ± 5–10 mm) were used, with 20 replicate trees per treatment (N. ditissima or sterile water). At the time for inoculation the branches were cut to 15 cm from the trunk using secateurs. To determine potential pre-existing N. ditissima in/on the trees, a 5 cm piece of the cut-off end of one of the five branches per tree was cut < 0.5 hour pre-inoculation and processed to use for TaqMan PCR assay. The fresh pruning wounds (on the branch still attached to the tree) were inoculated within ± 15 seconds by placing a 10 μl droplet (approximately 100 spores) of the conidial suspension (or sterile water) onto the surface of the cut wounds using a pipette. The droplet was sucked down by the wood within several minutes. In order to determine viability of the inoculum 100 μl of the conidial suspension was transferred onto water agar in duplicate and incubated overnight at ambient conditions. The percentage of germinated conidia was determined by assessing a total of 50 conidia with a stereo microscope after 20 hours.
Sampling
In order to determine the N. ditissima colonization process over time, the inoculated branches were sampled at four different time points, with one branch per tree per time point: after 3 hours (8 August), 2 weeks (23 August), 4 weeks (7 September) and 8 weeks (4 October). At each time, a piece of 5 cm was cut off the branch and placed in a 50 ml Falcon collection tube. Samples were kept cool in a closed box with ice and processed within three hours. At each time point, four discs (2-mm thick) were cut from the 5 cm cut-off piece using sterile blades: two discs between 2 and 6 mm distance from the inoculation site and two discs between 10 and 14 mm. Additionally, for sampling times at 4 and 8 weeks, two discs at 30 to 34 mm were taken (Tables 1 and 2). The fifth inoculated branch on each tree was not cut off. These were used as positive controls and assessed for symptom expression at each sampling time. The two discs per distance were used for N. ditissima detection using one disc for the TaqMan PCR assay and one for culturing. The discs for the TaqMan PCR assay were transferred to cryovial tubes, submerged in liquid nitrogen for 30 seconds and saved at -80 °C until further processing for DNA extraction. The discs used for culturing were transferred to cryovial tubes and placed in the fridge (4 °C). Weather conditions at the sampling times were: dry with 20–25 °C at the day of inoculation and at sampling after 2 weeks, rainy and 17 °C at sampling after 4 weeks and dry with 17 °C at sampling after 8 weeks.
DNA extraction and TaqMan PCR assay of N. ditissima from wood discs
Prior to DNA extraction, the discs were lyophilized and homogenized by beadbeating (6.35 mm RVS bead) with the Precellys (Bertin Technologies, Montigny-le-Bretonneux, France) for 2 × 15 seconds at 6000 rpm with a 5 seconds break. DNA was extracted with the MagAttract PowerSoil DNA extraction kit (Qiagen, Hilden, Germany) according to the protocol of the manufacturer. TaqMan PCR assays were performed using a 384-well format in a CFX384 Real-Time PCR Detection System (Bio-Rad Laboratories Inc., Hercules, CA, USA). For each TaqMan PCR assay 1 μl sample was mixed with 9 μl reaction mix containing 5 μl 2X PerfeCTa qPCR Toughmix (Quantabio, Beverly, MA, USA); 200 nM probe and 300 nM of each forward and reverse primer (Elena et al., 2022). The reaction conditions were: 95 °C for 2 min; 40 cycles of 95 °C for 15 seconds followed by 60 °C for 60 seconds. Data analysis was done by automatic threshold calculation within the Biorad CFX Manager software version 1.0 (Bio-Rad Laboratories Inc.). For each 384-well plate used, a 10-fold serial dilution ranging from 1 ng to 1 fg genomic DNA of N. ditissima isolate CBS 835.97 was run in parallel as a reference. If the 1 ng standard had a Ct shift of > 0.5 a fresh dilution series was made. A negative template control where 1 ul of sample is replaced by water was used in every 384-well plate. A separate PCR reaction, with a green fluorescent protein serving as an amplification control (AC) (Klerks et al., 2004), was performed for each sample to test possible inhibition. If AC amplification indicated inhibition of TaqMan-PCRs, measurements were repeated with 2- and 10-fold dilutions of the sample.
Isolation of N. ditissima from wood discs by culturing
For isolating of N. ditissima from wood, the discs were cut in eight pieces of approximately 2 × 2 mm. The pieces were placed on PDA plates amended with rifamycine and iprodione, both at 20 ppm (McCracken et al., 2003). Plates were kept at 20 °C in the dark and were assessed for fungal growth after one and three weeks when white mycelia were transferred to modified Matsushima-medium to induce spore production and incubated at ambient conditions and light for three weeks. In order to confirm identity of N. ditissima, cultures were rinsed with sterile water, conidia were dislodged, scraped off the media and poured through cheesecloth. N. ditissima conidia were identified in the residual suspensions using a stereomicroscope.
Data analysis
All analyses were performed using Genstat (VSN International, 2022). For the culturing method, detection was achieved when typical N. ditissima spores were identified. For the TaqMan PCR assay detection was achieved when discs contained amounts of target DNA above the limit of quantification (LOQ). The difference between detection probabilities for ‘Elstar’ and ‘Gala’ were tested employing Fisher’s exact test per time point and distance. This was done separately for the culturing method and the TaqMan PCR assay and only using trees inoculated with N. ditissima. The difference between detection probabilities for culturing and TaqMan PCR assay were also tested by means of Fisher’s exact test per cultivar, time point, and distance.
In order to represent the quantified target DNA concentration by the TaqMan PCR assay, the lyophilized weight of the discs was used to normalize the data. Ct values were converted to quantitative amounts of N. ditissima DNA using the standard curve and were log10 (target DNA) transformed for analyses and visualization. For each of the three conducted TaqMan PCR assay runs, a calibration curve consisting of a dilution series of isolate CBS 835.97 N. ditissima genomic DNA was prepared in nuclease free water in steps of 10–1 (1 ng to 1 fg DNA). There was a linear relationship between the amount of N. ditissima DNA and the corresponding Ct values with R2 ranging between 0.995 and 0.999 and slopes ranging between -3.256 and -3.304. The detection limit obtained was 1 fg of genomic DNA. To test whether the concentration of target DNA increases over time for discs at 2–6 mm of inoculated trees, a mixed model was fitted on the log10 scale with a random tree effect and fixed effects cultivar and time after inoculation and their interaction. In this analysis only discs with detected target DNA were used. The target DNA concentrations of discs at 10–14 and 30–34 mm were not statistically analyzed because of the low number of discs with detected target DNA.
Results
Detection of N. ditissima in wood discs by culturing
N. ditissima was detected by culturing in 2 of the discs from the control trees (N = 400, inoculated with sterile water) and from 42 of the discs inoculated with N. ditissima (N = 400) (Table 1). Of the discs inoculated with N. ditissima, 24 were obtained from ‘Elstar’ and 18 from ‘Gala’ (N = 200). The distance with the highest numbers of detection was at 2–6 mm with 35 discs, compared to 3 and 4 at 10–14 mm and 30–34 mm, respectively. Comparing the detection probability for the inoculated trees between the cultivars per distance and timepoint only revealed a significant difference at 8 weeks and 2–6 mm distance (P = 0.041) (Table 1).
Detection of N. ditissima in wood discs by TaqMan PCR assay
DNA of N. ditissima was detected in 48 of the discs from the control trees (N = 400)) of which 11 were recovered from ‘Elstar’ and 37 from ‘Gala’ (N = 200, Table 2). N. ditissima was detected by TaqMan PCR assay in 207 of the discs from trees inoculated with N. ditissima, in 122 ‘Gala’ discs and 85 ‘Elstar’ discs. For the pre-inoculation samples, N. ditissima was detected in 5 of the 80 discs, these were all discs of the cultivar Gala. For three distance x timepoint combinations, the detection of N. ditissima in inoculated ‘Gala’ trees was significantly greater than in ‘Elstar’ (Table 2). Overall, the number of discs with detected N. ditissima of the inoculated trees increased with time after inoculation and decreased with the distance from the inoculation point. In all discs from the inoculated trees taken at 2–6 mm after 2, 4 and 8 weeks, except two, N. ditissima was detected by TaqMan PCR assay (Table 2). Detection in 10–14 mm discs of inoculated trees was much smaller compared to 2–6 mm, but increased over time: 2 discs after 3 hours, 5 after 4 weeks, 15 after 4 weeks and 21 after 8 weeks with a significant higher number of discs for ‘Gala’ than ‘Elstar’ at 2 weeks and 8 weeks. Detection in 30–34 mm discs of inoculated trees was 7 after 4 weeks and 16 after 8 weeks with more discs for ‘Gala’ than ‘Elstar’ after 8 weeks.
Comparison of detection by culturing and TaqMan PCR assay
Of the 42 discs in which N. ditissima was detected by culturing, 35 were also detected by the TaqMan PCR assay in the same branch. Of the seven samples negative for N. ditissima four were sampled at 30–34 mm 8 weeks after inoculation, two from ‘Gala’ and two from ‘Elstar’. Two were sampled after 3 hours, one from each cultivar and one was obtained at 4 weeks from ‘Gala’ at 30–34 mm. The Ct values for these samples were between 37.6 and 40. Per cultivar, time point and distance, a Fisher’s exact test was employed to test whether the probability that detection by culturing was equal to that of the TaqMan PCR assay. When significant, the chance of detection using the TaqMan PCR assay was larger than by culturing, which was the case for 12 of the 20 different cultivar x distance x timepoint combinations (Table 3).
Quantity of target DNA detected using the TaqMan PCR assay
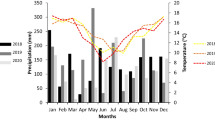
The mixed model analysis revealed that the interaction between cultivar and time was not significant (P = 0.066), that there is no indication of a cultivar effect (P = 0.255) and a very significant time effect (P < 0.001). Pairwise comparisons of the means for the different time points revealed that all means were significantly different (P < 0.001) except after 4 weeks and 8 weeks (P = 0.073). In the discs taken at 2–6 mm of the inoculated trees, there was an increase over time of the target DNA concentration starting with an average of 2 fg/mg and 8 fg/mg after 3 hours up to 54,303 and 43,909 fg/mg after 8 weeks for ‘Elstar’ and ‘Gala’, respectively (Fig. 1). At 10–14 mm a different pattern was observed. Very low levels (< 0,5 fg/mg) of N. ditissima were measured at 3 hours and 2 weeks. After 4 weeks, N. ditissima was detected with an average of 33 fg/mg and 4 fg/mg in ‘Elstar’ and ‘Gala’, respectively. This decreased in ‘Elstar’ to an average of 1 fg/mg, but measured a similar amount in ‘Gala’ after 8 weeks. At 30–34 mm the average concentration of target DNA was 0.2 fg/mg in ‘Elstar’ both after 4 and 8 weeks whereas in ‘Gala’ the concentration increased from 0.3 fg/mg after 4 weeks to 2.5 fg/mg after 8 weeks.
The concentrations of Neonectria ditissima (Nd) DNA in wooden discs of two apple cultivars, Elstar and Gala, of trees inoculated with N. ditissima (Nd inoculated) or sterile water (Control). DNA concentrations were calculated in discs cut from the tree branches at three distances from the inoculation point, 2–6 mm, 10–14 mm and 30–34 mm, at four time points, 3 hours, 2 weeks, 4 weeks and 8 weeks, after inoculation. The percentages of discs where no DNA was detected are presented at the x-axis (%Non)
The target DNA concentrations in control discs (inoculated with sterile water) were overall much lower than for discs inoculated with N. ditissima with some exceptions (Fig. 1). These included two discs from ‘Elstar’, one at 2–6 mm at 2 weeks and one at 30–34 mm at 8 weeks. Ten ‘Gala’ discs showed values higher than the N. ditissima inoculated samples, one at 10–14 mm at 2 weeks and four at 8 weeks, one at 30–34 mm at 4 weeks and four at 8 weeks.
Quantifying the N. ditissima DNA in the discs using the TaqMan PCR assay allowed following the colonization process over time through the branches of 20 trees in both cultivars (Supplemental Fig. 1A and B). In all branches the quantity of N. ditissima declined with a longer distance from the inoculation point. The concentration of target DNA was greater with 1.03 fg/mg in 10–14 mm discs compared to 0.57 fg/mg in 2–6 mm discs once. In 7 trees, no target DNA was detected at 10–14 mm distance from the inoculation point, but it was detected at 30–34 mm distance. These were ‘Gala’ tree 5, 9, 12 and 16 at 4 weeks, ‘Elstar’ tree 8 at 4 weeks and ‘Elstar’ tree 1 and 20 at 8 weeks. In five control discs (N = 80) sampled before inoculation in ‘Gala’ a pre-existing level of N. ditissima was measured of 6.84, 123.09, 1.19, 0.56 and 1.33 fg/mg.
Symptom development (formation canker lesions) was evaluated on all experimental branches. None of the branches used in the experiment for inoculations and sampling with either N. ditssima or water showed visible symptoms. On the branches inoculated as a positive control, no symptoms were visible after 2 and 4 weeks. Two out of the 20 branches of ‘Gala’ and three of the 20 branches of ‘Elstar’ demonstrated lesions of circa 1 cm length from the pruning wound after 8 weeks.
Discussion
In this study the accumulation of N. ditissima in apple wood was measured up to 34 mm over 8 weeks in apple wood after inoculating pruning wounds, while no symptoms had developed in the sampled branches during the experiment. This is the first time colonization of N. ditissima was detected and quantified in the absence of symptoms of European canker. These results confirm that the higher sensitivity of the used TaqMan PCR assay compared to previously reported assays allowed for earlier and deeper detection of the pathogen in wood. Olivieri et al. (2021) used the qPCR developed by Ghasemkhani et al. (2016) and detected N. ditissima at 10–15 mm from a canker lesion of leaf scars and pruning wounds after 4 and 7 months of inoculation indicating the need for more time required for accumulation of N. ditissima to be detected. The ability to detect N. ditissima at lower quantities than previously reported may reopen discussions on several attributes in the epidemiology of this disease e.g. the concept of systemic infection and should be reinvestigated.
Two types of media have been reported for consistent recovery of N. ditissima from symptomless woody tissues, a semi-selective PDA and apple sap amended water agar (ASAWA). The semi-selective PDA was reported to recover N. ditissima from asymptomatic tissue at 5-10 mm distance from inoculated pruning wounds after 2 weeks at 100% and therefore very suitable to this study (Olivieri et al., 2021). ASAWA recovered N. ditissima from asymptomatic inoculated wounds up to 100% after 8 weeks (Amponsah et al, 2014; Scheper et al., 2019a, b; Walter et al., 2016; Xu et al., 2021). Using the semi-selective PDA, we detected N. ditissima in 25 out of 120 samples taken at 2–6 mm over 8 weeks. Microconidia are less active than macroconidia (Scheper et al., 2015; Wesche & Weber, 2022), therefore the lower numbers of detection in our study could have been caused by the used inoculum with 100 microconidia per wound versus the 10000 macroconidia per wound used by Olivieri et al. (2021). Using inoculum of isolates with a higher number of macroconidia would be closer to field inoculum (Scheper et al., 2010) and could have resulted in a higher detection rate in culturing as well as by the TaqMan PCR assay.
Seven samples were negative using the TaqMan PCR assay but positive by culturing. A direct comparison could not be made because adjacent discs were used for TaqMan PCR assay and culturing. In addition, not all microconidia of different fungal species can be distinguished on Matsushima-medium from N. ditissima (Scheper et al., 2014) potentially causing false positives. Future research should include the DNA sequencing of these isolates to confirm their identity. Using the TaqMan PCR assay, detection of N. ditissima occurred at very low levels around the LOQ. In 9% of the cases, no detection occurred at 10–14 mm but N. ditissima was detected at 30–34 mm. In these cases, the corresponding Ct values for these measurements were between 37.2 and 40. These values are close to the set LOQ of 37. Depending on the objective of an experiment and the setting of the LOQ these measurements could be classified as false negatives.
The TaqMan PCR assay detected N. ditissima in a high number of discs of the water-inoculated trees, suggesting that the pathogen was already present inside the trees. These plants were 2 years old. Even though they looked disease-free at the start and end of the experiment, the presence of existing natural infection cannot be excluded. Interestingly, pre-existence of N. ditissima detected by the TaqMan PCR assay used in this research was also reported by Elena et al. (2022), who decided to use ‘Elstar’ in their experiments based on a lower background level compared to other cultivars. The higher concentration of N. ditissima in discs 10–34 mm from the site of inoculation in water-inoculated trees compared to N. ditissima-inoculated trees may be due to variations in virulence between our inoculum and field inoculum (Scheper et al., 2010).
The positive control inoculation demonstrated low numbers of symptom development. After 8 weeks, 2 and 3 of the ‘Gala’ and ‘Elstar’ branches (N = 20 per cultivar), respectively, showed symptoms. The inoculum used in this study consisted of microconidia which may be an explanation for slow disease progression. Scheper et al. (2015) showed that an inoculation with an isolate producing 1% macroconidia started to develop symptoms after 7 weeks compared to < 5 weeks for isolates with over 60% macroconidia and that the isolate was significantly less aggressive. These and our observations are also in line with Wesche and Weber’s (2022) findings of a slower germination and elongation of microconidia compared to macroconidia and symptom development on fruit. Another explanation could be that suboptimal weather conditions may have slowed down the colonization. Fungal infections and disease development are generally favoured by temperatures between 5 °C and 20 °C and high humidity (Latorre et al., 2002; Wenneker et al., 2017). The day of inoculation was dry and had temperatures higher than that potentially drying out the tissues and slowing down infections. Our observations however are not uncommon in outdoor experiments, similar inoculation experiments of pruning wounds in the UK resulted in ~ 5% incidence after 8 weeks (30% after 12 weeks) (Olivieri et al., 2021), and 32% incidence after 16 weeks (Xu & Ridout, 1998).
The TaqMan PCR assay has been used successfully for the quantification of N. ditissima DNA in apple branches treated with various candidate antagonists in assays for screening new biological control agents (Elena et al., 2022). Other applications of this assay could include comparative studies on the invasive behavior of the fungus on susceptible versus partially resistant cultivars. N. ditissima was detected by TaqMan PCR assay in more ‘Gala’ discs compared to ‘Elstar’, but it is unclear if this could have been caused by varietal differences in wood colonization. Furthermore, the assay could be further incorporated into multiplex assays for the detection of multiple pathogens or other target sequences (Luo et al., 2017) or can assist in the refinement of pruning time and distances. In these experiments care should be taken that molecular methods detect all the target DNAof a pathogen, living and dead, in host material. An accumulation of the target DNA over time should therefore be included in the experiment. Børve et al. (2018) demonstrated that some disease development starts through asymptomatic infected scion wood. Early detection and diagnosis of N. ditissima in nursery plant material could help prevent disease development and spread at a later, stage of apple tree development (Wenneker et al., 2017). To use a TaqMan PCR assay for pre-screening of this planting material, a thorough evaluation of the thresholds defining the biological relevance of the measured N. ditissima DNA quantity for disease development is needed as well as a strategic sampling and a possible pooling approach for the large amounts of plant material produced in the nurseries.
Based on our current findings pruning practices should focus on cutting the whole branch rather than a part of the branch as was previously concluded (Olivieri et al., 2021; Xu et al., 2021). This study demonstrated that the TaqMan PCR assay could detect N. ditissima at very low and unlikely amounts in the host (Wesche & Weber, 2022) and proved therefore potential to further assess the internal colonization over time and further refine pruning practices. Future studies using isolates producing high numbers of macroconidia and assessing wood at further distances from an inoculation site over a longer period of time are suggested to further establish our understanding of the colonization pattern of N. ditissima representative for the infections in nature.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Amponsah, N. T., Walter, M., & Scheper, R. W. A. (2014). Agar media for isolation of Neonectria ditissima from symptomatic and asymptomatic apple tissues and production of infective conidia. New Zealand Plant Protection, 67, 116–122. https://doi.org/10.30843/nzpp.2014.67.5741
Amponsah, N. T., Walter, M., Beresford, R. M., & Scheper, R. W. A. (2015). Seasonal wound presence and susceptibility to Neonectria ditissima infection in New Zealand apple trees. New Zealand Plant Protection, 68, 250–256.
Beresford, R. M., & Kim, K. S. (2010). Identification of regional climatic conditions favorable for development of European canker of apple. Phytopathology, 101, 135–146.
Børve, J., Kolltveit, S. A., Talgø, V., & Stensvand, A. (2018). Apple rootstocks may become infected by Neonectria ditissima during propagation. Acta Agriculturae Scandinavica, Section B — Soil & Plant Science, 68(1), 16–25. https://doi.org/10.1080/09064710.2017.1351578
CBS. (2021). Centraal Bureau voor Statistiek. Fruitteelt; oogst en teeltoppervlakte appels en peren. Retrieved May 12, 2021 from https://www.cbs.nl/nl-nl/cijfers/detail/84499NED
Crowdy, S. H. (1949). Observations on apple canker III. The anatomy of the stem canker. Annals of Applied Biology, 36(4), 483–495. https://doi.org/10.1111/j.1744-7348.1949.tb06944.x
Crowdy, S. H. (1952). Observations on apple canker. Annals of Applied Biology, 39(4), 569–580. https://doi.org/10.1111/j.1744-7348.1952.tb01071.x
Dewey, F. M., Li, R., & Swinburne, T. R. (1995). A monoclonal antibody immunoassay for the detection of Nectria galligena in apple fruit and woody tissues. Bulletin OEPP/EPPO Bulletin, 25, 65–73.
Dubin, H. J., & English, H. (1974). Factors affecting apple leaf scar infection by Nectria galligena conidia. Phytopathology, 64, 1201–1203.
Elena, G., Groenenboom-de Haas, B. H., Houwers, I., de Lange, E., Schnabel, S. K., & Köhl, J. (2022). Systematic stepwise screening of new microbial antagonists for biological control of European canker. Biological Control, 105009. https://doi.org/10.1016/j.biocontrol.2022.105009
Ghasemkhani, M., Holefors, A., Marttila, S., Dalman, K., Zborowska, A., Rur, M., Rees-George, J., Nybom, H., Everett, K. R., Scheper, R. W. A., & Garkava-Gustavsson, L. (2016). Real-time PCR for detection and quantification, and histological characterization of Neonectria ditissima in apple trees. Trees, 30, 1111–1125. https://doi.org/10.1007/s00468-015-1350-9
Klerks, M. M., Zijlstra, C., & van Bruggen, A. H. C. (2004). Comparison of real-time PCR methods for detection of Salmonella enterica and Escherichia coli O157:H7, and introduction of a general internal amplification control. Journal of Microbiological Methods, 59(3), 337–349. https://doi.org/10.1016/j.mimet.2004.07.011
Langrell, S. R. H. (2002). Molecular detection of Neonectria galligena (syn. Nectria galligena). Mycological Research, 106(3), 280–292. https://doi.org/10.1017/S095375620200552X
Langrell, S. R. H., & Barbara, D. J. (2001). Magnetic capture hybridisation for improved PCR detection of Nectria galligena from lignified apple extracts. Plant Molecular Biology Reporter, 19(1), 5–11. https://doi.org/10.1007/BF02824073
Latorre, B. A., Rioja, M. E., Lillo, C., & Muñoz, M. (2002). The effect of temperature and wetness duration on infection and a warning system for European canker (Nectria galligena) of apple in Chile. Crop Protection, 21(4), 285–291. https://doi.org/10.1016/S0261-2194(01)00099-0
Luo, Y., Gu, S., Felts, D., Puckett, R. D., Morgan, D. P., & Michailides, T. J. (2017). Development of qPCR systems to quantify shoot infections by canker-causing pathogens in stone fruits and nut crops. Journal of Applied Microbiology, 122(2), 416–428. https://doi.org/10.1111/jam.13350
Matsushima, T. (1961). A new medium for identification of hyphomycetes. Transactions of the Mycological Society of Japan, 2, 118–120.
McCracken, A. R., Berrie, A., Barbara, D. J., Locke, T., Cooke, L. R., Phelps, K., Swinburne, T. R., Brown, A. E., Ellerker, B., & Langrell, S. R. H. (2003). Relative significance of nursery infections and orchard inoculum in the development and spread of apple canker (Nectria galligena) in young orchards. Plant Pathology, 52(5), 553–566. https://doi.org/10.1046/j.1365-3059.2003.00924.x
Olivieri, L., Saville, R. J., Gange, A. C., & Xu, X. M. (2021). Limited asymptomatic colonization of apple tree shoots by Neonectria ditissima following infection of leaf scars and pruning wounds. Plant Pathology, 70(8), 1838–1849. https://doi.org/10.1111/ppa.13419
Scheer, H. V. D. (1980). Kanker bij vruchtbomen. Proefstation voor de fruitteelt Wilhelminadorp.
Scheper, R. W. A., Fisher, B. M., & Wood, P. N. (2010). Pathogenicity of field and laboratory grown inoculum of Neonectria galligena on potted apple trees. New Zealand Plant Protection, 63(abstract only), 280.
Scheper, R. W. A., Fisher, B. M., Amponsah, N. T., & Walter, M. (2014). Effect of culture medium light and air circulation on sporulation of Neonectria ditissima. New Zealand Plant Protection, 67, 123–132.
Scheper, R. W. A., Frijters, L., Fisher, B. M., & Hedderley, D. I. (2015). Effect of freezing of Neonectria ditissima inoculum on its pathogenicity. New Zealand Plant Protection, 68, 257–263.
Scheper, R. W. A., Fisher, B. M., Bowen, J. K., Amponsah, N. T., & Hedderley, D. I. (2019a). Successive passaging through an apple host of six low-virulent Neonectria ditissima isolates increased virulence in one of them. New Zealand Plant Protection, 72, 103–116. https://doi.org/10.30843/nzpp.2019.72.300
Scheper, R. W. A., Vorster, L., Turner, L., Campbell, R. E., Colhoun, K., McArley, D., Murti, R., Hodson, A., Beresford, R. M., Stock, M., Fisher, B. M., Hedderley, D. I., & Walter, M. (2019b). Lesion development and conidial production of Neonectria ditissima on apple trees in four New Zealand regions. New Zealand Plant Protection, 72, 123–134. https://doi.org/10.30843/nzpp.2019.72.302
Swinburne, T. R. (1975). European canker of apple (Nectria galligena). Review of Plant Pathology, 54, 787–799.
VSN International. (2022). Genstat for Windows, 22nd ed. Hemel Hempstead: VSN International. http://www.genstat.co.uk
Walter, M., Roy, S., Fisher, B. M., Mackle, L., Amponsah, T., Curnow, T., Campbell, R. E., Braun, P., Reineke, A., & Scheper, R. W. A. (2016). How many conidia are required for wound infection of apple plants by Neonectria ditissima? New Zealand Plant Protection, 69, 238–245.
Weber, R. W. S. (2014). Biology and control of the apple canker fungus Neonectria ditissima (syn. N. galligena) from a Northwestern European perspective. Erwerbs-Obstbau, 56(3), 95–107. https://doi.org/10.1007/s10341-014-0210-x
Weber, R. W. S., & Børve, J. (2021). Infection biology as the basis of integrated control of apple canker (Neonectria ditissima) in Northern Europe. CABI Agriculture and Bioscience, 2(1), 5. https://doi.org/10.1186/s43170-021-00024-z
Wenneker, M., de Jong, P. F., Joosten, N. N., Goedhart, P. W., & Thomma, B. P. H. J. (2017). Development of a method for detection of latent European fruit tree canker (Neonectria ditissima) infections in apple and pear nurseries. European Journal of Plant Pathology, 148(3), 631–635. https://doi.org/10.1007/s10658-016-1115-3
Wesche, J., & Weber, R. W. S. (2022). Are microconidia infectious principles in Neonectria ditissima? Journal of Plant Diseases and Protection. https://doi.org/10.1007/s41348-022-00669-6
Xu, X. M., & Ridout, M. S. (1998). The effects of inoculum dose, duration of wet period, temperature and wound age on infection by Nectria galligena of pruning wounds on apple. European Journal of Plant Pathology, 104(5), 511–519. https://doi.org/10.1023/a:1008689406350
Xu, X. M., Olivieri, L., Gange, A. C., Vorster, L., Rice, D., Campbell, R. E., & Walter, M. (2021). Does apple canker develop independently on leaf scars of a single apple shoot? New Zealand Plant Protection, 74(2S), S9–S19. https://doi.org/10.30843/nzpp.2021.74.11739
Acknowledgements
The authors would like to thank Engelien Kerkhof, Renske Bos and Yvonne Griekspoor for their technical assistance, Lia Groenenboom-de Haas for her contributions to the molecular analyses and Dr Leone Olivieri (NIAB-EMR) for advice on culturing methods.
Funding
This study was funded by the Dutch Ministry of Agriculture, Nature and Food Quality and a consortium consisting of 10 companies lead by the Dutch Fruit Growers Organisation (NFO), and was part of the project KV 1605–033 ‘PPS Integrale ketenaanpak vruchtboomkanker’.
Author information
Authors and Affiliations
Contributions
Conceptualization: D.O.C. Harteveld, P.F. de Jong, J. Köhl, M. Wenneker; Methodology: I. Houwers, D.O.C. Harteveld; Data analysis: P. Goedhart, D.O.C. Harteveld; Writing—original draft preparation: D.O.C. Harteveld; Writing—review and editing: I. Houwers, M. Wenneker, J. Köhl; Resources: M. Wenneker, J. Köhl: Funding acquisition: P.F. de Jong, M. Wenneker; Supervision: M. Wenneker, J. Köhl.
Corresponding author
Ethics declarations
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
Conflict of interest
There is no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
10658_2023_2662_MOESM1_ESM.pdf
Supplementary file1 (PDF 621 KB) Supplemental figure 1 Quantity of N. ditissima DNA (Nd DNA) in fg per mg lyophilized wood of discs cut at three distances from the inoculated pruning wound of 20 apple trees of cultivars Elstar (A) and Gala (B) at four timepoints. Timepoints were 3 hours, 2 weeks, 4 weeks and 4 weeks. At 3 hours and 2 weeks discs were cut at 2-6 and 10-14mm from the inoculation point, at 4 and 8 weeks discs were also cut at 30-34mm. The LOQ is the limit of quantification
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Harteveld, D.O.C., Goedhart, P.W., Houwers, I. et al. Detecting the asymptomatic colonization of apple branches by Neonectria ditissima, causing European canker of apple. Eur J Plant Pathol 166, 291–301 (2023). https://doi.org/10.1007/s10658-023-02662-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-023-02662-7