Abstract
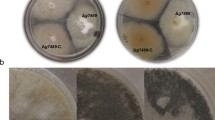
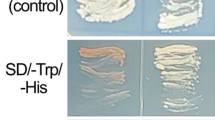
The protein kinase HOG1 in high osmolarity glycerol pathway is one of mitogen-activated protein kinases and plays an important role in fungi cellular responses to external stress and stimuli. In this study, AlHOG1, a yeast HOG1 homolog, was isolated from tobacco pathogenic fungus Alternaria longipes and its disrupted mutants were generated using homologous recombination. Compared with the wild-type strain, the mutants grew slower under Minimal-Media, produced less conidia on Potato-Dextrose-Agar and showed a retardation phenotype in conidial germination rate. Colony color observation indicated that AlHOG1 most likely negatively regulated melanin production under nutrients-deficient or osmotic stress. The same action mode of AlHOG1 on hyphal growth in response to certain carbon or nitrogen sources was speculated. Additionally, the disrupted strains were sensitive to osmotic and oxidative stress, and showed increased resistance to dicarboximide, phenylpyrrole and carbendazim fungicides. Meanwhile, results from the comparative analysis between the AlHOG1- and the AlHK1 (encoding a group III histidine kinase)-disrupted mutants showed that AlHOG1 mediated-pathway was partly regulated by the upstream AlHK1 kinase for cellular resistance to salts osmotic stress and that AlHK1 was a primary regulator for cellular sugar and fungicides stress adaptation and likely modulated both AlHOG1-mediated pathway and other unknown mechanisms. Noteworthily, AlHOG1 but not AlHK1 was confirmed to be associated with high concentration glycerol adaptation, which is the first report to our knowledge. Overall, our results indicated that AlHOG1 was involved in multiple physiological processes in A. longipes, including mycelial and conidial development, melanin production, nutrients sensing and multi-stress responses.






Similar content being viewed by others
References
Adam, A. L., Kohut, G., & Hornok, L. (2008). Cloning and characterization of a HOG-type MAP kinase encoding gene from Fusarium proliferatum. Acta Phytopathologica et Entomologica Hungarica, 44(1), 1–13.
Brewster, J. L., & Gustin, M. C. (2014). Hog1: 20 years of discovery and impact. Science Signaling. doi:10.1126/scisignal.2005458
Christianson, T. W., Sikorski, R. S., Dante, M., Shero, J. H., & Hieter, P. (1992). Multifunctional yeast high-copy-number shuttle vectors. Gene, 110(1), 119–122.
Colot, H. V., Park, G., Turner, G. E., Ringelberg, C., Crew, C. M., Litvinkova, L., et al. (2006). A high-throughput gene knockout procedure for Neurospora reveals functions for multiple transcription factors. Proceedings of the National Academy of Sciences of the United States of America, 103(27), 10352–10357.
R Core Team. (2015). R: a language and environment for statistical computing. R foundation for statistical computing. http://www.R-project.org/.
Dixon, K. P., Xu, J. R., Smirnoff, N., & Talbot, N. J. (1999). Independent signaling pathways regulate cellular turgor during hyperosmotic stress and appressorium-mediated plant infection by Magnaporthe grisea. Plant Cell, 11(10), 2045–2058.
Du, C., Sarfati, J., Latge, J. P., & Calderone, R. (2006). The role of the sakA (Hog1) and tcsB (sln1) genes in the oxidant adaptation of Aspergillus fumigatus. Medical Mycology, 44(3), 211–218.
Eisenman, H. C., & Casadevall, A. (2012). Synthesis and assembly of fungal melanin. Applied Microbiology and Biotechnology, 93(3), 931–940.
Eliahu, N., Igbaria, A., Rose, M. S., Horwitz, B. A., & Lev, S. (2007). Melanin biosynthesis in the maize pathogen Cochliobolus heterostrophus depends on two mitogen-activated protein kinases, Chk1 and Mps1, and the transcription factor Cmr1. Eukaryotic Cell, 6(3), 421–429.
Esquivel-Naranjo, E. U., Garcia-Esquivel, M., Medina-Castellanos, E., Correa-Pérez, V. A., Parra-Arriaga, J. L., Landeros-Jaime, F., et al. (2016). A Trichoderma atroviride stress-activated MAPK pathway integrates stress and light signals. Molecular Microbiology. doi:10.1111/mmi.13355.
Hagiwara, D., Takahashi-Nakaguchi, A., Toyotome, T., Yoshimi, A., Abe, K., Kamei, K., et al. (2013). NikA/TcsC histidine kinase is involved in conidiation, hyphal morphology, and responses to osmotic stress and antifungal chemicals in Aspergillus fumigatus. PloS One, 8(12), e80881.
Igbaria, A., Lev, S., Rose, M. S., Lee, B. N., Hadar, R., Degani, O., et al. (2008). Distinct and combined roles of the MAP kinases of Cochliobolus heterostrophus in virulence and stress responses. Molecular Plant-Microbe Interactions, 21(6), 769–780.
Izumitsu, K., Yoshimi, A., & Tanaka, C. (2007). Two-component response regulators Ssk1p and Skn7p additively regulate high-osmolarity adaptation and fungicide sensitivity in Cochliobolus heterostrophus. Eukaryotic Cell, 6(2), 171–181.
Izumitsu, K., Yoshimi, A., Kubo, D., Morita, A., Saitoh, Y., & Tanaka, C. (2009). The MAPKK kinase ChSte11 regulates sexual/asexual development, melanization, pathogenicity, and adaptation to oxidative stress in Cochliobolus heterostrophus. Current Genetics, 55(4), 439–448.
James, T. Y., Kauff, F., Schoch, C. L., Matheny, P. B., Hofstetter, V., Cox, C. J., et al. (2006). Reconstructing the early evolution of fungi using a six-gene phylogeny. Nature, 443(7113), 818–822.
Joubert, A., Bataille-Simoneau, N., Campion, C., Guillemette, T., Hudhomme, P., Iacomi-Vasilescu, B., et al. (2011). Cell wall integrity and high osmolarity glycerol pathways are required for adaptation of Alternaria brassicicola to cell wall stress caused by brassicaceous indolic phytoalexins. Cellular Microbiology, 13(1), 62–80.
Kawasaki, L., Sanchez, O., Shiozaki, K., & Aguirre, J. (2002). SakA MAP kinase is involved in stress signal transduction, sexual development and spore viability in Aspergillus nidulans. Molecular Microbiology, 45(4), 1153–1163.
Kojima, K., Takano, Y., Yoshimi, A., Tanaka, C., Kikuchi, T., & Okuno, T. (2004). Fungicide activity through activation of a fungal signalling pathway. Molecular Microbiology, 53(6), 1785–1796.
Lamb, T. M., Finch, K. E., & Bell-Pedersen, D. (2012). The Neurospora crassa OS MAPK pathway-activated transcription factor ASL-1 contributes to circadian rhythms in pathway responsive clock-controlled genes. Fungal Genetics and Biology, 49(2), 180–188.
Lenassi, M., Vaupotic, T., Gunde-Cimerman, N., & Plemenitas, A. (2007). The MAP kinase HwHog1 from the halophilic black yeast Hortaea werneckii: coping with stresses in solar salterns. Saline Systems. doi:10.1186/1746-1448-3-3.
Leng, Y., & Zhong, S. (2015). The role of mitogen-activated protein (MAP) kinase signaling components in the fungal development, stress response and virulence of the fungal cereal pathogen Bipolaris sorokiniana. PloS One, 10(5), e0128291.
Lin, C. H., & Chung, K. R. (2010). Specialized and shared functions of the histidine kinase- and HOG1 MAP kinase-mediated signaling pathways in Alternaria alternata, a filamentous fungal pathogen of citrus. Fungal Genetics and Biology, 47(10), 818–827.
Liu, W., Leroux, P., & Fillinger, S. (2008). The HOG1-like MAP kinase Sak1 of Botrytis cinerea is negatively regulated by the upstream histidine kinase Bos1 and is not involved in dicarboximide- and phenylpyrrole-resistance. Fungal Genetics and Biology, 45(7), 1062–1074.
Liu, W., Soulie, M. C., Perrino, C., & Fillinger, S. (2011). The osmosensing signal transduction pathway from Botrytis cinerea regulates cell wall integrity and MAP kinase pathways control melanin biosynthesis with influence of light. Fungal Genetics and Biology, 48(4), 377–387.
Luo, Y., Yang, J., Zhu, M., Yan, J., Mo, M., & Zhang, K. Q. (2008). Characterization of mutations in AlHK1 gene from Alternaria longipes: implication of limited function of two-component histidine kinase on conferring dicarboximide resistance. Journal of Microbiology and Biotechnology, 18(1), 15–22.
Luo, Y. Y., Yang, J. K., Zhu, M. L., Liu, C. J., Li, H. Y., Lu, Z. B., et al. (2012). The group III two-component histidine kinase AlHK1 is involved in fungicides resistance, osmosensitivity, spore production and impacts negatively pathogenicity in Alternaria longipes. Current Microbiology, 64(5), 449–456.
Maayan, I., Beenstock, J., Marbach, I., Tabachnick, S., Livnah, O., & Engelberg, D. (2012). Osmostress induces autophosphorylation of Hog1 via a C-terminal regulatory region that is conserved in p38alpha. PloS One, 7(9), e44749.
Mao, C., Chen, P., Dai, Q., Yang, L., & Huang, J. (2014). Biological characteristics of an Hog1 MAPK homologous gene FoHog1 knock-out mutant of Fusarium oxysporum f. Sp. cubense. Acta Microbiologica Sinica, 54(11), 1267–1278.
Mehrabi, R., Zwiers, L. H., de Waard, M. A., & Kema, G. H. (2006). MgHog1 regulates dimorphism and pathogenicity in the fungal wheat pathogen Mycosphaerella graminicola. Molecular Plant-Microbe Interactions, 19(11), 1262–1269.
Moriwaki, A., Kubo, E., Arase, S., & Kihara, J. (2006). Disruption of SRM1, a mitogen-activated protein kinase gene, affects sensitivity to osmotic and ultraviolet stressors in the phytopathogenic fungus Bipolaris oryzae. FEMS Microbiology Letters, 257(2), 253–261.
Murakami, Y., Tatebayashi, K., & Saito, H. (2008). Two adjacent docking sites in the yeast Hog1 mitogen-activated protein (MAP) kinase differentially interact with the Pbs2 MAP kinase kinase and the Ptp2 protein tyrosine phosphatase. Molecular and Cellular Biology, 28(7), 2481–2494.
Nicholls, P., Sternin, Y., Loewen, J., Jennings, T., & Tattrie, B. (1996). Hydration as a control mechanism in membrane proteins: the case of cytochrome c oxidase. Faraday Discussions, 103, 313–323.
Nimmanee, P., Woo, P. C., Kummasook, A., & Vanittanakom, N. (2015). Characterization of sakA gene from pathogenic dimorphic fungus Penicillium marneffei. International Journal of Medical Microbiology, 305(1), 65–74.
Park, S. M., Choi, E. S., Kim, M. J., Cha, B. J., Yang, M. S., & Kim, D. H. (2004). Characterization of HOG1 homologue, CpMK1, from Cryphonectria parasitica and evidence for hypovirus-mediated perturbation of its phosphorylation in response to hypertonic stress. Molecular Microbiology, 51(5), 1267–1277.
Posas, F., Wurgler-Murphy, S. M., Maeda, T., Witten, E. A., Thai, T. C., & Saito, H. (1996). Yeast HOG1 MAP kinase cascade is regulated by a multistep phosphorelay mechanism in the SLN1-YPD1-SSK1 "two-component" osmosensor. Cell, 86(6), 865–875.
Segmuller, N., Ellendorf, U., Tudzynski, B., & Tudzynski, P. (2007). BcSAK1, a stress-activated mitogen-activated protein kinase, is involved in vegetative differentiation and pathogenicity in Botrytis cinerea. Eukaryotic Cell, 6(2), 211–221.
Staben, C., Jensen, B., & Singer, M. (1989). Use of a bacterial hygromycin B resistance gene as a dominant selectable marker in Neurospora crassa transformation. Fungal Genet Newsl, 36, 79–81.
Tamura, K., Stecher, G., Peterson, D., Filipski, A., & Kumar, S. (2013). MEGA6: molecular evolutionary genetics analysis version 6.0. Molecular Biology and Evolution, 30(12), 2725–2729.
Thompson, J. D., Gibson, T. J., Plewniak, F., Jeanmougin, F., & Higgins, D. G. (1997). The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Research, 25(24), 4876–4882.
Wang, M., Chen, C., Zhu, C., Sun, X., Ruan, R., & Li, H. (2014). Os2 MAP kinase-mediated osmostress tolerance in Penicillium digitatum is associated with its positive regulation on glycerol synthesis and negative regulation on ergosterol synthesis. Microbiological Research, 169(7–8), 511–521.
Winston, F., Dollard, C., & Ricupero-Hovasse, S. L. (1995). Construction of a set of convenient Saccharomyces cerevisiae strains that are isogenic to S288C. Yeast, 11(1), 53–55.
Xue, T., Nguyen, C. K., Romans, A., & May, G. S. (2004). A mitogen-activated protein kinase that senses nitrogen regulates conidial germination and growth in Aspergillus fumigatus. Eukaryotic Cell, 3(2), 557–560.
Yago, J. I., Lin, C. H., & Chung, K. R. (2011). The SLT2 mitogen-activated protein kinase-mediated signalling pathway governs conidiation, morphogenesis, fungal virulence and production of toxin and melanin in the tangerine pathotype of Alternaria alternata. Molecular Plant Pathology, 12(7), 653–665.
Yoshimi, A., Kojima, K., Takano, Y., & Tanaka, C. (2005). Group III histidine kinase is a positive regulator of Hog1-type mitogen-activated protein kinase in filamentous fungi. Eukaryotic Cell, 4(11), 1820–1828.
Zhang, D., Yang, Y., Castlebury, L. A., & Cerniglia, C. E. (1996). A method for the large scale isolation of high transformation efficiency fungal genomic DNA. FEMS Microbiology Letters, 145(2), 261–265.
Zhang, Y., Lamm, R., Pillonel, C., Lam, S., & Xu, J. R. (2002). Osmoregulation and fungicide resistance: the Neurospora crassa os-2 gene encodes a HOG1 mitogen-activated protein kinase homologue. Applied and Environmental Microbiology, 68(2), 532–538.
Zhang, L., Yang, J. K., Niu, Q. H., Zhao, X. N., Ye, F. P., Liang, L. M., et al. (2008). Investigation on the infection mechanism of the fungus Clonostachys rosea against nematodes using the green fluorescent protein. Applied Microbiology and Biotechnology, 78(6), 983–990.
Zhang, Y., Zhao, J., Fang, W., Zhang, J., Luo, Z., Zhang, M., et al. (2009). Mitogen-activated protein kinase hog1 in the entomopathogenic fungus Beauveria bassiana regulates environmental stress responses and virulence to insects. Applied and Environmental Microbiology, 75(11), 3787–3795.
Zheng, D., Zhang, S., Zhou, X., Wang, C., Xiang, P., Zheng, Q., et al. (2012). The FgHOG1 pathway regulates hyphal growth, stress responses, and plant infection in Fusarium graminearum. PloS One, 7(11), e49495.
Acknowledgments
This work was kindly supported by the National Natural Science Foundation of China (No. 31260028 and 31300068), the Open Foundation of Key Laboratory of Tobacco Chemistry of Yunnan Province (No. fw32104132-4), and the Program for Excellent Young Talents of Yunnan University (to Jin-Kui Yang). We thank Dr. KA Borkovich (University of California, USA) for the generous gift of plasmids pCSN44, pRS426 and yeast strain FY834.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
This study was funded by the National Natural Science Foundation of China (No. 31,260,028 and 31,300,068), the Open Foundation of Key Laboratory of Tobacco Chemistry of Yunnan Province (No. fw32104132–4), and the Program for Excellent Young Talents of Yunnan University.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Yin, Z., Bi, W., Mi, Q. et al. Conserved and divergent roles of the HOG1 kinase of Alternaria longipes in mycelial and conidial development, multi-stress responses, melanin production and pathogenicity. Eur J Plant Pathol 147, 415–430 (2017). https://doi.org/10.1007/s10658-016-1012-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-016-1012-9